A Randomized Study Comparing Levofloxacin, Omeprazole, Nitazoxanide, and Doxycycline versus Triple Therapy for the Eradication of Helicobacter pylori
Am J Gastroenterol. Nov 2011; 106(11): 1970–1975.
doi: 10.1038/ajg.2011.306
Abstract
OBJECTIVES:
Resistance to standard Helicobacter pylori (HP) treatment regimens has led to unsatisfactory cure rates in HP-infected patients. This study was designed to evaluate a novel four-drug regimen (three antibiotics and a proton pump inhibitor (PPI)) for eradication of HP infection in treatment-naive patients.
METHODS:
Patients with a diagnosis of HP gastritis or peptic ulcer disease confirmed using endoscopy and stool antigen testing were eligible for inclusion in this study. All patients underwent a washout period of 6 weeks from any prior antibiotic or PPI usage. Patients were then randomized to either levofloxacin, omeprazole, nitazoxanide, and doxycycline (LOAD) therapy for 7 days (LOAD-7) or 10 days (LOAD-10), including levofloxacin 250 mg with breakfast, omeprazole 40
mg with breakfast, omeprazole 40 mg before breakfast, nitazoxanide (Alina) 500
mg before breakfast, nitazoxanide (Alina) 500 mg twice daily with meals and doxycycline 100
mg twice daily with meals and doxycycline 100 mg at dinner, or lansoprozole, amoxicillin, and clarithromycin (LAC) therapy for 10 days, which included lansoprozole 30
mg at dinner, or lansoprozole, amoxicillin, and clarithromycin (LAC) therapy for 10 days, which included lansoprozole 30 mg, amoxicillin 1
mg, amoxicillin 1 g with breakfast and dinner, and clarithromycin 500
g with breakfast and dinner, and clarithromycin 500 mg with breakfast and dinner. HP eradication was confirmed by stool antigen testing at least 4 weeks after cessation of therapy.
mg with breakfast and dinner. HP eradication was confirmed by stool antigen testing at least 4 weeks after cessation of therapy.
RESULTS:
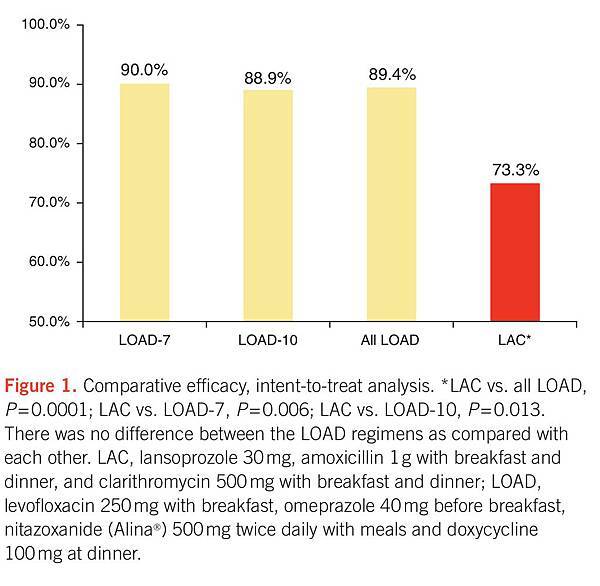
Intention-to-treat analysis revealed significant differences (P<0.05) in the respective eradication rates of the LOAD therapies (88.9% (80/90) LOAD-10, 90% (81/90) LOAD-7, 89.4% (161/180) for combined LOAD) compared with those receiving LAC, 73.3% (66/90). There were no differences in adverse effects between the groups.
CONCLUSIONS:
This open-label, prospective trial demonstrates that LOAD is a highly active regimen for the treatment of HP in treatment-naive patients. A large randomized controlled trial is warranted to further evaluate the efficacy of this regimen.
INTRODUCTION
Helicobacter pylori (HP) is a small, Gram-negative spirochete inhabiting the mucous layer overlying the gastric epithelial cells in humans. It is the most common chronic human bacterial infection and the most common cause of gastritis, with incidence rates as high as 50% worldwide (1). Furthermore, according to the World Health Organization, HP is classified as a type 1 carcinogen and is the primary cause of peptic ulcer disease, gastric carcinoma, and mucosa-associated lymphoid tissue lymphomas (1,2,3,4,5,6,7).
Current guidelines from the American College of Gastroenterology and the European Helicobacter Study Group recommend either a clarithromycin-based triple therapy (a proton pump inhibitor (PPI) plus amoxicillin and clarithromycin) or a bismuth quadruple therapy (a PPI plus bismuth, metronidazole, and tetracycline) as a standard of care in the treatment of confirmed HP infections (4,7). However, a study by Rokkas et al. (8), based on the Maastricht III guidelines, indicated that treatment with a PPI, amoxicillin, and clarithromycin regimen as first-line therapy will fail in ~30% of patients on an intention-to-treat (ITT) basis, and treatment with PPI, bismuth, metronidazole, and tetracycline as second-line therapy will fail in another 30%, leaving ~10% of the total patient population in need of an alternative regimen. Patient tolerability, adherence, and drug resistance rates play a major role in treatment selection and efficacy. Drug resistance has the greatest impact on clarithromycin therapy, with HP resistance rates ranging from 10 to 30% (2,9,10,11), and actual cure rates for PPI, amoxicillin, and clarithromycin triple therapy consistently falling below 80% (5,6,8,10,12). In contrast, global resistance rates to metronidazole have been reported to range from 25 to 66%, but some investigators believe resistance might be overcome with increased doses of metronidazole (2,4,9,10,11,12,13). Unfortunately, this increase in dosage can lead to the potential for intolerable side effects and pill burden, thus affecting treatment tolerability and adherence (4).
In response to increasing rates of resistance with the aforementioned three- and four-drug regimens, newer treatment regimens aimed at eradicating the organism more effectively are increasing in popularity (1,3,5,6,7,8,9,10,12). This study evaluates a novel four-drug regimen, three antibiotics and a PPI, for the eradication of HP infection in treatment-naive patients.
METHODS
Study patients
Consecutive patients with dyspeptic symptoms undergoing upper endoscopy were recruited for study participation. To be included in the study, patients must have had HP-induced gastritis confirmed by endoscopy (four-quadrant stomach biopsy (antro–corpus–incisura); Caris Life Sciences, Irving, TX) and monoclonal stool antigen testing (Premier Platinum HPsA PLUS, Meridian Bioscience, Cincinnati, OH). Exclusion criteria were as follows: partial gastrectomy or gastric malignancy; active bleeding; age <20 years; pregnancy; prior HP treatment or infection; recent history and treatment for Clostridium difficile infection; current use of a PPI, H2 receptor antagonist, antacid, anticoagulant, or misoprostol; recent use of antibiotics (within 6 weeks), or allergy to any medication used in the study.
Study design
A randomized, open-label trial was conducted to compare the tolerability and efficacy of a novel four-drug regimen in eradicating HP gastritis. The study compared the LOAD regimen (levofloxacin (Levaquin®; Ortho-McNeil-Janssen Pharmaceuticals, Raritan, NJ), omeprazole (Prilosec®; AstraZeneca Pharmaceuticals LP, Wilmington, DE), nitazoxanide (Alinia®; Romark Laboratories, LC, Tampa, FL), and doxycycline (Vibramycin®; Pfizer, New York, NY)) with the current standard of care as represented by the LAC regimen (lansoprazole (Prevacid®; Takeda Pharmaceuticals North America, Deerfield, IL), amoxicillin (Amoxil®; GlaxoSmithKline, Research Triangle Park, NC), and clarithromycin (Biaxin®; Abbott Laboratories, Abbott Park, IL)). Before randomization, patients underwent a washout period of 6 weeks from any antibiotic or PPI use, and provided written informed consent before the initiation of therapy. Eligible patients were then randomized to either LOAD therapy for 7 days (LOAD-7) or 10 days (LOAD-10; levofloxacin 250 mg with breakfast, omeprazole 40
mg with breakfast, omeprazole 40 mg before breakfast, nitazoxanide 500
mg before breakfast, nitazoxanide 500 mg twice daily with meals, and doxycycline 100
mg twice daily with meals, and doxycycline 100 mg at dinner) or LAC therapy for 10 days (lansoprozole 30
mg at dinner) or LAC therapy for 10 days (lansoprozole 30 mg, amoxicillin 1
mg, amoxicillin 1 g with breakfast and dinner, clarithromycin 500
g with breakfast and dinner, clarithromycin 500 mg with breakfast and dinner). Randomization was performed in blocks of three, where the assignment of treatment was drawn in a blinded manner from an envelope. Patients were instructed to return to the clinic at least 4 weeks after the cessation of therapy for repeat stool antigen testing for HP. An ad hoc analysis of those initially meeting the definition of a “cure” (HP-negative by stool antigen at least 4 weeks after the cessation of therapy) was performed. These patients were asked to return to the clinic approximately 1 year later to test for HP recurrence (recrudescence or reinfection).
mg with breakfast and dinner). Randomization was performed in blocks of three, where the assignment of treatment was drawn in a blinded manner from an envelope. Patients were instructed to return to the clinic at least 4 weeks after the cessation of therapy for repeat stool antigen testing for HP. An ad hoc analysis of those initially meeting the definition of a “cure” (HP-negative by stool antigen at least 4 weeks after the cessation of therapy) was performed. These patients were asked to return to the clinic approximately 1 year later to test for HP recurrence (recrudescence or reinfection).
Outcome assessments
The primary efficacy outcome was measured as relative eradication rates between the three regimens, at least 4 weeks after the cessation of therapy, with patient tolerability and compliance addressed as secondary end points. At the cessation of HP therapy, all anti-secretory agents were discontinued and stool antigen testing for HP was performed at least 4 weeks after therapy to evaluate for complete eradication. Usage of a monoclonal antibody stool antigen test to assess HP eradication has been validated in a large meta-analysis and was approved by the United States Food and Drug Administration (14,15). During the course of therapy, all patients were contacted by phone at least four times to assess for side effects and adherence. Helicobacter pylorirecrudescence or reinfection was assessed using a 13C–urea breath test (OAPI BreathTeK Otsuka Pharmaceutical, Rockville, MD) and stool antigen testing as previously described. Patients were instructed to discontinue any anti-secretory agent at least 2 weeks before follow-up testing.
The study protocol was approved by the institutional review board at Long Island Jewish Hospital-Forest Hills, and performed in accordance with the Declaration of Helsinki, the International Conference on Harmonisation Good Clinical Practice Guidelines, and applicable local laws and regulations. Signed informed consent was obtained from each patient before study enrollment.
RESULTS
Clinical outcomes
In total, 653 patients (N=653) were evaluated from a community referral base between February 2008 and January 2010. These patients reported symptoms of dyspepsia (n=439, 67.2%), abdominal pain (n=114, 17.5%), gastroesophageal reflux (n=89, 13.6%), early satiety (n=8, 1.2%), and loss of appetite (n=3, 0.5%). Of these patients, 270 (41.3%) were enrolled with confirmed HP gastritis or non-bleeding peptic ulcers as documented by endoscopy (four-quadrant stomach biopsies) and stool antigen testing. Overall, 278 patients (42.6%) were excluded because of recent or ongoing usage of PPI, H2 receptor antagonist, bismuth, or antacid; 79 (12.1%) patients were excluded because of recent usage of nonsteroidal anti-inflammatory drugs, Cox-II inhibitors, or misoprostol; 24 (3.7%) patients were excluded because of recent antibiotic usage; and 2 (0.3%) declined to participate. Baseline characteristics and patient demographics are further elucidated in Table 1. Approximately two-thirds of those enrolled were of Asian descent; however, there was a broad distribution of patients representing the six distinct regions of the continent.
No difference in patient adherence among LOAD-10, LOAD-7, and LAC regimens was detected. Overall, 94.4% (85/90), 96.7% (87/90), and 94.4% (85/90) of patients completed the LOAD-10, LOAD-7, and LAC regimens, respectively. In the LOAD-7 and LOAD-10 groups, eight patients discontinued therapy owing to gastrointestinal distress (n=6), dizziness (n=1), or palpitations (n=1). Five patients in the LAC arm discontinued therapy secondary to a minor skin rash (n=2) or gastrointestinal distress (n=3). Follow-up data are unavailable for one patient in the LOAD-10 arm who left the country during treatment; this patient was therefore considered to be a treatment failure. As depicted in Figure 1, ITT analysis in the patient population revealed that eradication rates were 88.9% (80/90) for LOAD-10, 90% (81/90) for LOAD-7, 89.4% (161/180) for combined LOAD regimen patients, and 73.3% (66/90) for LAC. There was no significant difference between LOAD-7 and LOAD-10 in eradication rates or adverse events. When compared with LAC in the ITT population, significant differences were detected between LOAD-7, LOAD-10, and combined LOAD therapy, P=0.013, P=0.006, and P=0.0001, respectively. Per protocol eradication rates comparing LOAD-7 and LOAD-10 patients with LAC-10 patients revealed respective cure rates of 93.6% (161/172) and 77.6% (66/85), P=0.0003 (Figure 2). As depicted in Table 2, there was no difference in reported adverse events between the groups.
 mg, amoxicillin ...
mg, amoxicillin ...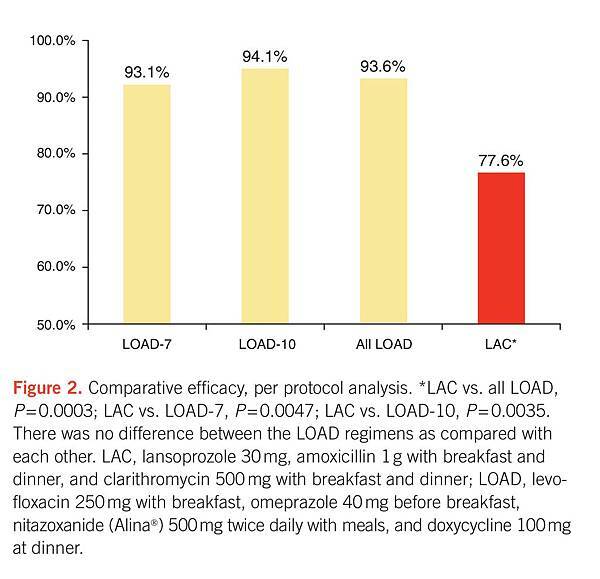
 mg, amoxicillin...
mg, amoxicillin...Although eradication of HP was of primary concern, recurrence (recrudescence or reinfection) data were collected on 218 “cures” (n=152 LOAD, n=75 LOAD-7, and n=77 LOAD-10; n=66 LAC) at 1 year (range 12 to 14 months) after the completion of therapy using stool antigen and urea breath testing. Nine patients were lost to follow-up or refused retesting. The incidence of recurrence was 11.5% (25/218) for all patients, 9.2% (14/152) for LOAD, and 16.7% (11/66) for LAC, P=0.16.
Indistinguishable symptoms of recurrence made delineating between true recrudescence and reinfection difficult. Of note, 80% (20/25) of the patients who were HP positive 1 year later had traveled out of the country to areas of high HP prevalence (Asia and Latin America). Most of these, 15/25 (60%), had traveled to Asia: Bangladesh (n=4), India (n=3), Kazakhstan (n=3), Nepal (n=2), Turkey (n=2), and Mongolia (n=1).
DISCUSSION
HP resistance to metronidazole and clarithromycin in standard therapy has led to a decline in cure rates and increased the demand for a more efficacious, yet tolerable, therapy. Although there are few regional data regarding HP resistance in our community, previous studies indicate that, in New York and the Northeast region of the United States, antibiotic resistance ranges from 9 to 15% for clarithromycin and 5 to 31% for metronidazole, and is negligible to nonexistent for amoxicillin and tetracycline (2,11). Fluoroquinolone resistance is of concern especially in areas of high fluoroquinolone use; unfortunately, regional data on HP resistance to this class of drugs are not available.
A meta-analyses including over 53,000 patients indicated the cure rate for the recommended first-line therapy is <80%, which is far below the desired 90% cure rate for any HP regimen (16,17,18). Rokkas et al. (8) highlight a greater concern—at least 10% of HP patients will fail standard first- and second-line regimens. In response to rising resistance rates, therapies including concomitant quadruple therapy and sequential therapy have emerged as alternatives to standard therapy (12,18,19,20). Although sequential therapy is innovative, the regimen is also very complex, requiring mid-therapy transition from dual to triple therapy (18,19,20). Comparatively, concomitant quadruple therapy appears to be an effective, safe, and well-tolerated alternative to triple therapy and is less complex than sequential therapy (20).
Our study indicates that a unique four-drug regimen (LOAD) is potentially more efficacious than the standard of care (LAC) for the treatment of HP in a treatment-naive population. Overall, patients receiving LAC had an unsatisfactory cure rate of 73.3% ITT (77.6% per protocol); this outcome is similar to those reported by others (4,8,21,22). On the other hand, the LOAD cure rate of 89.4% ITT (93.6% per protocol) meets the criteria that content experts deem acceptable (22,23). Furthermore, there was a trend toward fewer patients receiving LOAD (9.2%) therapy having HP recurrence 1 year later as compared with those receiving LAC therapy (16.7%). Notably, a majority of those positive for HP 1 year later had traveled out of the United States to areas of known high HP prevalence; in addition, it is possible that noncompliance and antibiotic resistance had roles in disease recurrence. However, these results are in line with a meta-analysis by Niv and Hazazi (24) indicating much higher HP recurrence rates in developing countries.
The antibiotics used in this study (levofloxacin, doxycycline, and nitazoxanide) were selected because of ease of dosing (once or twice daily) and supportive data indicating these agents are highly active against HP (25,26,27). A pilot study in patients previously failing treatment successfully used a similar regimen LEND (levofloxacin, esomeprazole, nitazoxanide, and doxycycline) to achieve a comparable ITT eradication rate of 90% (27/30) (28).
Levofloxacin, a bactericidal fluoroquinolone antibiotic, has activity against HP due primarily to the drug's activity on bacterial DNA gyrase (29). Levofloxacin has been advocated for use in second- and third-line “rescue” regimens (4,5,6,7,8,12,30,31). Unfortunately fluoroquinolone resistance, especially in patients who have routinely received a fluoroquinolone for other indications, is of particular concern (4,12,29). A lower dose of levofloxacin was used to aid in the tolerability of this four-drug regimen, however, as levofloxacin is a concentration-dependent agent, higher doses may increase eradication rates. Although the optimal dose of levofloxacin is yet to be determined, our study demonstrated efficacy using the lower dose of 250 mg once daily. Considering the LOAD regimen was well tolerated overall, an increase in levofloxacin dosage may be warranted.
mg once daily. Considering the LOAD regimen was well tolerated overall, an increase in levofloxacin dosage may be warranted.
Doxycycline, a tetracycline analog, has bacteriostatic properties through the inhibition of bacterial protein synthesis (32). The current literature indicates there is little to no resistance to this class (2,9,11), thus supporting the use of tetracyclines as superior alternatives for rescue therapies (33,34). Although tetracycline is traditionally dosed four times a day, doxycycline offers the advantages of once- or twice-daily dosing and thus potentially improving compliance and tolerability (32). In our study, LOAD therapy takes advantage of the prolonged half-life of doxycycline by using a single 100 mg dose at dinner. Dosing doxycycline only once a day may help to increase tolerability while decreasing pill burden, but, as in the case with levofloxacin, it is possible that twice-daily dosing may improve clinical outcomes.
mg dose at dinner. Dosing doxycycline only once a day may help to increase tolerability while decreasing pill burden, but, as in the case with levofloxacin, it is possible that twice-daily dosing may improve clinical outcomes.
Nitazoxanide is a thiazolide antibiotic indicated for use in adults and children for the treatment of Cryptosporidium and Giardia infections. The bactericidal activity of nitazoxanide is a result of the drug's ability to prevent anaerobic energy metabolism via inhibition of the pyruvate oxidoreductase enzyme (35). Unlike metronidazole, nitazoxanide has been shown to be non-mutagenic for HP (35) and to possess anti-vacuolating toxin activity (36). In vitro studies indicate nitazoxanide is a potent agent against HP and other anaerobes, having activity against metronidazole-resistant strains (27,36). In addition to the in vitro data, others have reported clinical success using nitazoxanide in a two-drug regimen combined with a PPI (27) or sucralfate 37 and a three-drug regimen with a PPI and amoxicillin (38). Clinical studies with nitazoxanide have used total daily doses ranging from 1,000 to 2,000 mg (27,28,37,38) when given in combination with other antibiotics, a dose of 500
mg (27,28,37,38) when given in combination with other antibiotics, a dose of 500 mg twice daily has been successful (28,38). This lower dose of nitazoxanide is generally better tolerated and helps in regard to capping the total cost of therapy.
mg twice daily has been successful (28,38). This lower dose of nitazoxanide is generally better tolerated and helps in regard to capping the total cost of therapy.
Current guidelines recommend a PPI as part of any standard therapy regimen in the treatment of HP (4,7). PPIs enhance therapy by effectively increasing gastric pH, which serves to enhance the activity of the antibiotics and disrupt the acidic environment preferred by HP (39). Omeprazole was selected for use because at the time this study was initiated, it was the only generic option. Comparisons of the available PPIs in the treatment of HP have been reviewed elsewhere and are beyond the scope of this paper (40).
Although the data indicate LOAD therapy is statistically superior to the standard of care, this study has several limitations. Patients were enrolled from a single center, thus narrowing the population size and the applicability to larger populations. Although the population studied was diverse, approximately two-thirds of the patients were of Asian descent. However, the distribution of Asian patients was quite diverse as well, with patient representation from six distinct regions of the continent (South Asia, East Asia, Southeast Asia, the Middle East, Central Asia, and Russia). This unique global distribution of patients is primarily due to our location in New York, which is convenient to many ethnically distinct neighborhoods. Presumably, symptom improvement would follow HP eradication; unfortunately, in this study patients were not assessed for symptom improvement. Secondary to constraints in funding, we were unable to perform HP susceptibility and DNA testing for antibiotic resistance and differentiation of recrudescence and reinfection. Another limitation of the study is the use of a telephone survey to assess for compliance, which is probably not optimal; non-compliance may have been associated with treatment failures and recurrence. On the other hand, this type of less-stringent follow-up may serve as an indicator of expected clinical outcomes in community practice. Finally, patients receiving therapy were not blinded, potentially leaving a margin of error due to surveillance bias.
In summary, this prospective trial demonstrates that LOAD is a highly active and well-tolerated regimen for the treatment of HP infection in treatment-naive patients. The novel LOAD regimen combines three antibiotics with a PPI, offering the advantages of good tolerability, twice-a-day dosing, low pill burden (five per day), minimal in vitro HP resistance, and superior therapeutic outcomes as compared with a standard triple-therapy regimen. A large, randomized controlled clinical trial is warranted to confirm the therapeutic superiority of this regimen as a first- or second-line treatment option over the standard recommended therapies for HP eradication.








 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 