先了解一下端粒是啥:
我們都知道複製的動物壽命都不長,以桃莉羊為例,原本一隻正常健康的羊可以活 15年,但是複製羊卻只能活5年,這是為什麼呢?原因可能在於我們生物的染色體尾端有一段與其他部分不同的序列(稱之為端粒,Telomere),只要每經過一次染色體複製分裂長度就會變短,但是人體內又有一種端粒酶可以修復端粒的長度,而端粒酶在兒童及年輕人身上尤其常見,不過隨著年紀的增長端粒酶的修復功能會逐漸減弱,端粒的長度也隨之變短,等到端粒的長度已經短到一個程度時,就代表死亡已經接近。
資料來源:NEJM Volume 361:2353-2365
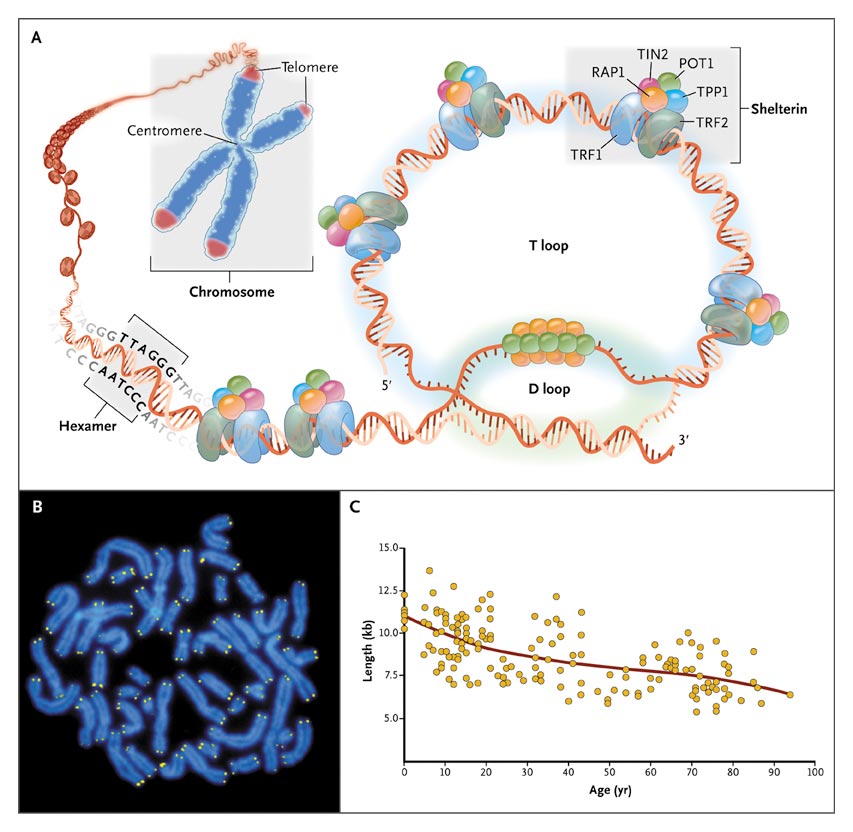
Telomere Structure.
As shown in Panel A, telomeres are located at the ends of linear chromosomes; they are composed of hundreds to thousands of tandem DNA repeat sequences: hexameric TTAGGG in the leading strand and CCCTAA in the lagging strand in humans. Protective proteins associated with telomere DNA are collectively termed shelterin (TRF1, TRF2, TIN2, POT1, TPP1, and RAP1). The 3' end of the telomeric leading strand terminates as a single-stranded overhang, which folds back and invades the double-stranded telomeric helix, forming the T loop. As shown in Panel B, telomeres can be directly visualized under the microscope at the ends of metaphase chromosomes (four telomere signals per chromosome) by fluorescence in situ hybridization (FISH). (Image provided by Peter Lansdorp, M.D., Ph.D.) Average telomere length can be measured by several methods: a technique that combines flow cytometry and FISH (flow-FISH), Southern blotting, and a quantitative polymerase-chain-reaction (qPCR) assay. Flow-FISH can measure the telomere length in different cell subgroups, such as granulocytes or CD4+ T lymphocytes; Southern blotting reveals length and length heterogeneity; and qPCR is a rapid assay that requires very small amounts of DNA. As shown in Panel C, the average length of telomeres in human leukocytes varies, ranging from approximately 11 kb at birth (in umbilical-cord blood) to 6 kb at 90 years of age. Telomere loss is most rapid early in life, and over a life span it is not linear but follows a third-order polynomial. Data are from Yamaguchi et al.
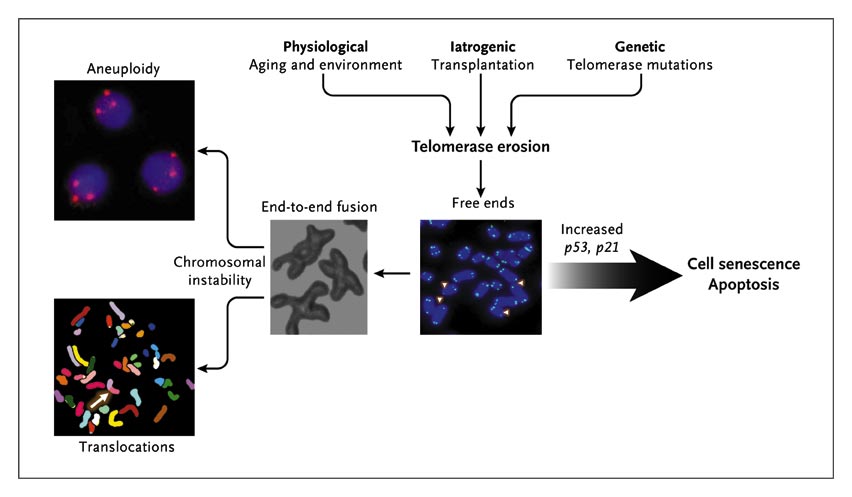
Consequences of Telomere Erosion in the Cell.
Telomeres inexorably shorten with every cell division, and telomere attrition is an inevitable physiological consequence of aging. Telomere shortening also may be iatrogenic; for example, telomere shortening occurs after bone marrow transplantation, in which highly proliferative hematopoietic stem cells and progenitor cells reconstitute hematopoiesis. Environmental factors also may accelerate telomere loss. In addition, telomere attrition may be genetic; there may be an inherited inability to elongate telomeres as a result of mutations in components of the telomerase complex. When telomeres become critically short, inappropriately capped chromosomes or telomere-free ends emerge, which lead to cell senescence or apoptosis. If the cell overrides senescence and continues to proliferate (e.g., because of inactive p53), uncapped telomeres may cause end-to-end fusion of chromosomes, breakage-fusion-bridge cycles, aneuploidy, and chromosomal translocations.
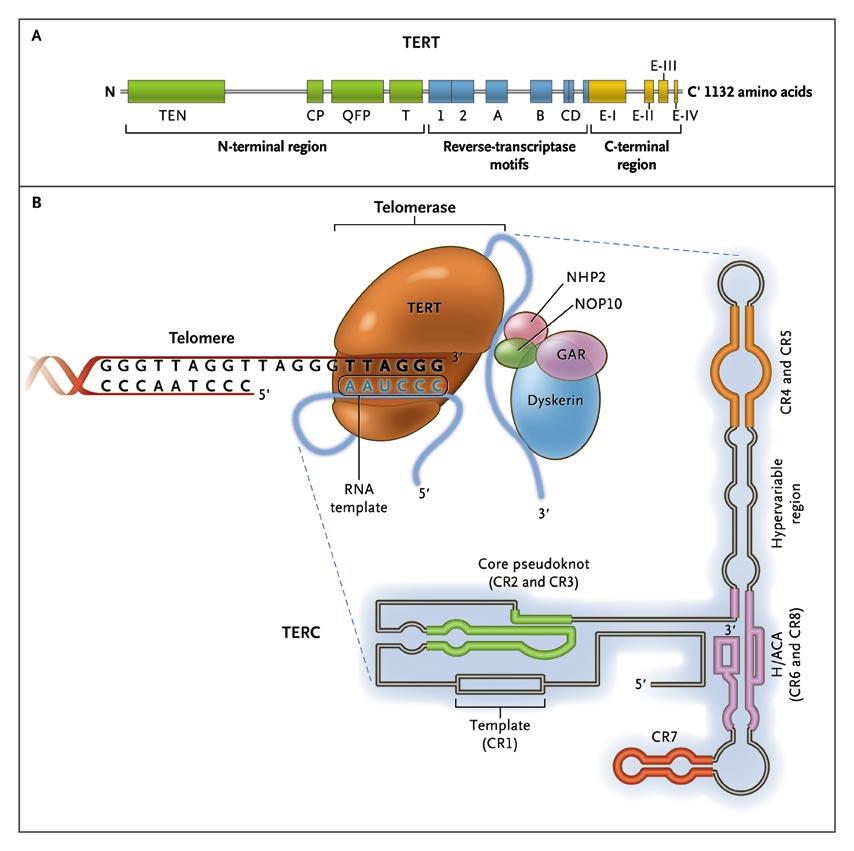
The Telomerase Complex and Its Components.
The enzyme telomerase reverse transcriptase (TERT), its RNA component (TERC), the protein dyskerin, and other associated proteins (NHP2, NOP10, and GAR1) are shown. Telomerase catalytically adds TTAGGG hexameric nucleotide repeats to the 3'-hydroxyl end of the telomeric leading strand, using a specific sequence in the RNA component as the template. TERT contains three major domains: the N-terminal region, the reverse-transcriptase motifs, and the C-terminal region, all containing evolutionarily conserved motifs. TERC contains 451 nucleotides in seven conserved regions (CR1 through CR7), including the template (CR1), and an H/ACA box, a hairpin nucleotide sequence characteristic of a class of small nucleolar RNAs involved in RNA processing.
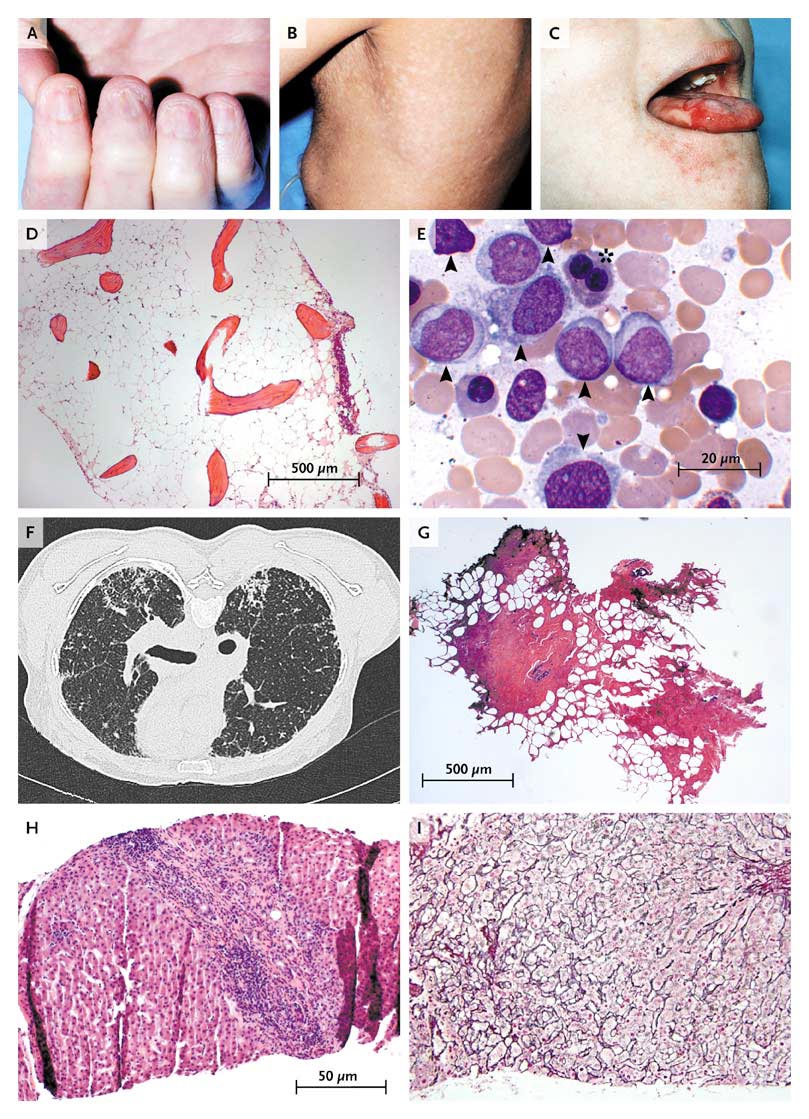
Pathologic Consequences of Telomere Erosion in Organs and Tissues.
The childhood syndrome dyskeratosis congenita is characterized clinically by the mucocutaneous triad of nail dystrophy (Panel A), reticular skin hyperpigmentation or hypopigmentation (Panel B), and leukoplakia (Panel C) (reprinted from Alter,13 with the permission of the publisher). In the bone marrow, telomere shortening confers a predisposition to aplastic anemia (Panel D, Giemsa stain) and progression to myelodysplasia and acute myeloid leukemia (Panel E, Giemsa stain). Leukemic bone marrow is characterized by an increased number of myeloid blasts (arrowheads) and dysplasia or dyserythropoiesis (asterisk). In the lungs, telomere attrition can be clinically manifested as pulmonary fibrosis and radiologically characterized by diffuse fibrosis predominantly in the subpleural region (Panel F). Histologically, fibrotic zones alternate with less affected parenchyma (Panel G, hematoxylin and eosin). Telomere shortening in the liver has diverse histologic appearances, including cirrhosis with inflammation (Panel H, hematoxylin and eosin) and nodular regenerative hyperplasia (Panel I, reticulin stain).
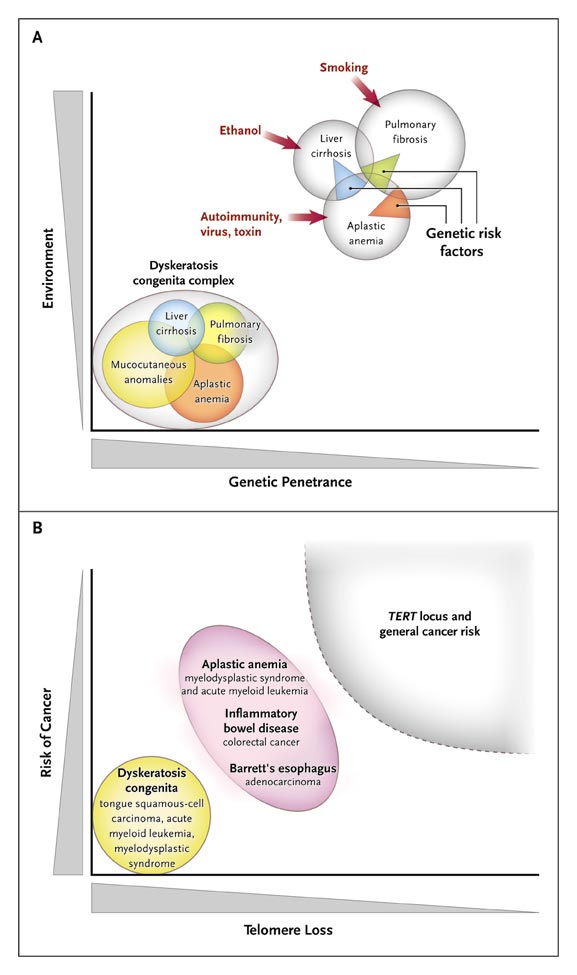
Telomere Erosion and Human Diseases.
Panel A shows a Venn diagram of mutations of the telomerase complex and human telomere diseases. Dyskeratosis congenita is the most evident and severe manifestation of genetic lesions causing telomere diseases, with high genetic penetrance and congenital clinical manifestations. However, telomerase mutations may be less penetrant and induce single-organ damage in adults without suggestive family histories and the classic physical signs of dyskeratosis congenita. Thus, telomerase mutations represent risk factors rather than genetic determinants in aplastic anemia, pulmonary fibrosis, and liver cirrhosis. Environmental, epigenetic, and other genetic factors probably contribute to disease development in these patients. Panel B shows the relationship between telomere shortening and the risk of cancer. In dyskeratosis congenita, in which genetic penetrance is high, the risk of the development of cancer — particularly head and neck squamous-cell carcinoma and acute myeloid leukemia — also is elevated. In addition, patients with aplastic anemia are at risk for the development of clonal malignant disorders, but the risk is lower than that among patients with dyskeratosis congenita. Similarly, short telomeres appear to predict the progression of chronic inflammatory gastrointestinal states to adenocarcinoma. In multiple genomewide association studies, the TERT locus has appeared as a significant susceptibility locus for a variety of cancers, but at relatively low odds ratios. Shaded areas representing diseases and disease states are not drawn to scale.





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 