這不是一般避孕藥,這是事後的,不可以拿來當做一般避孕用,如果你想多知道一點一般避孕藥,可以參考這邊:常見避孕藥比較
在你使用之前,我還是要再次提醒你,使用避孕藥是有風險的,不管是對身體健康還是對於避孕效果,都是有一定度的風險在,請在進行性行為之前,審慎評估。
今天的主角:
他有兩種包裝:



主成分就是:30 mg ulipristal acetate.
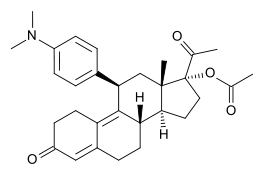
適應症為:
Emergency contraception within 120 hours (5 days) of unprotected sexual intercourse or contraceptive failure.
其他注意事項:
Concomitant use with an emergency contraceptive containing levonorgestrel is not recommended (看下一段).
Use in women with severe asthma insufficiently controlled by oral glucocorticoid is not recommended.
Emergency contraception with ellaOne is an occasional method. It should in no instance replace a regular contraceptive method. In any case, women should be advised to adopt a regular method of contraception.
Although the use of ellaOne does not contraindicate the continued use of regular hormonal contraception, ellaOne may reduce its contraceptive action (看下一段). Therefore, after using emergency contraception, it is recommended that subsequent acts of intercourse be protected by a reliable barrier method until the next menstrual period starts.
Repeated administration of ellaOne within the same menstrual cycle is not advisable, as safety and efficacy of ellaOne after repeated administration within the same menstrual cycle has not been investigated.
Emergency contraception with ellaOne does not prevent pregnancy in every case. No data is available on the efficacy of ellaOne for women who have had unprotected intercourse more than 120 hours before ellaOne intake. In case of doubt, delay of more than 7 days in next menstrual period, abnormal bleeding at the expected date of menses, or symptoms of pregnancy, pregnancy should be excluded by a pregnancy test.
If pregnancy occurs after treatment with ellaOne, as for all pregnancies, the possibility of an ectopic pregnancy should be considered. Ectopic pregnancy may continue, despite the occurrence of uterine bleeding.
After ellaOne intake menstrual periods can sometimes occur earlier or later than expected by a few days. In approximately 7% of the women, menstrual periods occurred more than 7 days earlier than expected. In 18.5% of the women a delay of more than 7 days occurred, and in 4% the delay was greater than 20 days.
This medicinal product contains lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine.
交互作用(Interaction with other medicinal products and other forms of interaction):
Ulipristal acetate is metabolized by CYP3A4 in vitro. No specific drug interaction studies have been performed in vivo.
• Potential for other medicinal products to affect ulipristal acetate:
CYP3A4 inducers (e.g. rifampicin, phenytoin, phenobarbital, carbamazepine, ritonavir, St John's wort/Hypericum perforatum) may reduce plasma concentrations of ulipristal acetate and may result in decrease in efficacy. Concomitant use is therefore not recommended. Enzyme induction wears off slowly and effects on the plasma concentrations of ulipristal acetate may occur even if a woman has stopped taking an enzyme inducer within the last 2![]() 3 weeks.
3 weeks.
Concomitant administration of medicinal products that increase gastric pH (e.g. proton pump inhibitors, antacids and H2-receptor antagonists) may reduce plasma concentrations of ulipristal acetate and may result in decrease in efficacy. Concomitant use is therefore not recommended.
Potent CYP3A4 inhibitors (e.g. ketoconazole, itraconazole, telithromycin, clarithromycin, nefazodone) may increase exposure to ulipristal acetate. The clinical relevance is unknown.
• Potential for ulipristal acetate to affect other medicinal products:
Because ulipristal acetate binds the progesterone receptor with high affinity, it may interfere with the action of progestogen-containing medicinal products:
- Contraceptive action of combined hormonal contraceptives and progestogen-only contraception may be reduced
- Concomitant use of ulipristal acetate and emergency contraception containing levonorgestrel is not recommended.
其他賦形劑:
Povidone K30 Croscarmellose sodium Magnesium stearate
Lactose monohydrate





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 