This Journal feature begins with a case vignette highlighting a common clinical problem. Evidence supporting various strategies is then presented, followed by a review of formal guidelines, when they exist. The article ends with the author's clinical recommendations.
A
55-year-old woman with severe, persistent asthma requiring glucocorticoid
therapy for the past 3 months presents for care. Her medications include
albuterol, inhaled fluticasone with salmeterol, montelukast, and prednisone (at
a dose of 10 mg per day). In the past, she received several intermittent
courses of prednisone at a dose of 15 mg or more per day. Her weight is 45.5 kg
(100 lb), and her height 157.5 cm (62 in.); the body-mass index (the weight in
kilograms divided by the square of the height in meters) is 18. Scattered
wheezing is heard during expiration. Findings on vertebral percussion and
rib-cage compression are unremarkable. How should her case be evaluated and
managed to minimize the risk of fractures?
THE CLINICAL PROBLEM
Glucocorticoid therapy is the most common cause of secondary osteoporosis and the leading iatrogenic cause of the disease.1-3 Often, the presenting manifestation is fracture, which occurs in 30 to 50% of patients receiving long-term glucocorticoid therapy.4 Glucocorticoid-induced osteoporosis predominantly affects regions of the skeleton that have abundant cancellous bone, such as the lumbar spine and proximal femur. In patients with glucocorticoid-induced osteoporosis, the loss of bone mineral density is biphasic; it occurs rapidly (6 to 12% loss) within the first year and more slowly (approximately 3% loss yearly) thereafter.5 However, the risk of fracture escalates by as much as 75% within the first 3 months after the initiation of therapy, typically before there is a substantial decline in bone mineral density, suggesting that there are adverse effects of glucocorticoids on bone that are not captured by bone densitometry.6 Several large case–control studies have shown strong associations between exposure to glucocorticoids and the risk of fractures.4,6,7 An increase in the risk of vertebral and hip fractures occurs rapidly after the start of treatment and has been reported to occur with doses as small as 2.5 to 7.5 mg of prednisolone per day (equivalent to 3.1 to 9.3 mg of prednisone per day). In a cohort study involving patients 18 to 64 years of age, continuous treatment with 10 mg of prednisone per day for more than 90 days, for a variety of indications, as compared with no exposure to glucocorticoids, was associated with an increase in hip fractures by a factor of 7 and an increase in vertebral fractures by a factor of 17.7 Furthermore, an increase in the risk of fractures has been reported with the use of inhaled glucocorticoids, as well as with alternate-day and intermittent oral regimens.3
Risk Factors
Risk factors associated with glucocorticoid-induced osteoporosis are listed in Table 1

Risk Factors for Glucocorticoid-Induced Osteoporosis.
. One factor whose importance has been recognized in the past decade is the activity of the 11β-hydroxysteroid dehydrogenase (11β-HSD) system, a prereceptor modulator of glucocorticoid action.11 Two isoenzymes, 11β-HSD1 and 11β-HSD2, catalyze conversion between hormonally active glucocorticoids (e.g., cortisol or prednisolone) and inactive glucocorticoids (e.g., cortisone or prednisone). The 11β-HSD1 enzyme is an activator, and the 11β-HSD2 enzyme is an inactivator. The increased risk of fracture with glucocorticoid administration in the elderly may be explained in part by the increase in 11β-HSD1 that occurs with aging. The risk of glucocorticoid-induced osteoporosis appears to be similar in men and women and among various ethnic groups.13
Pathogenesis
Histomorphometric studies in patients with glucocorticoid-induced osteoporosis consistently show fewer osteoblasts and an increased prevalence of osteocyte apoptosis, as compared with normal controls1,3,4,14,15 (Figure 1
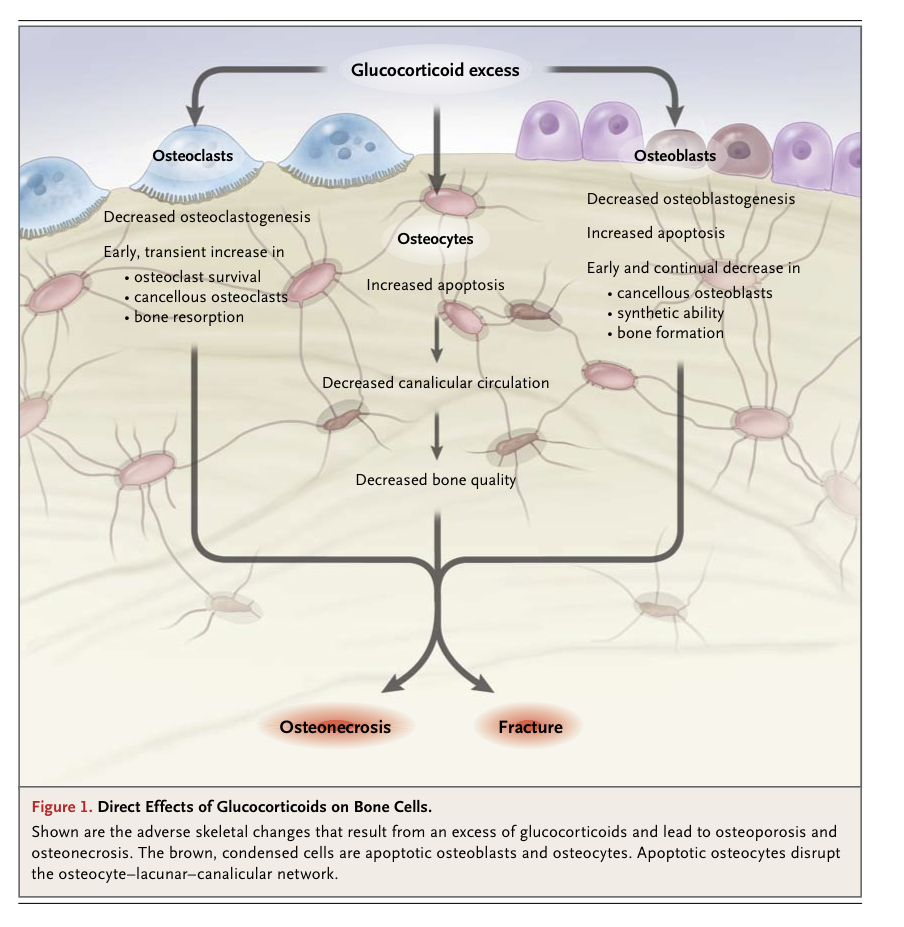
Direct Effects of Glucocorticoids on Bone Cells.
). The increased osteocyte apoptosis is associated with decreases in vascular endothelial growth factor, skeletal angiogenesis, bone interstitial fluid, and bone strength.16 Thus, glucocorticoid-induced apoptosis of osteocytes could account for the loss of bone strength that occurs before the loss of bone mineral density17 and the observed mismatch between bone mineral density and the risk of fracture in patients with glucocorticoid-induced osteoporosis.3,4, Glucocorticoid excess also directly reduces osteoclast production, but the lifespan of osteoclasts is prolonged, in contrast to the decrease in the lifespan of osteoblasts. Therefore, with long-term therapy, the number of osteoclasts is usually maintained in the normal range, whereas the number of osteoblasts plummets and bone formation is substantially reduced. 14,18 These histologic features contrast with the increased bone formation and resorption that are typical of postmenopausal osteoporosis or increased parathyroid hormone secretion and indicate that, contrary to previous assumptions, hypogonadism and secondary hyperparathyroidism are not central to the pathogenesis of glucocorticoid-induced osteoporosis.19-22
STRATEGIES AND EVIDENCE
Evaluation
Physicians who prescribe glucocorticoids should educate their patients about side effects and complications, including not only osteoporosis and osteonecrosis but also cataracts and glaucoma, hypokalemia, hyperglycemia, hypertension, hyperlipidemia, weight gain, fluid retention, susceptibility to bruising, decreased resistance to infection, impaired healing, myopathy, adrenal insufficiency, and the glucocorticoid withdrawal syndrome.3 Patients receiving long-term glucocorticoid therapy should wear medication identification jewelry. Malpractice suits precipitated by a failure of physicians to document disclosure of the skeletal complications to patients are not rare,23 yet these complications are often ignored in clinicians' discussions with patients about the use of glucocorticoids.13
Measurement of the patient's height is important, since loss of height suggests the possibility of prevalent vertebral fractures, with an associated increase in the risk of future fractures. Laboratory testing that should be performed before treatment is prescribed includes measurements of serum 25-hydroxyvitamin D, creatinine, and calcium levels (in addition to glucose, potassium, and lipid levels). Since bone turnover after long-term glucocorticoid therapy is low, tests of biochemical markers of bone metabolism are usually not helpful.4,24-27 Measurement of bone mineral density and vertebral morphologic assessment or plain films are often recommended to assess the patient for vertebral fractures, but the disparity between bone quantity and bone quality in glucocorticoid-induced osteoporosis makes measurements of bone mineral density insensitive for identifying patients at risk.2 However, measurements of bone mineral density may be useful for follow-up assessments after an intervention. The use of the World Health Organization fracture prevention algorithm (FRAX) is not recommended in the case of patients with glucocorticoid-induced osteoporosis, since it does not take into account the current and cumulative dose of glucocorticoids and the duration of therapy and underestimates the risk of glucocorticoid-induced fractures. Furthermore, bone mineral density at the femoral neck is used in the algorithm, but vertebral fractures are more common than hip fractures in patients with glucocorticoid-induced osteoporosis, and the inclusion of the common risk factors for postmenopausal osteoporosis in the algorithm may not be applicable to patients with glucocorticoid-induced osteoporosis.24
In patients treated with glucocorticoids who report persistent hip, knee, or shoulder pain, especially pain that occurs with joint movement or that is associated with tenderness or reduced range of motion, magnetic resonance imaging should be performed to rule out osteonecrosis.23 The incidence of osteonecrosis among patients who take glucocorticoids has been estimated to be between 5 and 40%; higher doses of glucocorticoids and prolonged treatment are associated with greater risk, although osteonecrosis may also occur with short-term exposure to high doses, including those administered intraarticularly (typically 40 to 80 mg of methylprednisolone) and in the absence of osteoporosis. The mechanisms that have been postulated for the development of osteonecrosis include fat embolism, vascular thrombosis, and osteocyte apoptosis.28,29
Treatment
All patients should receive adequate calcium supplementation (1200 mg per day in divided doses) and adequate vitamin D supplementation (800 to 2000 U per day), but these precautions alone are not sufficient to prevent fractures.4,24-27 Bisphosphonates are considered to be the first-line options for the treatment of glucocorticoid-induced osteoporosis (Table 2
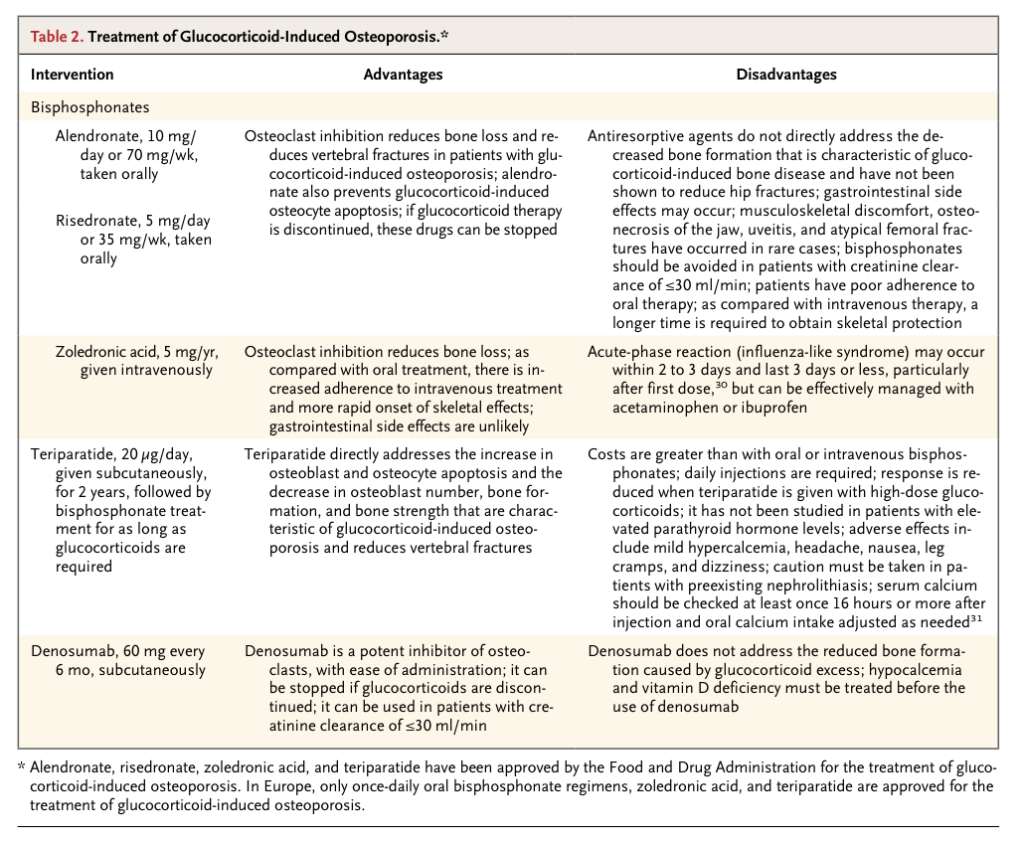
Treatment of Glucocorticoid-Induced Osteoporosis.
)4; alendronate, risedronate, and zoledronic acid are approved by the Food and Drug Administration (FDA) for this indication, although there is controversy regarding the doses and duration of glucocorticoid treatment that necessitate intervention to reduce the risk of fractures. In randomized, double-blind, placebo-controlled trials, including patients with a variety of underlying diseases and irrespective of bone mineral density, alendronate and risedronate increased bone mineral density at the lumbar spine and femoral neck and reduced the relative risk of glucocorticoid-induced vertebral fractures by about 40%4,27,32,33; patients in these trials typically had been taking 10 to 20 mg of prednisone daily or the equivalent for at least 1 year before enrollment, although the dose range and the duration of treatment varied widely. In another randomized trial involving patients treated with glucocorticoids, zoledronic acid was noninferior to risedronate in increasing bone mineral density at the lumbar spine.30
Alendronate decreases glucocorticoid-induced apoptosis of osteocytes,34 which may play a role in the preservation of bone strength.17 However, glucocorticoids antagonize the effects of nitrogen-containing bisphosphonates in inducing apoptosis of osteoclasts and inhibiting bone resorption.35,36 Perhaps as a consequence, bisphosphonates appear to be less effective in the protection of bone mineral density in patients with glucocorticoid-induced osteoporosis than they are in patients with other forms of osteoporosis. The average percentage increase in bone mineral density at the lumbar spine and femoral neck in patients with glucocorticoid-induced osteoporosis after treatment with alendronate at a dose of 10 mg per day for 2 years was 3.9% and 0.6%, respectively — considerably less than that reported in women with postmenopausal osteoporosis (about 7% and 3.6%, respectively) or in men with osteoporosis (7% and 2.5%, respectively), even though the latter two groups were, on average, 10 years older than the patients with glucocorticoid-induced osteoporosis.32,37,38 Moreover, the evidence to support the use of bisphosphonates in the treatment of patients with glucocorticoid-induced osteoporosis is not as strong as the evidence for their use in the treatment of patients with postmenopausal osteoporosis; the primary end point in the trials of glucocorticoid treatment was bone mineral density rather than the occurrence of fractures, and most trials were only 12 to 24 months in duration and were not powered to study hip fractures.
A limitation of oral bisphosphonate therapy is poor adherence to treatment — a well-recognized problem even in the case of agents that are administered weekly or monthly. Administration of zoledronic acid as a once-yearly infusion avoids this problem and provides rapid skeletal protection. For protection from fractures in patients who have received prolonged glucocorticoid therapy (e.g., 10 mg per day or more of prednisone for longer than 90 days), intravenous bisphosphonate therapy may be preferable to oral therapy. On the basis of estimates that the maximal absorption of alendronate when it is taken orally on an empty stomach is about 0.7% and that the molar potency of alendronate is lower than that of intravenous zoledronic acid by a factor of 10, it is estimated that a patient would need 90 days of treatment with alendronate at a dose of 70 mg per week to receive a dose equivalent to 5 mg of zoledronic acid delivered in 15 minutes,39 although these regimens have not been compared with respect to rates of fracture in patients with glucocorticoid-induced osteoporosis. Since substantial loss of bone mineral density has been observed in patients who discontinue bisphosphonate therapy while continuing to take glucocorticoids, it is usually recommended that bisphosphonate therapy be continued for at least as long as the glucocorticoids are prescribed3,4,27,40; drug holidays are not considered to be appropriate for patients who are being treated with glucocorticoids.
In a 2-year, randomized, controlled, open-label trial involving patients with osteonecrosis of the femoral head, patients who received alendronate therapy, as compared with those who received no treatment, had decreased pain and delayed expansion of lesions and were less likely to need surgery.41 A prospective, observational study showed that patients with osteonecrosis had a sustained reduction in pain and improvement in ambulation within months after the initiation of alendronate therapy.42 In both studies, the most common cause of osteonecrosis was the use of glucocorticoids.
Although bisphosphonates are useful in treating osteonecrosis of the hip, these drugs are associated with the development of osteonecrosis of the jaw.43 Osteonecrosis of the jaw is characterized by exposed maxillofacial bone for at least 8 weeks and typically occurs after a dental extraction or other invasive procedure.43 Most reported cases of osteonecrosis of the jaw have occurred in patients with osteolytic breast cancer or multiple myeloma who have received frequent, high doses of intravenous bisphosphonates. In patients with osteoporosis treated with bisphosphonates, the estimated risk of osteonecrosis of the jaw is 1 case per 10,000 to 100,000 patient-years.43 Before prescribing bisphosphonates, the clinician should perform an oral examination and encourage the patient to be examined by a dentist. Concurrent use of bisphosphonates and glucocorticoids may slightly increase the risk of osteonecrosis of the jaw. Bisphosphonates may also be associated with atypical subtrochanteric femoral fractures, but if there is an association, the risk is low (about 2 cases per 10,000 patient-years).44
An alternative to bisphosphonates is teriparatide, recombinant human parathyroid hormone 1-34, which is approved by the FDA for the treatment of glucocorticoid-induced osteoporosis. In an 18-month, randomized, double-blind trial comparing teriparatide with alendronate in patients with glucocorticoid-induced osteoporosis, teriparatide increased spinal bone mineral density over a shorter period and to a greater extent than did alendronate and also reduced vertebral fractures by 90%.45 Daily subcutaneous administration of parathyroid hormone prevents the expected glucocorticoid-induced increase in osteoblast and osteocyte apoptosis and decrease in osteoblast number, bone formation, and bone strength.46 However, the effect of teriparatide is somewhat compromised by high-dose glucocorticoid therapy47; a lesser increase in bone mineral density at the lumbar spine has been noted in patients taking more than 15 mg of prednisone per day, as compared with those taking less than 5 mg per day. In addition, host factors (e.g., the underlying illness and associated weight loss, medications, reduced renal function, and low levels of insulin-like growth factor I) may contribute to the diminished efficacy of teriparatide in patients with glucocorticoid-induced osteoporosis, as compared with patients who have other forms of osteoporosis.46 Disadvantages of teriparatide include the cost and the risk of mild hypercalcemia (Table 2).31
Another potential treatment option is denosumab, a humanized monoclonal antibody to the receptor activator of nuclear factor-κB ligand (RANKL), which is approved by the FDA for the prevention of vertebral, nonvertebral, and hip fractures in women with postmenopausal osteoporosis but is not currently approved for the treatment of glucocorticoid-induced osteoporosis. 48 Administered as a subcutaneous injection every 6 months, denosumab rapidly decreases bone resorption. In a subgroup analysis of a 12-month, randomized, placebo-controlled trial of denosumab in patients receiving methotrexate treatment for rheumatoid arthritis, patients receiving denosumab, prednisone (≤15 mg per day), and methotrexate had increases in bone mineral density at the lumbar spine and total hip that were similar to those in patients receiving methotrexate and denosumab alone, and the rate of side effects was similar in these groups.49 Denosumab may have a role in treating patients taking glucocorticoids who have stable serum calcium levels and who are not candidates for bisphosphonate or teriparatide therapy because of side effects or a history of renal insufficiency, although, as with other agents, more data are needed regarding the effect of denosumab on the risk of fracture.
Vertebroplasty and kyphoplasty are sometimes performed to treat painful vertebral fractures, but in controlled trials, these procedures have not been found to be superior to sham procedures, and the risks include leakage of the cement and an increase in the risk of additional fractures in patients receiving glucocorticoids.50
AREAS OF UNCERTAINTY
More data are needed to predict the risk of fractures among patients taking glucocorticoids and to establish clinical thresholds for intervention.2 Effective strategies are required to educate physicians about the importance of counseling patients who are receiving long-term glucocorticoid therapy regarding the risk of fractures. Additional studies are needed to determine the minimum dose of glucocorticoids and duration of therapy that warrant interventions to prevent fractures and to better understand the ways in which other risk factors for bone loss should guide decisions regarding therapy. Some clinicians prescribe drugs for the prevention of fractures for virtually every patient who requires glucocorticoid therapy and discontinue these drugs only when the glucocorticoids are discontinued, but the benefits, risks, and cost-effectiveness of this strategy, as compared with a more selective approach to the initiation of pharmacotherapy, have not been assessed. There is no evidence that medication to prevent fractures is needed with occasional dose-pack prescriptions, annual short-term (e.g., 7 to 10 days) high-dose intravenous or oral therapy (<1 g of cumulative exposure), or replacement therapy for patients with hypopituitarism, adrenal insufficiency, or congenital adrenal hyperplasia, provided that the replacement doses are not excessive.
GUIDELINES
Guidelines from the American College of Rheumatology, the National Osteoporosis Foundation, the Royal College of Physicians, and the Belgium Bone Club vary somewhat in their recommendations (Table 3
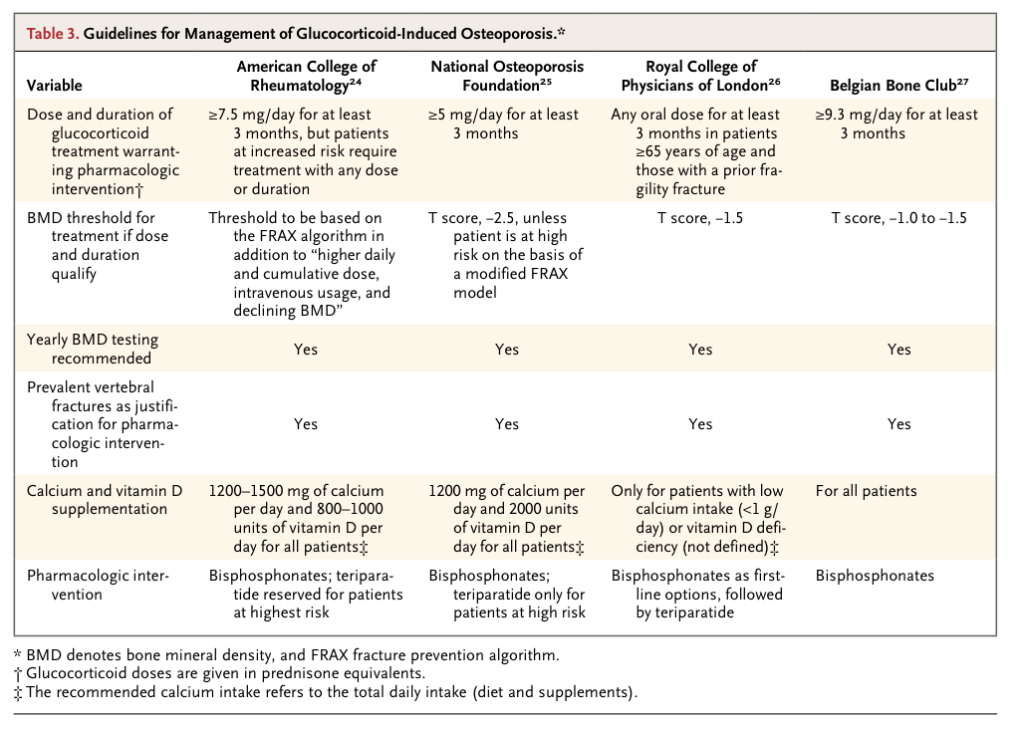
Guidelines for Management of Glucocorticoid-Induced Osteoporosis.
). 24-27 The well-recognized early increase in the risk of fracture associated with the use of glucocorticoids, the lack of certainty with respect to a known minimum dose and duration of glucocorticoid therapy that does not increase the risk of fracture, and available trials showing increased bone density (and in some cases reductions in the rate of vertebral fracture) with antiresorptive therapy in patients taking prolonged courses of glucocorticoids (mostly at doses greater than 10 to 20 mg of prednisone per day) serve as the basis of the recommendations. However, high-quality data are lacking to determine the precise risk of fractures associated with doses of prednisone that are less than 5 to 7.5 mg daily or with intermittent regimens of glucocorticoids, as well as the appropriate care of patients who are taking these regimens.
CONCLUSIONS AND RECOMMENDATIONS
The woman described in the vignette, who is slender, has been taking prednisone at a dose of 10 mg daily for 3 months, and previously received higher doses of glucocorticoids, is at considerable risk for glucocorticoid-induced osteoporosis. Other asthma therapies should be used as efficiently as possible in an effort to taper the prednisone. The assessment should include a measurement of bone mineral density, and I would also recommend a vertebral morphologic assessment or plain films to look for vertebral fractures. Adequate intake of calcium and vitamin D should be encouraged. Because of her long-term use of glucocorticoids, her age, and the low body-mass index, if prednisone cannot be discontinued (or if she has low bone mineral density or vertebral fractures), she should be advised about therapies to reduce her risk of fracture; bisphosphonates (alendronate, risedronate, and zoledronic acid) and teriparatide are approved by the FDA for these indications and should be continued for as long as the patient requires prednisone. In the absence of data from trials directly comparing the risk of fracture associated with the various therapies, the choice of medication should take into account the cost, the convenience of administration, and the side effects. The more rapid onset of action with zoledronic acid or teriparatide, as compared with oral bisphosphonates, is a potential advantage of these medications, but that advantage must be weighed against the greater costs of those drugs and, in the case of teriparatide, the need for daily injections.





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 