The US Food and Drug Administration (FDA) has approved the use of belimumab (Benlysta, Human Genome Sciences and GlaxoSmithKline) in combination with standard therapies to treat active autoantibody-positive systematic lupus erythematosus.
This is the first lupus drug to be approved since 1955, when the FDA approved hydroxychloroquine (Plaquenil) and corticosteroids. In 1948, aspirin was approved to treat lupus.
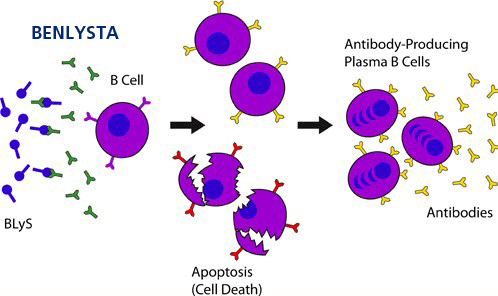
Belimumab is a B-lymphocyte stimulator protein inhibitor that is thought to decrease the amount of abnormal B cells, which is hypothesized to be a mechanism of action in lupus.
The safety and effectiveness of belimumab was demonstrated in 2 clinical trials that randomized a total of 1684 patients to receive either belimumab or placebo in combination with standard therapy. Treatment with belimumab plus standard therapy reduced disease activity and possibly decreased the number of severe flares and steroid use.
Patients with active lupus that involved the kidneys or central nervous system and those who were previously treated with a B-cell-targeted therapy or intravenous cyclophosphamide were excluded from participating in the trials.
Study participants of African American or African descent did not significantly respond to belimumab. Additional studies will be conducted to definitively determine the safety and efficacy of belimumab in this population.
Common adverse effects reported with belimumab include nausea, diarrhea, fever, and infusion-site reactions. It is suggested that patients be treated with an antihistamine prior to a belimumab infusion.
A greater number of deaths and serious infections were reported in patients treated with belimumab than in those treated with placebo. Live vaccines should not be administered during treatment with belimumab.
It is estimated that lupus afflicts up to 1.5 million Americans, and it disproportionately affects black women.





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 