手術前後使用抗凝血藥物要注意的事情
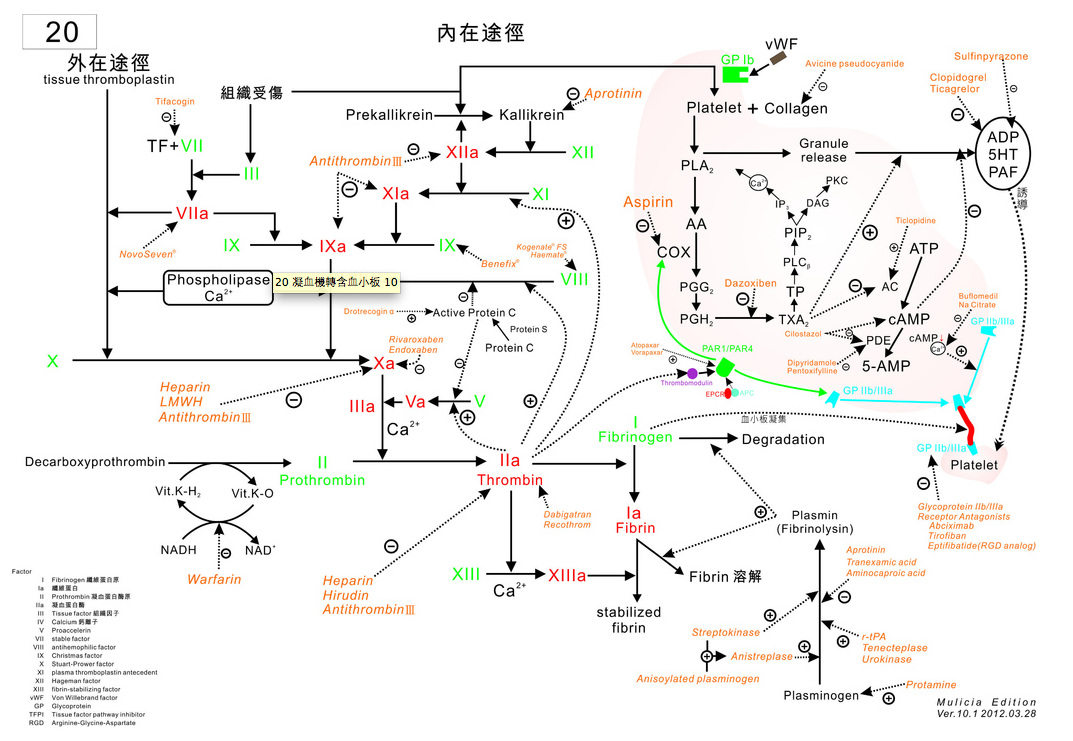
順便替大家複習一下凝血機轉
More than 6 million patients in the United States receive long-term anticoagulation therapy for the prevention of thromboembolism due to atrial fibrillation, placement of a mechanical heart-valve prosthesis, or venous thromboembolism.1 In addition, dual antiplatelet therapy (combination treatment with aspirin and a thienopyridine) after the placement of a coronary-artery stent has dramatically increased. Annually, 10% of patients taking antithrombotic agents undergo surgical or other invasive procedures that require temporary discontinuation of therapy.2 Although the goal is to minimize thromboembolic events and major hemorrhage in the periprocedural period, data from randomized, controlled trials in this area are limited, and many recommendations are derived from single-center cohorts, particularly with regard to bridging anticoagulation therapy. In addition, there are minimal data on procedure-specific bleeding rates in this patient population.
Guidelines from scientific societies with graded levels of evidence, as well as prior review articles, provide direction for periprocedural management of antithrombotic agents.2-12 This review provides approaches and recommendations that are based on recent changes in national guidelines2,13 for patients undergoing invasive procedures while receiving antithrombotic therapy, including newer antithrombotic agents.14,15
GENERAL CONCEPTS
The question of whether antithrombotic therapy should be suspended in a patient who will be undergoing an invasive procedure involves balancing the risk of postprocedural bleeding with continued treatment against the thrombotic risk with suspension of treatment and use of bridging anticoagulation therapy. In general, a patient undergoing a procedure that is associated with a low risk of bleeding (low-risk procedure) can safely continue antithrombotic therapy and should do so, particularly if the patient is at high risk for a thromboembolic event (high-risk patient). Conversely, a patient undergoing a high-risk procedure can temporarily discontinue antithrombotic agents safely if the patient is at low risk for a thromboembolic event (low-risk patient).
The decision-making process is challenging when patients at moderate-to-high risk for thromboembolic events undergo high-risk procedures. Management also differs between elective and emergency procedures. A discussion among a clinician specializing in periprocedural management of antithrombotic agents and coagulation disorders, the primary provider prescribing these agents, and the proceduralist is essential. Ideally, this communication should occur well in advance of the procedure to maximize patient safety and facilitate patient education.2 Appropriate decision making requires knowledge of thrombotic risk, procedure-related bleeding risk, concepts of bridging anticoagulation therapy, and timing of cessation and reinitiation of antithrombotic therapy. The first step in antithrombotic management is to assess the risk of thromboembolic events during the period when antithrombotic agents are to be discontinued.
ASSESSMENT OF THROMBOTIC RISK
Atrial Fibrillation
Periprocedural risks of discontinuing antithrombotic therapy are extrapolated from risks outside the periprocedural period. For patients with nonvalvular atrial fibrillation, important determinants of the risk of stroke include the CHADS2 score16 (Table 1
)
and, more recently, the CHA2DS2-VASc score, which includes cardiovascular atherosclerotic disease and female sex as additional risk factors.17 Scores on the CHADS2 range from 0 to 6, with higher scores indicating a greater risk of stroke; congestive heart failure, hypertension, diabetes, and an age of 75 years or older are each assigned 1 point, and prior stroke or transient ischemic attack is assigned 2 points. Scores on the CHA2DS2-VASc range from 0 to 9, with higher scores indicating greater risk. For the CHA2DS2-VASc score, an age of 65 to 74 years is assigned 1 point and an age of 75 years or older is assigned 2 points. Valvular atrial fibrillation implies the coexistence of severe valvular heart disease (mechanical valvular prosthesis or mitral-valve repair), and affected patients are at high risk for thromboembolism.
Mechanical Heart Valves and Venous Thromboembolism
Risk factors for thromboembolic events in patients with one or more mechanical heart valves and venous thromboembolism are outlined in Table 2
.
The risk is influenced by the type, number, and location of valvular prostheses, as well as by the presence or absence of associated heart failure, atrial fibrillation, history of thromboembolism, and intracardiac thrombi.19,20
In patients with venous thromboembolism, the risks of recurrent thrombosis, thrombus propagation, and embolization are elevated for 3 months after the diagnosis and initiation of anticoagulation therapy.21 The risk of recurrence differs depending on whether the venous thromboembolism was provoked (in which case the risk decreases with resolution of the underlying risk factor) or unprovoked (i.e., idiopathic) (Table 2).22
Cancer
Patients with cancer have an increased risk of periprocedural thrombosis owing to cancer-specific prothrombotic activity, hormonal therapy, angiogenesis inhibitors, radiotherapy, and the presence of indwelling central venous catheters.23 Concurrently, there is an increased risk of bleeding24because of the administration of prophylactic agents for the prevention of venous thromboembolism, chemotherapy-related hepatic and renal dysfunction and thrombocytopenia, and tumor friability. An increasing number of outpatients with cancer-related thrombosis and a history of recurrent thrombosis before the cancer diagnosis are receiving long-term parenteral anticoagulation therapy.25
Coronary Stents
Some patients with coronary stents may require dual antiplatelet therapy indefinitely. Premature discontinuation of antiplatelet therapy in anticipation of a surgical or other invasive procedure may lead to stent thrombosis26,27 and precipitation of myocardial infarction, with a mortality rate of 50% or higher.2
The risk of stent thrombosis differs between bare-metal stents and drug-eluting stents. The risk of thrombosis is highest within 6 weeks after the placement of a bare-metal stent and within 3 to 6 months after the placement of a drug-eluting stent28; antiplatelet therapy is required for at least 1 month after placement of a bare-metal stent and for 1 year after placement of a drug-eluting stent.29 After acute coronary syndromes, continuation of dual antiplatelet therapy is recommended for up to 12 months in patients with bare-metal stents and for at least 12 months in patients with drug-eluting stents, unless the risk of bleeding is excessive.30 The optimal duration of dual antiplatelet therapy for patients with coronary stents remains unknown. However, recent studies suggest that 6 to 12 months may be appropriate, with little to gain from a longer duration.31,32
ASSESSMENT OF PERIPROCEDURAL BLEEDING RISKS
The risk of major periprocedural bleeding depends on the type of procedure, and additional risk factors include residual effects of antithrombotic agents, active cancer and chemotherapy, history of bleeding, and reinitiation of antithrombotic therapy within 24 hours after the procedure.33 Table S1 in the Supplementary Appendix, available with the full text of this article at NEJM.org, shows commonly performed procedures and associated bleeding risks; classification into high-risk and low-risk categories is based on guidelines and reviews coupled with expert opinion within our institution.
Grades of bleeding severity are not standardized across specialties.34 The American Society for Gastrointestinal Endoscopy designates low-risk procedures as those with clinical rates of bleeding of 1.5% or less.35 In the absence of specific risk stratification, we propose that high-risk procedures are those with a rate of bleeding of more than 1.5% among patients not receiving antithrombotic agents. In addition, procedures that can result in intracranial, intraspinal, intraocular, retroperitoneal, intrathoracic, or pericardial bleeding are high-risk, with bleeding in these locations classified as major.34 Neuraxial anesthesia is a high-risk procedure.36,37
For other procedures, we determine the severity of bleeding using criteria similar to those used for gastrointestinal endoscopic procedures (Table S2 in the Supplementary Appendix).38
BRIDGING ANTICOAGULATION THERAPY
Bridging anticoagulation therapy is designed to minimize the risk of thromboembolism in high-risk patients when anticoagulation therapy is suspended (Table 1 and Table 2) and to minimize the risk of bleeding after high-risk procedures (Table S2 in the Supplementary Appendix). The need for bridging depends on the duration of action of the anticoagulant agent and the potential for reversing anticoagulation. In most cases, bridging anticoagulation therapy is used in patients receiving warfarin. Once warfarin has been discontinued and the international normalized ratio (INR) falls below the therapeutic range, intravenous unfractionated heparin or subcutaneous low-molecular-weight heparin is administered for 3 to 5 days. The heparin agent is withdrawn before the procedure, with the timing based on whether unfractionated heparin or low-molecular-weight heparin is used, and is usually readministered 48 hours after the procedure, if hemostasis is secured.
Although the use of bridging anticoagulation therapy in high-risk patients is considered the standard of care, it has been evaluated in only two randomized, controlled trials39,40 and remains controversial.41,42 The results of an ongoing trial of the use of bridging therapy in high-risk patients are awaited.43 The available data are difficult to interpret because high-risk and low-risk patients and high-risk and low-risk procedures have been pooled. Older studies used nonperioperative data to estimate perioperative risks of stroke and thromboembolism during a period of 8 to 10 days of warfarin interruption. Rates of thrombosis were estimated to be quite low (0.1 to 0.4% among patients with a mechanical heart valve and <0.1 to 0.2% among those with atrial fibrillation39). However, actual overall rates (with and without bridging) are higher: 1.2% among patients with a mechanical heart valve, 0.9% among those with atrial fibrillation, and 1.8% among those with venous thromboembolism.44 Corresponding rates of major bleeding (with and without bridging) are 2.7%, 2.0%, and 1.9%, respectively.44
A recent meta-analysis showed that periprocedural bridging therapy with heparin increased the overall risk of major bleeding without a significant decrease in the risk of thromboembolic events.42 This conclusion has been affirmed in the study by Birnie et al. in this issue of theJournal.40 This single-blind, randomized study involved patients at moderate-to-high risk for thromboembolic events who were undergoing pacemaker or defibrillator surgery. A clinically significant device-pocket hematoma was more common in the heparin-bridging group (16.0%) than in the continued-warfarin group (3.5%). Major surgical and thromboembolic complications were rare in both treatment groups.
When bridging therapy is required for high-risk patients with an estimated creatinine clearance of less than 30 ml per minute, the use of unfractionated heparin is preferred. High-dose unfractionated heparin (therapeutic anticoagulation)2 is commonly used, with monitoring of the activated partial-thromboplastin time. For low-risk patients, such as those with an episode of venous thromboembolism more than 3 months before the planned procedure, prophylactic low-dose heparin can be used for bridging.2 In moderate-risk patients, the decision to use bridging therapy and the degree of intensity of bridging therapy should be individualized and the patient's wishes considered.2
Our approach to bridging therapy, shown in Table 3
,
is consistent with published guidelines.2 The protocol is as follows. Stop warfarin 5 days before a high-risk procedure, and when the INR falls below the therapeutic range, begin low-molecular-weight heparin at a therapeutic dose. For patients with a mechanical heart valve or atrial fibrillation, use enoxaparin at a dose of 1 mg per kilogram of body weight, administered every 12 hours, or dalteparin at a dose of 100 IU per kilogram, administered every 12 hours. For patients with venous thromboembolism, use enoxaparin at a dose of 1.5 mg per kilogram or dalteparin at a dose of 200 IU per kilogram once daily. The final dose (either enoxaparin at a dose of 1 mg per kilogram or dalteparin at a dose of 100 IU per kilogram) should be administered 24 hours before the procedure.
Check the INR on the morning of the procedure. Restart warfarin therapy immediately after the procedure if hemostasis is secured, and reinstitute treatment with subcutaneous low-molecular-weight heparin or intravenous unfractionated heparin at a therapeutic dose (without bolus) 48 hours after the procedure if no bleeding has occurred, with the exception that for patients undergoing endoscopic sphincterotomy, heparin therapy should be initiated after 72 hours.45Discontinue heparin therapy when the INR is in the therapeutic range (approximately 5 days later).
TIMING OF CESSATION OF ANTITHROMBOTIC THERAPY
When anticoagulant agents are discontinued in high-risk patients (including agents used for bridging therapy), the interval without anticoagulation therapy should be as short as possible, with the risk of thromboembolic events balanced against the risk of bleeding. The duration of action of the anticoagulant agent depends on several factors, including renal function (in the case of dabigatran, rivaroxaban, apixaban, and low-molecular-weight heparin), hepatic function (in the case of warfarin, rivaroxaban, and apixaban), and potential for reversing the effects (in the case of warfarin and heparin). Standardized monitoring recommendations are available for warfarin but not for the newer antithrombotic agents.46,47 Suggested monitoring for older and newer antithrombotic agents is presented in Table 4
.
我這邊有一張拼起來的給各位:
Warfarin
A relatively normal zone of hemostasis exists when the INR is 1.0 to 2.0, with the lower value corresponding to a coagulation factor level of 100% and the higher value corresponding to a level of 30%.53 The INR value at which the risk of bleeding increases is unknown, but the risk is assumed not to be elevated when the INR is 1.5 or less and is assumed to be elevated when the INR is more than 2.0.2,53 Thus, an INR of 1.5 or less is considered safe for high-risk procedures,2,36,54 although some clinicians recommend an INR of 1.2 or less for procedures with a high risk of bleeding into closed spaces (e.g., intracranial surgery)2 (Table S2 in the Supplementary Appendix).
An INR of 2.0 to 3.5 corresponds to therapeutic anticoagulation,53 and 93% of patients with an INR within this range have an INR of less than 1.5 approximately 5 days after warfarin therapy has been discontinued.48 The INR should be obtained within 24 hours before the procedure55 and corrected with vitamin K, if needed, except in the presence of mechanical heart valves.
When warfarin therapy is continued through the procedure, it is important that the INR before the procedure not be supratherapeutic. We adjust warfarin doses over a period of 5 days to aim for an INR of approximately 2.5 by the time of the procedure.
Heparin
Unfractionated heparin administered intravenously has a half-life of 60 to 90 minutes, and anticoagulant effects dissipate 3 to 4 hours after discontinuation. Thus, the infusion is stopped 4 to 6 hours before high-risk procedures.2 Low-molecular-weight heparin is administered subcutaneously at therapeutic doses for bridging and for the treatment of venous thromboembolism, with reduced doses for the prevention of venous thromboembolism in low-risk patients. The half-life of these agents is approximately 4 hours, and the last dose should be given 24 hours before the anticipated procedure at 50% of the total daily dose (i.e., enoxaparin at a dose of 1 mg per kilogram or dalteparin at a dose of 100 IU per kilogram).2
Newer Anticoagulant Agents
Direct factor Xa inhibitors include the oral agents rivaroxaban and apixaban. The timing of discontinuation of both agents before high-risk procedures depends on the creatinine clearance.53 The recommended durations are taken from package inserts, if the information is provided, or are derived from guidelines and drug pharmacokinetics (Table 4). However, because of the lack of available reversal agents, we prefer to take a more conservative approach, withholding these agents for slightly longer periods than those based on package inserts, guidelines, or pharmacokinetic data (i.e., 1 to 2 days longer than the specifications outlined in Table 4).44 Future studies should focus on more precise laboratory monitoring and reliable reversal of the newer anticoagulant agents.
Fondaparinux is a subcutaneously administered direct factor Xa inhibitor approved for the prevention and treatment of venous thromboembolism, with a half-life of 17 hours. It has been shown to be associated with acceptable rates of bleeding when discontinued more than 36 hours before cardiopulmonary bypass surgery.56
Direct thrombin inhibitors can be administered orally (dabigatran), subcutaneously (desirudin), and intravenously (argatroban and bivalirudin). Bivalirudin is used primarily during acute coronary interventions. Argatroban is used for the treatment of heparin-induced thrombocytopenia. Dabigatran is approved for the prevention of thromboembolic stroke in patients with nonvalvular atrial fibrillation, and the timing of discontinuation is based on the creatinine clearance (Table 4).57,58 Desirudin is used postoperatively for prophylaxis against deep-vein thrombosis in patients undergoing hip replacement. It has a half-life of 2 hours and should be discontinued 10 hours before high-risk procedures.
ANTIPLATELET AGENTS
Traditional Antiplatelet Agents
Aspirin is used alone or in combination with other antiplatelet agents. Low-dose aspirin alone does not substantially increase the risk of clinically important bleeding after invasive procedures.59-62
Dipyridamole reversibly inhibits platelet aggregation. It has an elimination half-life of 12 hours and a duration of action of approximately 2 days after discontinuation. Aspirin and dipyridamole individually do not substantially increase the risk of clinically important postprocedural bleeding but are sometimes discontinued before certain elective high-risk procedures. Administration of aspirin and dipyridamole together (i.e., Aggrenox [Boehringer Ingelheim]) probably increases the risk of postprocedural bleeding.63
Cilostazol, a phosphodiesterase inhibitor, is approved for the treatment of claudication and is used off-label in combination with antiplatelet drugs for patients with coronary artery disease64 and for those with ischemic cerebrovascular disease.65 Cilostazol does not increase the risk of bleeding when used alone. Platelet function returns to normal approximately 2 days after discontinuation.15
Other Antiplatelet Agents
Orally administered inhibitors of the adenosine diphosphate receptor P2Y12 include clopidogrel, ticlopidine, prasugrel, and ticagrelor (Table 4).15 The period during which therapy should be suspended ranges from 5 to 7 days for clopidogrel, ticagrelor, and prasugrel and possibly longer for ticlopidine (10 to 14 days).
INFERIOR VENA CAVA FILTERS
We do not recommend the routine placement of inferior vena cava filters for bridging.23 Whenever feasible, we suggest delaying elective surgical procedures until patients have received at least 3 months of anticoagulation therapy. The placement of an inferior vena cava filter (which should be removable, whenever possible, because of the risks of long-term adverse events with permanent filters) may be indicated if pulmonary thromboembolism or proximal deep-vein thrombosis has occurred within the previous 4 weeks and an urgent procedure is required. In such cases, filters can prevent pulmonary embolic events and allow the temporary discontinuation of anticoagulation therapy.66
PHARMACOLOGIC REVERSAL OF ANTICOAGULATION
When urgent or emergency procedures are required, there are various options for the management of antithrombotic agents. In some patients, a low-risk temporizing procedure may be carried out to delay the need for a definitive higher-risk procedure. When this approach is not possible, the administration of reversal agents may be considered if the risk of bleeding outweighs the risk of thrombotic events. Several reversal agents are available (Table 4). Further discussion of plasma and prothrombin complex concentrates is provided in the Supplementary Appendix.49,51
Antithrombotic Agents with Reversible Effects
In patients with an INR that is not supratherapeutic, the effect of warfarin can be reliably reversed within 24 to 48 hours by administering intravenous vitamin K. Reversal occurs within a few hours after the infusion of vitamin K and fresh-frozen plasma.53,67 The administration of either vitamin K or fresh-frozen plasma may cause clinical problems. High-dose vitamin K will delay the response to warfarin therapy when it is reinstituted. The administration of fresh-frozen plasma may lead to volume overload in patients with advanced cardiac or kidney disease.68 Prothrombin complex concentrates are preferred in cases of bleeding related to vitamin K antagonist treatment,49particularly for patients with heart failure, valvular heart disease, or renal failure, in whom a large-volume infusion of fresh-frozen plasma may result in volume overload.
Because unfractionated heparin has a short duration of action, reversal is not usually required. Protamine can completely reverse the action of unfractionated heparin and can partially reverse the action of low-molecular-weight heparin.
Antithrombotic Agents with Nonreversible Effects
Reliable reversibility of the effects of newer anticoagulant agents (direct thrombin inhibitors and direct factor Xa inhibitors) has not been proved.53 There is no clinical evidence that prothrombin complex concentrates effectively reverse major bleeding induced by these agents. The effects of rivaroxaban but not dabigatran were reversed in 12 healthy volunteers after the administration of 4-factor prothrombin complex concentrates,69 which contain factors II, VII, IX, and X and proteins C and S (see the Supplementary Appendix). It is unknown whether these data are applicable to patients undergoing invasive procedures.
In patients receiving dabigatran who have life-threatening bleeding that cannot be managed with supportive care and local hemostatic measures, hemodialysis or charcoal hemoperfusion can be considered.70 However, these interventions may not be feasible, given the bleeding risks associated with dialysis-catheter placement. Moreover, hemodialysis may not increase drug elimination in the absence of renal failure. Unlike dabigatran, rivaroxaban and apixaban are not dialyzable.
For patients receiving treatment with newer anticoagulant agents, when surgery is imminent but the timing is unpredictable (e.g., organ transplantation), we recommend switching to warfarin because its effects can be rapidly and reliably reversed.
RESUMPTION OF ANTITHROMBOTIC THERAPY
The reinitiation of antithrombotic therapy, particularly full-dose therapy, is a major determinant of the bleeding risk after invasive procedures. In contrast to full-dose anticoagulation therapy, prophylactic anticoagulation therapy is resumed once hemostasis is secured. In patients receiving bridging therapy, heparin at a therapeutic dose should be withheld for 48 hours after the procedure. If the risk of postprocedural bleeding is deemed acceptably low, full-dose anticoagulation therapy may be initiated after a shorter interval.
Because achieving full anticoagulation after the reinstitution of warfarin therapy takes several days, it can be reinstituted the evening of the day on which the procedure is performed, unless there is a substantial risk of delayed bleeding or unless reoperation is anticipated. We recommend delaying the reinitiation of treatment with dabigatran, rivaroxaban, or apixaban for at least 48 hours after high-risk procedures because the full anticoagulatory effect occurs shortly after administration and there are no reliable reversal agents for these medications.
Clopidogrel administered at maintenance doses has a delayed onset of action, and treatment can therefore be reinitiated within 24 hours after the procedure. Clopidogrel loading, which results in a rapid onset of action, can be used if the risk of bleeding is lower than anticipated because of a change in the procedure that was performed (e.g., endoscopic biopsy of a large colonic polyp rather than polypectomy). Treatment with other antiplatelet agents, including aspirin, can be reinitiated within 24 hours. We recommend caution when reinitiating treatment with prasugrel or ticagrelor because of their rapid onset of action, potent antiplatelet inhibition, and the lack of agents to reverse their effects.
Some gastrointestinal endoscopic procedures are associated with cautery-induced injury that may result in delayed bleeding 7 to 10 days after the procedure (e.g., polypectomy and biliary sphincterotomy). If antithrombotic therapy is reinstituted after a short interval, the full antithrombotic effects may coincide with the onset of delayed bleeding, although it is often not practical to withhold antithrombotic agents for a longer period.



















 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 