High-dose docosahexaenoic acid (DHA) supplementation in preterm infants is associated with lower risk for bronchopulmonary dysplasia (BPD) in the smallest infants and in male infants, according to the results of a multicenter, randomized controlled trial published online June 27 in Pediatrics.
(http://trialx.com/curebyte/2011/07/06/images-related-to-bronchopulmonary-dysplasia/)
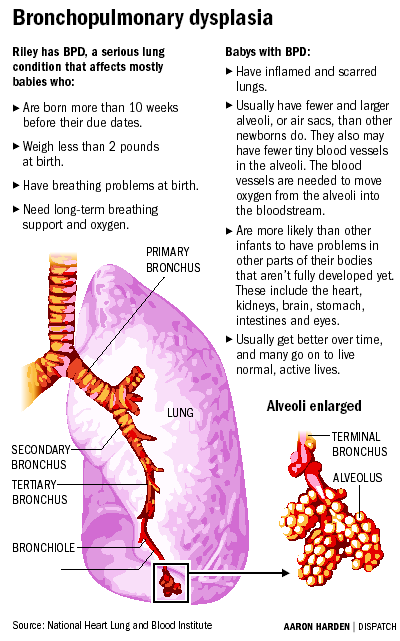
"Very preterm infants are at risk of [BPD] and are more likely to suffer from atopic conditions in later life," write Brett J. Manley, MBBS, from the Department of Newborn Research, Royal Women's Hospital in Victoria, Australia, and colleagues from the Docosahexaenoic acid for the Improvement in Neurodevelopmental Outcome (DINO) Steering Committee. "[DHA] is known to modulate inflammation and is postulated to modulate the neonatal immune response."
The study goal was to investigate the effect of DHA supplementation on long-term respiratory and atopic outcomes in preterm infants with gestational age younger than 33 weeks. Infants received expressed breast milk from their mothers, who were randomly assigned to receive supplementation with tuna oil capsules (high-DHA diet) or soy oil capsules (standard-DHA diet). During the first 18 months of life, incidence of BPD and parental reports of atopic conditions were recorded (follow-up rate, 93.5%).
Of 657 infants enrolled, 322 were randomly assigned to the high-DHA diet and 335 to the standard-DHA diet. The high-DHA diet was associated with a reduction in relative risk (RR) in BPD in boys (RR, 0.67; 95% confidence interval [CI], 0.47 - 0.96; P = .03) and in both boys and girls with birth weight lower than 1250 g (RR, 0.75; 95% CI, 0.57 - 0.98; P = .04). However, DHA supplementation was not associated with duration of respiratory support or of hospitalization, or with requirement for home oxygen.
The high-DHA group had lower risk for reported hay fever in all infants at either 12 or 18 months (RR, 0.41; 95% CI, 0.18 - 0.91; P =.03), and at either 12 or 18 months in boys (RR, 0.15; 95% CI, 0.03 - 0.64; P = .01). High DHA did not appear to have any protective effect on development of asthma, eczema, or food allergy.
"DHA supplementation for infants of <33 weeks' gestation reduced the incidence of BPD in boys and in all infants with a birth weight of <1250 g and reduced the incidence of reported hay fever in boys at either 12 or 18 months," the study authors write.
Limitations of this study include its reliance on parental recall for allergy outcomes.
"These results add weight to the argument for high-dose DHA supplementation in the most at-risk premature infants, and support previous evidence that DHA may have a role in reducing atopic conditions," the study authors conclude. "Future studies should be powered to achieve a difference in BPD incidence in DHA-supplemented infants. The optimal dose of DHA is uncertain, and although the dose used in the DINO trial should be compared with a higher dose strategy, any products designed for neonates born at <1250 g should contain at least 1% of the total fats as DHA."
The DINO trial was supported by a grant from the National Health and Medical Research Council of Australia. Treatment. Placebo capsules were donated by Clover Corporation, and infant formula was donated by Mead Johnson Nutritionals and Nutricia Australia. Some of the study authors report various financial relationships with Nestle, Fonterra, and/or Nutricia.
Pediatrics. Published online June 27, 2011. Abstract
根據線上刊載於6月27日小兒科(Pediatrics)期刊的多中心隨機控制試驗的結果,高劑量二十二碳六烯酸(DHA)補充品用於體型小、男性早產兒與降低肺支氣管發育不良(BPD)風險有關。
澳洲皇家婦女醫院新生兒研究部Brett J. Manley醫師與「二十二碳六烯酸改善神經發育結果(Docosahexaenoic acid for the Improvement in Neurodevelopmental Outcome,DINO)」指導委員會的同僚寫道,極早產嬰兒有BPD風險,爾後會比較可能發生特異性狀況;已知DHA可調節發炎,想當然可以調節新生兒的免疫反應。
研究目標是探討DHA補充品對於妊娠年齡33週以下早產兒的長期呼吸道與特異性結果;嬰兒們喝母奶,母親們被隨機指派接受鮪魚油膠囊補充品(高DHA飲食)或大豆油膠囊(標準DHA飲食);在出生後18個月內,紀錄BPD發生率和家長報告的特異性狀況(追蹤率為93.5%)。
納入的657名嬰兒中,322人被隨機指派接受高DHA飲食、335人為標準DHA飲食,高DHA飲食和男孩的BPD相對風險(RR)降低有關(RR為0.67;95%信心區間[CI]為0.47 - 0.96;P = .03),體重低於1250 g的男孩和女孩也是(RR為0.75;95% CI為0.57 - 0.98;P = .04),不過,DHA補充品和使用呼吸支持期間、住院期間、居家照護需要氧氣等無關。
高DHA組嬰兒不論在12或18個月時的枯草熱(花粉症/hay fever)風險較低(RR為0.41;95% CI為0.18 - 0.91;P =.03),以及12或18個月的男孩(RR為0.15;95% CI為0.03 - 0.64;P = .01),高DHA對於發生氣喘、濕疹或食物過敏並沒有顯示任何保護效果。
研究作者寫道,妊娠年齡33週以下嬰兒接受DHA補充品可降低男孩和所有體重小於1250 g嬰兒的BPD風險,並降低12或18個月男孩的枯草熱發生率。
研究限制包括僅依賴家長有關過敏結果的回憶。
研究作者結論表示,這些結果佐證高劑量DHA補充品對於多數有風險的早產兒很重要,也支持DHA可減輕特異性狀況的證據,未來的研究應加強探討DHA補充品對於嬰兒BPD發生率的差異;DHA的適當劑量還不清楚,不過DINO試驗所用的劑量偏向高劑量策略,指定用於體重小於1250 g新生兒之任何產品的脂肪應包括至少1%的DHA。
資料來源:http://www.24drs.com/professional/list/content.asp?x_idno=6566&x_classno=0&x_chkdelpoint=Y





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 