N Engl J Med 2012; 367:11-19July 5, 2012
Severe aplastic anemia is an acquired bone marrow disease characterized by trilineage marrow hypoplasia and a paucity of hematopoietic stem and progenitor cells due to an autoimmune attack on the bone marrow.1 The standard treatment for aplastic anemia is immunosuppressive therapy with horse antithymocyte globulin (ATG) and cyclosporine, and hematologic responses are observed in about two thirds of patients.2 Patients with disease that is refractory to immunosuppression and those who have a relapse after treatment may undergo allogeneic hematopoietic stem-cell transplantation (HSCT). However, 20 to 40% of patients without a suitable donor for HSCT continue to have severe cytopenias and are at risk for life-threatening hemorrhage due to thrombocytopenia and severe infections due to neutropenia.3
No standard therapies are available for patients who have aplastic anemia that is refractory to immunosuppression and are ineligible for HSCT, other than transfusions and treatment of infections. More than 40% of patients with disease that is refractory to immunosuppression die from bleeding or infection within 5 years after diagnosis.4 Although readministration of immunosuppressive therapy has been effective as salvage therapy in some patients, intensification of the regimen with more potent agents, such as rabbit ATG, sirolimus, or mycophenolate, has not improved the response rate.1,5
Thrombopoietin is the principal regulator of platelet production through binding of the receptor c-MPL on megakaryocytes, which results in platelet maturation and release.6 Hematopoietic stem cells and progenitor cells also express c-MPL on their cell surface,7 and the addition of recombinant thrombopoietin expands the pool of hematopoietic stem cells in culture.7 Knockout mice that are deficient in expression of the thrombopoietin receptor mpl, 8,9 the thrombopoietin ligand,10 or both have reduced numbers of hematopoietic stem and progenitor cells, and multilineage marrow failure eventually develops in patients with congenital amegakaryocytic thrombocytopenia who have MPLmutations.11 These observations suggest that stimulation of c-MPL–signaling pathways may overcome depletion of hematopoietic stem and progenitor cells in aplastic anemia.
Eltrombopag (Promacta) is an oral thrombopoietin mimetic that binds to c-MPL, promoting megakaryopoiesis and release of platelets from mature megakaryocytes.12 Eltrombopag increases platelet counts in healthy persons and is approved by the Food and Drug Administration for treatment of patients with chronic immune thrombocytopenic purpura.12 We hypothesized that eltrombopag might have activity in patients with aplastic anemia who continue to have severe thrombocytopenia after not having a response to one or more cycles of immunosuppression.
METHODS
Study Participants and Oversight
This trial, which was sponsored by the National Institutes of Health (NIH), was an investigator-initiated, nonrandomized, phase 2 study of eltrombopag in patients with aplastic anemia and severe persistent thrombocytopenia after immunosuppression. The study was approved by an NIH institutional review board and was monitored by an NIH data and safety monitoring board. Written informed consent was obtained from all patients. Eltrombopag was provided free of charge by GlaxoSmithKline, which played no role in the study design, data collection or analysis, or writing of the manuscript. No one who is not listed as an author contributed to the writing of the manuscript. All authors had full and independent access to all the data and vouch for the accuracy and completeness of the reported data and the fidelity of the study to the protocol. The study design is shown in Fig. 1S in the Supplementary Appendix, available with the full text of this article at NEJM.org. The study was conducted in accordance with the protocol, which is also available at NEJM.org.
Adults with a diagnosis of severe aplastic anemia, defined according to standard criteria,13 and thrombocytopenia (platelet count, ≤30,000 per cubic millimeter) that persisted after one or more courses of treatment with horse or rabbit ATG and cyclosporine were eligible to participate in the study. Exclusion criteria are described in Table 1S in the Supplementary Appendix. The most recent treatment with ATG and cyclosporine had to have been initiated more than 6 months before study enrollment.
Intervention
Consecutive eligible patients received eltrombopag at an initial oral dose of 50 mg once daily. If, after 2 weeks, the platelet count had not increased by 20,000 per cubic millimeter from baseline or platelet-transfusion requirements had not decreased, the dose was increased by 25 mg every 2 weeks to a maximum of 150 mg. Patients received transfusions as supportive care to maintain platelets at a level above 10,000 cells per cubic millimeter and hemoglobin at a level above 10 g per deciliter. Bone marrow morphologic features were monitored and metaphase cytogenetic analysis was performed at entry and at 3 months in all patients and then every 6 months in patients who had a response (Table 2S in the Supplementary Appendix).
End Points
The primary end points were defined according to hematologic responses and toxic effects at 12 weeks. A platelet response was defined as an increase of 20,000 cells per cubic millimeter or more above the baseline value, or independence from platelet transfusions for a minimum of 8 weeks in patients who were previously transfusion-dependent. An erythroid response in patients with a pretreatment hemoglobin level of less than 9 g per deciliter was defined as an increase in the hemoglobin level by 1.5 g per deciliter or more without transfusion of packed red cells or a reduction in the number of units of packed red cells transfused by at least 4 units for 8 consecutive weeks, as compared with transfusion requirements during the 8 weeks preceding study entry. A neutrophil response was defined as an absolute increase in the neutrophil count of more than 500 per cubic millimeter; in patients with a pretreatment count of less than 500 per cubic millimeter, a response was defined as an increase of more than 500 per cubic millimeter or at least a 100% increase over the baseline neutrophil count.
Patients who met response criteria for one or more lineages at 12 weeks were deemed to have had a response. Patients who met criteria for a response at 12 weeks continued to receive eltrombopag for an additional 4 weeks to ensure the stability of the response and then could continue to receive eltrombopag for as long as the response was maintained. Hematologic responses in additional lineages were assessed in patients who had a response and who participated in the extension study. Toxic effects were measured with the use of the National Cancer Institute Common Terminology Criteria for Adverse Events (version 3) (http://ctep.cancer.gov/reporting/ctc.html).
Secondary end points were changes in blood counts measured as continuous variables, plasma thrombopoietin levels, and bone marrow cellularity; morphologic characteristics; metaphase cytogenetic profile; and reticulin fibrosis. Reticulin was graded according to standard guidelines.14Plasma cytokine levels, telomere length, and the immunophenotype of peripheral-blood cells were measured as described previously.2,15,16
Statistical Analysis
We used the two-stage “minimax” design described by Simon,17 with a response probability of 10% or less as the null hypothesis and a response probability of 30% or more as the alternative hypothesis. The sample size of 25 patients was determined by testing the null hypothesis (response probability, ≤10%) versus the alternative hypothesis (response probability, ≥30%) at a significance level of 0.05 and a power of 80%. The null hypothesis would be accepted if the number of patients with a response at 12 weeks was 5 or less. A Bayesian stopping rule for safety was applied on the basis of the number of patients in whom possible treatment-related severe adverse events developed, including death, grade 4 toxic effects, and thrombosis or pulmonary embolism.18Summary statistics were used to describe the characteristics of the patients, baseline variables, and treatment responses. Data analysis was performed with the use of S-Plus software (TIBCO).
RESULTS
Characteristics of the Patients
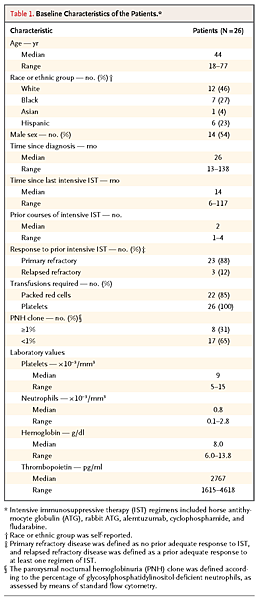
Between 2009 and 2011, we screened 47 patients with aplastic anemia and refractory cytopenia for eligibility; 26 were enrolled, and 25 received eltrombopag. In 1 patient, the diagnosis was changed from aplastic anemia to hypocellular myelodysplastic syndrome after study entry but before treatment initiation. The characteristics of the patients are listed in Table 1
TABLE 1Baseline Characteristics of the Patients.. One patient received granulocyte colony-stimulating factor (G-CSF) briefly during the study because of fever; therefore, the neutrophil lineage was excluded from the response assessment in this patient. One patient had a neutrophil response to cyclosporine before enrollment, despite persistent thrombocytopenia, and continued to receive the same dose of cyclosporine throughout the study. None of the other patients received immunosuppressive or cytokine therapy for at least 1 month before enrollment.
Adverse Events and Toxic Effects
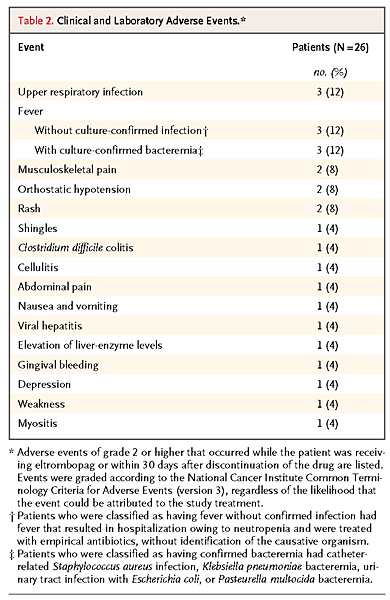
All but one patient received the maximum dose of 150 mg per day. Patients were asked about side effects at each visit, and they also participated in a quality-of-life survey that assessed symptoms during treatment. They did not find the side effects of eltrombopag limiting. Table 2
TABLE 2Clinical and Laboratory Adverse Events. lists adverse events of grade 2 or higher. Severe adverse events resulting in hospitalization included the following: abdominal pain and orthostatic hypotension in a patient with diabetic gastroparesis, in whom the drug was discontinued at the patient's request; a severe rash that was temporally related to the initiation of treatment with cephalosporin; severe gingival bleeding; and episodes of fever with neutropenia, some of which were linked to culture-confirmed infection. In our cohort, fever and recurrent infections, which are common in patients with aplastic anemia, occurred only in patients who did not have a response. One patient acquired hepatitis B virus infection during the study, with elevation of aminotransferase levels, and eltrombopag was discontinued at week 8. Diagnosis of a cataract in one patient at week 9, during examination for a “floater,” prompted the discontinuation of eltrombopag, although the patient met hematologic response criteria. The cataract could not be confirmed on subsequent examinations. One patient with a response discontinued eltrombopag at the response assessment at 12 weeks because of an increase in glycosylphosphatidylinositol (GPI)–deficient neutrophils from 40% to 57%, which met the exclusion criterion of more than 50%; the patient had no evidence of hemolysis or other signs of paroxysmal nocturnal hemoglobinuria.
Previous studies have aroused concern that chronic c-MPL stimulation may cause bone marrow fibrosis.19 Among 23 patients assessed, no increase in fibrosis or reticulin was noted on bone marrow–biopsy specimens obtained at 3 months, or on subsequent specimens obtained every 6 months from patients who had a response and continued to receive eltrombopag for up to 30 months. In these patients, the reticulin scores were 1+ or 0 (on a scale of 0 to 4+, with higher scores indicating myelofibrosis) (Fig. 2S in the Supplementary Appendix).
Clinical Response
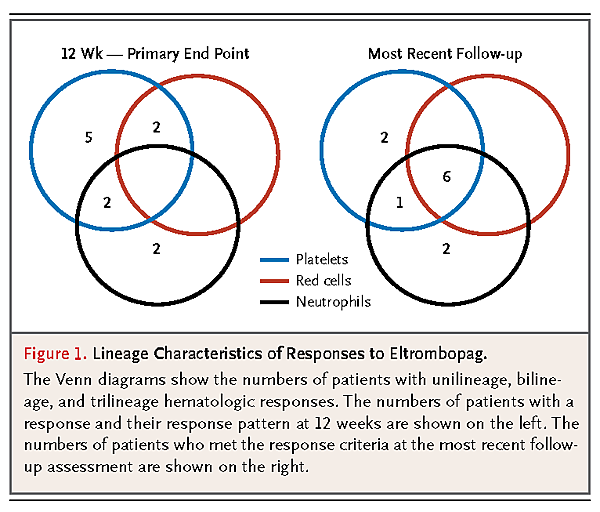
Eleven of 25 patients who received eltrombopag (44%) met primary response criteria in at least one lineage 12 weeks after the initiation of treatment. All 25 patients were dependent on platelet transfusions at enrollment, and 9 of the 11 patients with a response no longer required platelet transfusions at 12 weeks, including 2 patients who also had hemoglobin responses and 2 who also had neutrophil responses. The 2 remaining patients who had a response met only the criteria for a neutrophil response at 12 weeks (Figure 1
FIGURE 1Lineage Characteristics of Responses to Eltrombopag.).
Seven of 11 patients with a response continued to receive eltrombopag (at a dose of 150 mg per day) for a median of 16 months (range, 8 to 32). Of the 4 patients who did not continue to receive eltrombopag, 2 had only a neutrophil response, and 2 discontinued the drug because of possible toxic effects as described above. Patients with a response continued to have clinically significant improvements in blood counts, and some had responses in additional lineages; 1 patient eventually met response criteria in two lineages, and 6 patients eventually met response criteria in three lineages (Figure 1).
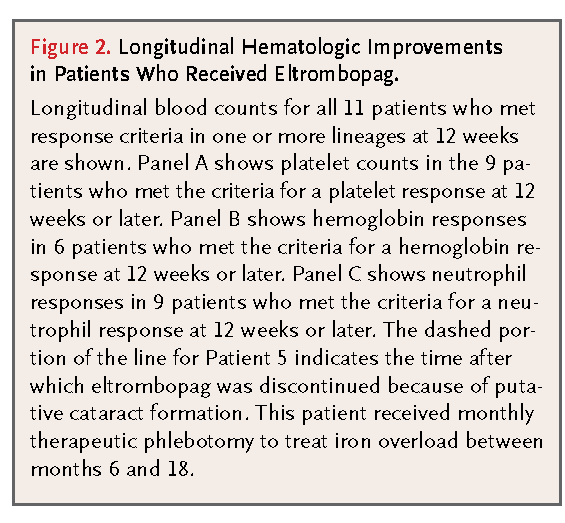
FIGURE 2Longitudinal Hematologic Improvements in Patients Who Received Eltrombopag. shows longitudinal measurements of blood counts in patients for each lineage with a response. A total of nine patients had a platelet response, with a median increase in the platelet count of 44,000 per cubic millimeter at longest follow-up (Figure 2A). A total of six patients had erythroid responses (Figure 2B), with a median increase in the hemoglobin level of 4.4 g per deciliter. Three patients who were previously dependent on red-cell transfusions no longer required them, and one was able to undergo phlebotomy for severe iron overload. A total of nine patients eventually had a neutrophil response, with a median increase of 1350 cells per cubic millimeter, including four patients with severe neutropenia at baseline (Figure 2C). Mean (±SE) blood counts in all three lineages in patients with a response are summarized in Fig. 3S in the Supplementary Appendix. Patient 5, who had an unconfirmed cataract, had a platelet response by 12 weeks and eventually had a trilineage response that persisted for 21 months, despite discontinuation of eltrombopag after only 9 weeks.
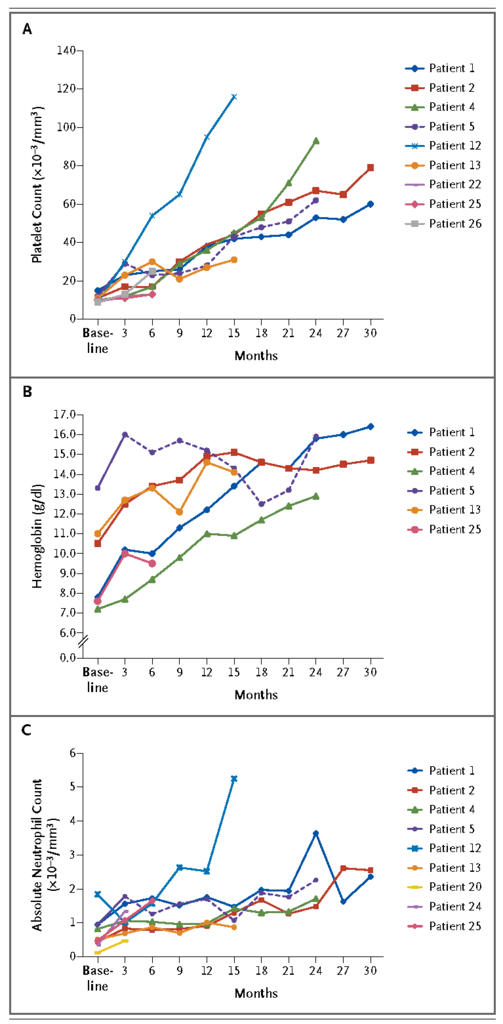
Serial bone marrow–biopsy specimens were assessed for cellularity. Three of four patients with hematologic responses who were followed for more than 8 months had normalization of cellularity (Figure 3
FIGURE 3Bone Marrow Cellularity at Baseline and at 8 Months or Longer in Four Patients with Trilineage Responses to Eltrombopag.). Immunohistochemical staining for CD34 showed normalization of the frequency of hematopoietic stem and progenitor cells (Fig. 4S in theSupplementary Appendix).
Relapse, Survival, and Clonal Evolution
In 8 of the 11 patients with a response, the response was maintained without a relapse for a median of 10 months; 7 of these patients have continued to receive eltrombopag. One patient with a neutrophil response had a relapse after 6 months of treatment; eltrombopag was discontinued, and she died from infectious complications related to profound neutropenia. Clonal evolution to monosomy 7 developed in 2 patients who did not have a response; 1 died after disease progression to myeloid leukemia, and a second patient prepared to undergo HSCT. One additional patient who did not have a response died from infection 6 months after drug discontinuation. Neither clonal evolution nor the myelodysplastic syndrome, as assessed by means of bone marrow examination and metaphase cytogenetic analysis every 6 months, developed in any patient with a hematologic response (Table 2S in the Supplementary Appendix).
Predictors of Response
Using nominal P values, we analyzed a number of baseline characteristics in patients with a response versus patients without a response. Age, neutrophil count, age-adjusted telomere length, plasma cytokine levels measured with the use of a multiplex assay, presence or absence of more than 1% GPI-deficient neutrophils, number of prior cycles of immunosuppression, time since the last cycle of immunosuppression, duration of aplastic anemia, T-cell immunophenotype, primary refractory disease versus relapsed refractory disease, and thrombopoietin level were not predictive of response. The mean reticulocyte count at baseline was predictive (47,864±8265 per cubic millimeter in patients with a response vs. 20,921±5254 per cubic millimeter in patients without a response, P=0.01). The baseline immature platelet count was 1183±145 per cubic millimeter in patients with a response versus 785±146 per cubic millimeter in patients without a response (P=0.06 by Student's t-test for means), with the top quartile of immature platelet counts predicting a response (P=0.03). The median time from the initiation of the most recent course of immunosuppression was 14 months (range, 6 to 117) in all patients and 17 months (range, 6 to 117) in those who had hematologic responses; these findings obviated concern that responses could indicate late effects of immunosuppression.
Thrombopoietin Level, Telomere Length, and Immunophenotype
Patients with aplastic anemia have markedly elevated thrombopoietin levels as compared with patients who have immune thrombocytopenic purpura or healthy persons.15,20 We measured serum thrombopoietin levels at baseline and after 12 weeks of treatment with eltrombopag. The mean baseline level was 2767 pg per milliliter (range, 1615 to 4618), as compared with 400 pg per milliliter in healthy controls,15 and was unchanged after 3 months of treatment.
The age-adjusted baseline telomere length of leukocytes was in the first quartile in 22 of 25 patients and did not change with 12 weeks of eltrombopag treatment. In the 11 patients with a response, we detected no significant change in telomere length at the most recent assessment (range, 12 to 117 weeks).
An immunophenotypic panel of T-cell subsets in blood was assessed at baseline and at 3 months (Table 3S in the Supplementary Appendix). There were no significant changes from baseline in the percentage or absolute numbers of T regulatory cells or other T-cell subsets in patients who were receiving eltrombopag, nor were there significant differences between patients with a response and those without a response.
DISCUSSION
The hallmark of severe aplastic anemia is an autoimmune attack against the bone marrow, resulting in a paucity of hematopoietic stem and progenitor cells.1 We administered eltrombopag, a synthetic small-molecule thrombopoietin agonist, in patients with refractory aplastic anemia to determine whether pharmacologic stimulation of the c-MPL receptor can improve hematopoiesis. We observed clinically significant responses in platelet, erythroid, and neutrophil lineages in 11 of 25 patients at 12 weeks, with normalization of bone marrow cellularity and trilineage hematopoiesis in patients with a response who continued to receive eltrombopag. These findings, which suggest that eltrombopag stimulates hematopoiesis at the level of primitive hematopoietic cells, are consistent with the results of previous laboratory studies.7-10 Previous studies of cytokines in aplastic anemia have not shown a benefit, probably because hematopoietic stem and progenitor cells do not have receptors for erythropoietin or G-CSF.21
Thrombopoietin levels are significantly elevated in patients with aplastic anemia, in contrast to thrombopoietin levels in patients with immune thrombocytopenic purpura, which are at the high end of the normal range.20 Eltrombopag does not compete with thrombopoietin for binding to c-MPL, since it binds in the membrane-spanning region outside the ligand-binding pocket of c-MPL, and it activates signaling through JAK-STAT (Janus-associated kinase–signal transducers and activators of transcription) and MAPK (mitogen-activated protein kinase) pathways.22 Eltrombopag may stimulate hematopoiesis by noncompetitive activation of c-MPL, despite elevated thrombopoietin levels, and it may activate signaling in a way that is distinct from that of thrombopoietin.
All 25 patients in our study who received eltrombopag were dependent on platelet transfusions at enrollment. In 9 patients, treatment with eltrombopag eliminated the need for platelet transfusions. The increases in platelet counts in these patients occurred over several months, whereas the response to eltrombopag in patients with immune thrombocytopenic purpura is more rapid, with an increase in platelet counts within 2 weeks after the start of treatment.23 The kinetics of the delayed response in aplastic anemia may reflect inherently slow cycling of hematopoietic stem and progenitor cells, as compared with the more rapid effects of the drug on maturing megakaryocytes. Patients in our study who did not have a response at 12 weeks might have had a response to more prolonged use of eltrombopag, given that the patients with a response at 12 weeks who participated in the extension study had additional lineage responses and markedly improved counts with continued treatment. The only patient or laboratory characteristics that predicted a response in this small cohort were less profoundly depressed baseline reticulocyte and immature platelet counts, which may reflect residual stem cells that can respond to eltrombopag. Response to initial immunosuppression also correlated with the baseline reticulocyte count in a previous study.24
It is possible that c-MPL stimulation enhances clonal evolution to myelodysplasia or leukemia. Clonal evolution occurs in 10 to 15% of patients with aplastic anemia overall, and the incidence of disease progression is highest in patients without a response and in those with short telomeres.15,25,26 Some studies have shown an association of prolonged G-CSF treatment with clonal evolution to monosomy 7 and the myelodysplastic syndrome, but this is more frequent in patients with severe, refractory aplastic anemia than in patients who have a good response to immunosuppression, and patients with refractory disease more often receive long-term treatment with G-CSF.27 In our cohort, monosomy 7 was detected at the conclusion of the study in two patients without a response, one of whom had extremely short telomeres. Patients with a response continued to receive the drug and were monitored by means of serial bone marrow biopsies and cytogenetic analysis; these patients did not have progression to myelodysplasia or clonal hematopoiesis. Given the uncertainty about this risk, it would be prudent to monitor patients with aplastic anemia who receive eltrombopag by performing serial bone marrow biopsies and cytogenetic analysis; in our opinion, treatment should be limited to clinical trials.
As with other autoimmune disorders such as multiple sclerosis and Crohn's disease, recovery in aplastic anemia may be limited not only by ongoing immune attack but also by profound depletion of tissue stem cells.28 Hematopoietic stem and progenitor cells have inherent expansion capabilities, but they may require exogenous stimulation to regenerate adequate hematopoiesis. Data are lacking on whether the addition of eltrombopag to up-front immunosuppressive therapy can preserve hematopoietic stem cells and progenitor cells and improve the rate and quality of responses in aplastic anemia. Expansion of hematopoietic stem cells and progenitor cells with eltrombopag might also benefit patients undergoing prolonged chemotherapy or speed recovery of hematopoiesis after umbilical-cord blood transplantation.





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 