Volume 18, Issue 5, May 2017, Pages 654-662 https://doi.org/10.1016/S1470-2045(17)30109-2
Summary
Background
Stomatitis is a class effect associated with the inhibition of mTORand is associated with everolimus therapy for breast cancer. Topical steroids might reduce stomatitis incidence and severity, and the need for dose reductions and interruptions of everolimus. Anecdotal use of topical steroid oral prophylaxis has been reported in patients with breast cancer. We aimed to assess dexamethasone-based mouthwashfor prevention of stomatitis in patients with breast cancer.
Methods
This US-based, multicentre, single-arm, phase 2 prevention study enrolled women aged 18 years and older with postmenopausal status who had histologically or cytologically confirmed metastatic hormone receptor-positive, HER2-negative breast cancer. Beginning on day 1 of cycle 1, patients received everolimus 10 mg plus exemestane 25 mg daily, with 10 mL of alcohol-free dexamethasone0·5 mg per 5 mL oral solution (swish for 2 min and spit, four times daily for 8 weeks). After 8 weeks, dexamethasone mouthwash could be continued for up to eight additional weeks at the discretion of the clinician and patient. The primary endpoint was incidence of grade 2 or worse stomatitis by 8 weeks assessed in the full analysis set (patients who received at least one dose of everolimus and exemestane and at least one confirmed dose of dexamethasone mouthwash) versus historical controls from the BOLERO-2 trial (everolimus and exemestane treatment in patients with hormone receptor-positive advanced breast cancer who were not given dexamethasone mouthwash for prevention of stomatitis). This trial is registered at ClinicalTrials.gov, number NCT02069093.
Findings
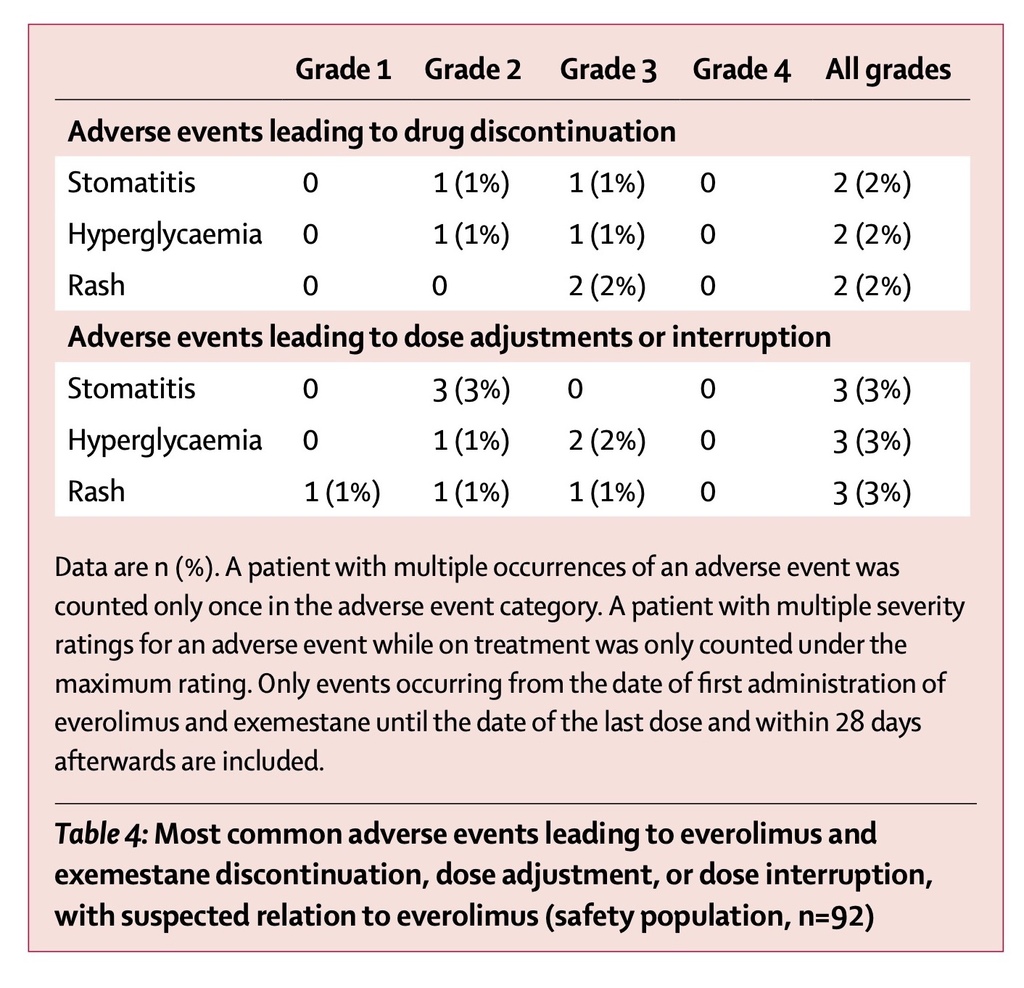
Between May 28, 2014, and Oct 8, 2015, we enrolled 92 women; 85 were evaluable for efficacy. By 8 weeks, the incidence of grade 2 or worse stomatitis was two (2%) of 85 patients (95% CI 0·29–8·24), versus 159 (33%) of 482 patients (95% CI 28·8–37·4) for the duration of the BOLERO-2 study. Overall, 83 (90%) of 92 patients had at least one adverse event. The most frequently reported grade 3 and 4 adverse events in the safety set were hyperglycaemia (seven [8%] of 92 patients), rash (four [4%]), and dyspnoea (three [3%]). Serious adverse events were reported in 20 (22%) patients; six (7%) were deemed treatment related, with dyspnoea (three [3%]) and pneumonia (two [2%]) reported most frequently. 12 (13%) of 92 patients had adverse events suspected to be related to treatment that led to discontinuation of everolimus and exemestane (the most common were rash, hyperglycaemia, and stomatitis, which each affected two [2%] patients).
Interpretation
Prophylactic use of dexamethasone oral solution substantially reduced the incidence and severity of stomatitis in patients receiving everolimus and exemestane and could be a new standard of oral carefor patients receiving everolimus and exemestane therapy.
Funding
Novartis Pharmaceuticals Corporation.
Introduction
The phosphoinositide 3-kinase (PI3K)–serine/threonine-protein kinases (AKT)–mTOR pathway is the most frequently altered signal transduction pathway in breast cancer, making it an attractive therapeutic target.1 Combination therapy with the oral mTORinhibitor everolimus and the aromatase inhibitor exemestane is approved for the treatment of advanced breast cancer in patients with hormone receptor-positive disease who have progressed on nonsteroidal aromatase inhibitors.2
The most common adverse event associated with everolimus is stomatitis, which is characterised by inflammation of the mucous membranes lining the mouth. Stomatitis is a class effect associated with mTOR inhibition that can adversely affect tolerability and treatment adherence.3, 4 The onset and pattern of mTOR inhibitor-associated stomatitis have been well characterised.4, 5, 6, 7 It presents as aphthous-like oral lesions, characterised by discrete shallow ulcerations.8, 9
Management of mTOR inhibitor-associated stomatitis by dose delays, reductions, or discontinuation of the mTOR inhibitor might compromise duration and intensity of therapy and might affect decisions to initiate treatment with mTOR inhibitors.3 Aphthous ulcers have been treated successfully by steroid-containing oral paste or rinse; anecdotal reports suggested that topical steroids could improve healing of everolimus-associated aphthous ulcers.3, 8, 10Until now, clinical evidence has been scarce to guide mTOR inhibitor-associated stomatitis prophylactic strategies.
Research in context
Evidence before this study
We searched PubMed for original research articles in English published between Jan 8, 2006, and Oct 14, 2016, with the terms “breast cancer”, “mTOR inhibitor”, “stomatitis”, and “steroid mouthwash”. We did not identify any phase 2 or 3 trials that investigated the prophylactic use of steroidmouthwash for stomatitis in women who are postmenopausal and treated with everolimus and exemestane for hormone receptor-positive, HER2-negative metastatic breast cancer.
Added value of this study
To our knowledge, this is the first and largest stomatitis prevention study completed that combines therapeutic management with proactive patient engagement to reduce the incidence and severity of stomatitis in patients who are postmenopausal and receiving everolimus and exemestane as treatment for hormone receptor-positive, HER2-negative metastatic breast cancer. Our results show that a commercially available, inexpensive, and well tolerated dexamethasone mouthwash resulted in a more than ten-times reduction in the incidence of grade 2 or higher stomatitis by 8 weeks compared with BOLERO-2 historical controls. Patient-reported outcomes monitoring diet and oral pain further substantiated the efficacy of the dexamethasone mouthwash. The favourable dose intensity for everolimus and exemestane that was recorded in this trial might be a consequence of the reduced incidence of stomatitis, as well as the fact that dexamethasone mouthwash treatment was well tolerated with minimal toxicity.
Implications of all the available evidence
Steroid mouthwash as prophylaxis for everolimus-related stomatitis should be considered as a new standard of oral care for patients who are postmenopausal and receiving everolimus and exemestane for treatment of hormone receptor-positive, HER2-negative metastatic or advanced breast cancer. Several ongoing clinical trials are assessing specific preventive and therapeutic rinses for oral mucositis or stomatitis and might further support a use of steroid-based prophylaxis for everolimus-related stomatitis. This preventative approach might eventually be considered as an important treatment option in many diseases for which everolimus is in active use.
In a meta-analysis of phase 3 trials, 89% of all-grade stomatitis was prominent within 8 weeks of starting everolimus.5 In the phase 3 BOLERO-2 trial of everolimus and exemestane therapy for patients with hormone receptor-positive, HER2-negative metastatic breast cancer, at a median follow-up of 13 months2 (IQR 9·1–16·2; Rugo HS, unpublished data), all-grade stomatitis occurred in 321 (67%) of 482 patients, including grade 2 or worse in 159 (33%) and grade 3 severity in 39 (8%; appendix p 9).11
We did this phase 2 SWISH trial (dexamethasone mouthwash for everolimus-related stomatitis prevention in hormone receptor-positive metastatic breast cancer) to evaluate 0·5 mg/5 mL dexamethasone mouthwash for the prevention of stomatitis in postmenopausal women receiving everolimus and exemestane for hormone receptor-positive metastatic breast cancer. We used data from the BOLERO-2 trial as a historical control cohort for comparisons.
Methods
Study design and participants
SWISH was a US-based, phase 2, single-arm trial that assessed alcohol-free dexamethasone mouthwash for the prevention of stomatitis in patients treated with everolimus and exemestane for hormone receptor-positive, HER2-negative metastatic breast cancer. Eligible patients were enrolled at 23 investigational sites from academic and community settings in the USA (appendix p 6) on the basis of the following key inclusion criteria: women aged 18 years or older who were postmenopausal and had metastatic histologically or cytologically confirmed hormone receptor-positive, HER2-negative breast cancer, newly prescribed everolimus 10 mg and exemestane 25 mg, Eastern Cooperative Oncology Group (ECOG) performance status of 0–2; adequate renal function (serum creatinine ≤1·5 × upper limit of normal), and a willingness to self-report oral pain using a visual analogue scale and dietary intake using the Normalcy of Diet Scale in a diary. Patients with active stomatitis, oral mucositis, or mouth ulcers; those receiving other anticancer therapies (except bisphosphonates or denosumab); those with severe or uncontrolled medical conditions including diabetes mellitus; and those requiring corticosteroids or other immunosuppressive drugs, were excluded (appendix p 8). There were no restrictions regarding previous treatments and thus no washout period.
The study protocol and informed consent were reviewed and approved by the institutional review board, independent ethics committee, or research ethics board before study initiation. All eligible patients included in the study provided written informed consent.
Procedures
On day 1 of cycle 1, patients initiated treatment with everolimus 10 mg plus exemestane 25 mg orally administered once daily and 10 mL of alcohol-free dexamethasone 0·5 mg per 5 mL oral solution (which is commercially available in the USA) to swish for 2 min (a timer was provided) and spit four times daily for 8 weeks. Patients were instructed not to take anything by mouth for 1 h after dosing of dexamethasone oral solution for a total of 4 h per day. The duration of one cycle was 28 days (4 weeks). After 8 weeks, dexamethasone mouthwash could be continued for up to 8 additional weeks at the discretion of the clinician and patient (appendix p 2). A 28 day safety follow-up started when patients had discontinued mouthwash treatment. Patients with grade 1 stomatitis any time during the study were advised to swish 10 mL of a 0·9% saltwater solution and spit, followed by the same regimen of dexamethasone mouthwash 10–15 min later. Patients were allowed to take oral nystatin suspension or other topical antifungal agents after the dexamethasone mouthwash according to clinician preference based on the protocols of individual institutions and practices.
Dose modification or interruption of everolimus was allowed for management of adverse events. Everolimus use was paused in the case of grade 2 or 3 stomatitis until recovery to a maximum severity of grade 1 stomatitis. Dexamethasone was held in the case of grade 3 stomatitis at the investigator's discretion. Patients were discontinued from the study or from everolimus and exemestane for the following reasons: interruption of everolimus for more than 4 consecutive weeks; diagnosis of grade 4 stomatitis; disease progression requiring a treatment change; intolerance to mouthwash regimen; or withdrawal of consent. Dexamethasone mouthwash was discontinued only if a patient withdrew from the protocol or study.
Safety assessments were done according to Common Terminology Criteria for Adverse Events, version 4.0 (CTCAE 4.0). Adverse events were monitored throughout the study duration, then for 28 days after the last treatment dose (the last dose of everolimus and exemestane or dexamethasone) or study discontinuation. Laboratory assessments of complete blood count, chemistry profile, and liver function testing for adverse event monitoring were done at screening or baseline; at cycle 1, day 28; and at cycle 2, day 28.
An educational video about stomatitis was provided to each patient with instructions about brushing their teeth at least twice daily with a soft-bristled toothbrush, daily flossing, and maintenance of good oral hygiene. Patients completed daily logs to document mouthwash usage and patient-reported outcomes including oral hygiene, Normalcy of Diet Scale scores, and visual analogue scale scores. The Normalcy of Diet Scale assesses the patient's ability to eat a normal diet by ranking food categories from easy-to-eat to difficult-to-eat (0=non-oral feeding; 100=normal diet with no restrictions). Ratings are based on the highest-ranking food that the patient could eat.12, 13 The visual analogue scale is a method to self-report oral pain (0=no pain; 10=severe pain).14, 15
Pharmacokinetic assessment of everolimus was done in evaluable patients who had a predose blood sample for analysis (obtained at cycle 1, day 28), took everolimus and exemestane dose 20–28 h before the blood sample, had no dose interruptions within 4 days of their predose blood sample being taken, and did not vomit within 4 h of their last dose of everolimus and exemestane.
Outcomes
The primary endpoint was the incidence of grade 2 or worse stomatitis by 8 weeks, compared with historical controls from the BOLERO-2 trial.7 To better assess the clinical effect of mTORinhibitor-associated stomatitis on patients, we evaluated the primary endpoint using a composite assessment. Stomatitis was graded by the investigator with a composite score of CTCAE 4.0, visual analogue scale, and Normalcy of Diet Scale. Grade 2 or worse stomatitis required confirmation by the investigator or treating physician by physical examination or interview (assessed according to CTCAE 4.0) and at least one of the following criteria: Normalcy of Diet scale score of 50 or lower, or a visual analogue scale score of 7 on two consecutive days or a score of 8, 9, or 10 on any one day.
Secondary endpoints were assessment through 8 weeks of the average number of times per day the dexamethasone mouthwash regimen was done; dose intensity of everolimus and exemestane throughout the 8 weeks; incidence of all-grade stomatitis throughout the 8 weeks; and time to resolution of grade 2 or worse stomatitis to grade 1 or below.
Statistical analysis
The full analysis set included patients who received at least one dose of everolimus and exemestane and at least one confirmed dose of dexamethasone mouthwash and was used for the efficacy analyses (stomatitis composite assessment by 8 weeks). The safety population included patients who received at least one dose of everolimus and exemestane and had at least one post-baseline safety assessment. The sample size was based on the 33% incidence of grade 2 or worse stomatitis reported on-study in historical controls of BOLERO-2 (appendix p 9),11 which led us to postulate an absolute reduction of 13% in the current study. A test of incidence rate R was done with our null hypothesis (H0: R≥0·33) and alternative hypothesis (Ha: R<0·33) with a one-sided significance level of 0·05. For a one-sided α of 0·05, 80% power, and with a χ2 test, 73 evaluable patients were required. Planned enrolment was 97 patients, to account for the fact that an anticipated 25% patients would be non-evaluable.
Secondary endpoints were summarised descriptively by frequency and day of mouthwash regimen and dose intensity of everolimus and exemestane. The incidences of all-grade and specific grade of stomatitis were summarised by counts, percentages, and 95% CIs at 8 weeks and for the entire treatment duration. Time to resolution of grade 2 or worse stomatitis was based on the number of days from diagnosis of grade 2 or worse stomatitis to resolution to grade 1 or lower. Patient-reported visual analogue scale scores and Normalcy of Diet Scale scores were summarised at baseline and by change from baseline in the full analysis set. Oral hygiene measures were also obtained from the full safety population. All patients who were excluded for analysis of the primary endpoint because of unconfirmed dexamethasone use were included in the sensitivity analysis as having grade 2 or worse stomatitis. SAS version 9.3 was used for statistical analyses of the primary and secondary endpoints.
This trial is registered with ClinicalTrials.gov, number NCT02069093.
Role of the funding source
The study was designed by the steering committee, in collaboration with the funder, who provided dexamethasone mouthwash, participated in regulatory and ethics approval, monitored safety and data management, and did statistical analyses. All authors had full access to study data, were involved in the interpretation and analyses of the data, the development and approval of the report, and had final responsibility for the decision to submit for publication.
Results
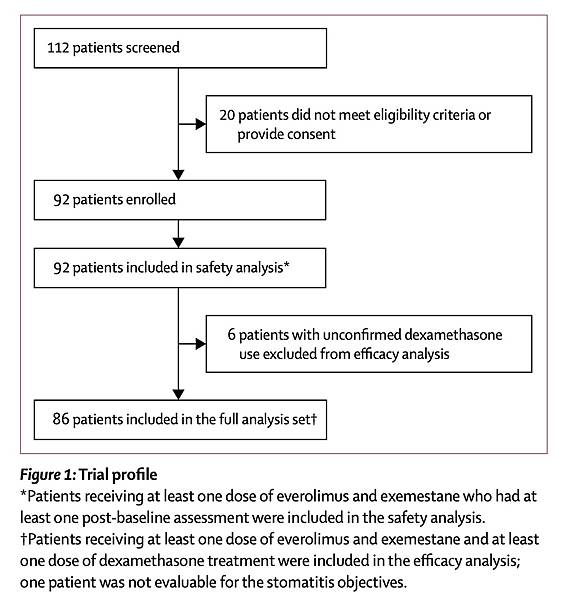
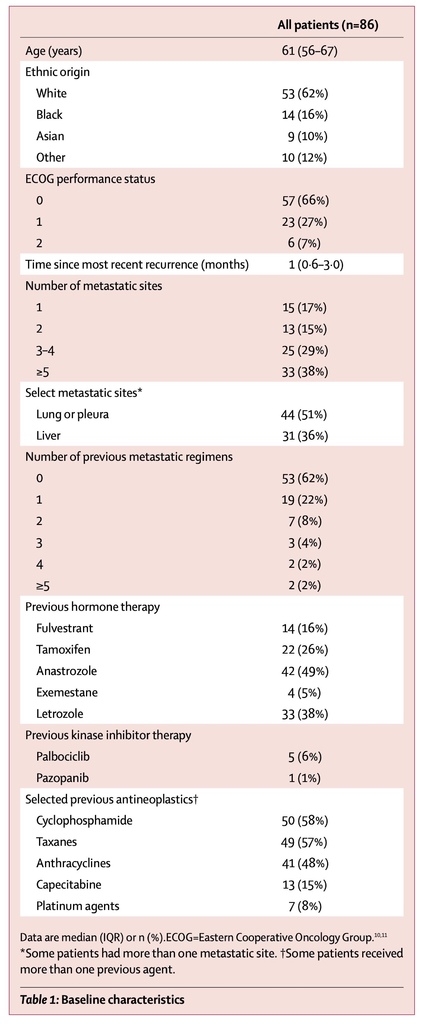
Between May 28, 2014, and Oct 8, 2015, 112 patients were screened and 92 eligible patients from 23 centres were enrolled in the SWISH trial (figure 1; appendix pp 6, 7). Of the 92 women enrolled, 86 were evaluable for efficacy; all 92 were evaluable for safety (figure 1). Six women were unevaluable for efficacy because administration of one or more doses of dexamethasone mouthwash was not confirmed. In the 86 evaluable patients, the median age was 61 years (IQR 56–67), 53 (62%) were white, 80 (93%) had an ECOG performance status of 0 or 1 and 33 (38%) received everolimus and exemestane treatment as a second-line or subsequent-line therapy in the advanced or metastatic setting (table 1) versus 385 (79%) of 485 for BOLERO-2.7
In the full analysis set of 86 patients treated with everolimus and exemestane and dexamethasone mouthwash, one patient with grade 1 stomatitis was not evaluable because of missing Normalcy of Diet Scale assessment. The primary endpoint of grade 2 or worse stomatitis by 8 weeks occurred in two (2% [95% CI 0·29–8·24]) of 85 patients (figure 2) in SWISH compared with 159 (33% [95% CI 28·8–37·4]) of 482 for the duration of BOLERO-2 study (p<0·0001).11Additionally, the proportion of stomatitis occurrence by 8 weeks for BOLERO-2 was calculated (figure 2; appendix p 9). Grade 2 stomatitis resolved to grade 1 or lower after 11 days and 15 days in the two affected SWISH patients, respectively. Two patients with clinical grade 2 stomatitis were classified as grade 1 due to the minimal impact of stomatitis on the visual analogue scale or Normalcy of Diet Scale.
Two patients developed oral candidiasis during antifungal prophylaxis (one treatment related and one not) that resolved with nystatin or oral fluconazole treatment. Six (30%) of 20 patients who received oral antifungal prophylaxis of nystatin developed stomatitis during the study. There was no apparent association between antifungal prophylaxis and prevention of stomatitis or oral candidiasis.
The prespecified analysis of the effect of dexamethasone treatment on patient-reported outcomes was also assessed. Among the 72 patients evaluable at 8 weeks, 68 (94%) reported an ability to maintain a normal diet (score 100) or a diet with no or few restrictions (score 90–100) by 8 weeks. One patient each had a diet score of 80 (all meat) or 50 (soft foods chewable foods) and two patients had a score of 40 (soft foods requiring no chewing). The mean oral pain score was less than 1 at all visits (range 0·1–0·6). In terms of oral hygiene, by the end of week 8, 75 (90%) of 83 patients brushed their teeth once or twice daily, 78 (94%) of 83 patients used a soft-bristled toothbrush, and 60 (73%) of 82 patients flossed daily, one person did not report their flossing routine. The baseline data for hygiene indicate that all 92 patients brushed at least once a day at baseline; of 91 patients, 62 flossed and 29 did not floss at baseline. In the 75 patients with complete ECOG scores available, 66 (88%) maintained or improved their ECOG status (appendix p 3).
Discussion
The results of the SWISH trial show that a commercially available, inexpensive, and well tolerated dexamethasone mouthwash is effective in the prevention of or reduction in the incidence and severity of all-grade stomatitis, especially grade 2 or worse stomatitis at 8 weeks compared with historical controls from the BOLERO-2 trial. Patient-reported outcomes (ability to maintain a normal diet and oral pain scores) further substantiated the efficacy of dexamethasone mouthwash. Reduction of stomatitis and tolerability of the dexamethasone mouthwash might have contributed to the favourable dose intensities for everolimus recorded in this trial; everolimus dose intensity was slightly higher than in BOLERO-2. Good oral hygiene, compliance with administration of mouthwash, and patient education about early onset and management of stomatitis might have further improved tolerability and adherence.
The treatment goal for patients with advanced breast cancer is to delay disease progression while minimising treatment-related adverse events. mTOR inhibitor-associated stomatitis is a mTOR class effect that can diminish oral food intake, drug adherence, and quality of life.5 Given the clinical benefit and potential long-term use of everolimus and exemestane, this study provides compelling evidence of a practical management strategy to effectively prevent or reduce a class-specific toxicity associated with everolimus.
The SWISH study population represents a real-world practice population of patients who were eligible for everolimus and exemestane therapy. As such, small differences in patient characteristics exist between the SWISH and BOLERO-2 populations based on study design. The manner in which the SWISH case report forms were completed made it challenging to ascertain the setting and specific regimens of previous chemotherapy, making it difficult to determine the proportion of patients with previous chemotherapy in specific settings (although the major chemotherapies used in this study population are listed in table 1). Overall, patients in this trial seem to be less heavily pretreated than those in BOLERO-2, which could suggest that they were less prone to developing stomatitis. However, the risk of stomatitis does not seem to correlate with the number of previous lines of therapy. Data from BOLERO-4,16 in which everolimus and letrozole were administered in the first-line metastatic setting, showed all-grade stomatitis in 137 (68%) of 202 patients and grade 3 stomatits in 13 (6%) of 202 patients, which are similar to the rates reported in BOLERO-2.
Although our study design did not include more than four cycles of the study-supplied steroid mouthwash and did not report on the proportion of patients with stomatitis after stopping the mouthwash, most stomatitis events from the safety set occurred within the first 8 weeks of treatment, which is consistent with larger datasets.3 In a meta-analysis of phase 3 trials across indications, 89% of first episodes of stomatitis occurred by 8 weeks.3 In BOLERO-2, 83% of grade 2 or worse stomatitis events occurred by 8 weeks, further supporting the rationale that the 8 week cutoff is sufficient to capture most events. Clearly, dexamethasone prophylaxis had a profound effect in SWISH: it reduced the proportion of all-grade stomatitis by 65% and grade 2 or worse stomatitis by 91% compared with BOLERO-2 by 8 weeks. This pattern reinforces the importance of prophylaxis initiation concurrent with everolimus and exemestane treatment and the rationale for an early and short-term intervention. As such, a reasonable approach based on these data would be to use the steroid-based mouthwash for the first two to four cycles of everolimus and exemestane treatment and then on an as-needed basis for signs or symptoms of stomatitis. Concurrent treatment with a steroid-based mouthwash and mTOR inhibitor during the period of highest risk for stomatitis is an effective strategy to maximise the benefit-to-risk ratio for patients. This approach has the potential to allow broader use of everolimus and exemestane with less toxicity in other indications for which the incidence of stomatitis ranges from 59% in advanced renal cell carcinoma3 to 70% in advanced neuroendocrine tumours.2
A strength of our study was that the eligibility criteria reflected the regulatory approved indication for everolimus and exemestane treatment. The most frequently reported adverse events were consistent with those reported in previous trials of everolimus and exemestane in breast cancer;5, 7 no new safety concerns were noted. Adverse events associated with the dexamethasone mouthwash were minimal (two patients had oral candidiasis), suggesting that the mouthwash presents minimal risk to patients. More than 74% of patients continued the mouthwash for more than 8 weeks, suggesting a benefit in the prevention or treatment of a later event of stomatitis with minimal risk. However, whether or not continuous preventive usage of dexamethasone mouthwash over a longer period could further increase the incidence of candidiasis remains unknown.
To the best of our knowledge, SWISH is the first and largest stomatitis prevention study combining therapeutic management with patient engagement to reduce the incidence and severity of mTOR inhibitor-associated stomatitis in women with hormone receptor-positive metastatic breast cancer. Our results are consistent with anecdotal reports of steroid-based oral rinses for prevention or amelioration of everolimus-related stomatitis.3, 8, 10, 17 These findings are further supported by interim results from a study of 47 patients treated with everolimus, an aromatase inhibitor, and prophylactic hydrocortisone-based or prednisolone oral solution for 12 weeks.18 Specifically, SWISH patients (in the safety set) with 13 weeks' median exposure to everolimus and exemestane reported an incidence of all-grade stomatitis (25 [27%] of 92 patients) that was similar to that reported by Jones and colleagues18 (12 [25%] of 48) in their 12 week study. Grade 2 or worse stomatitis was reduced by more than three times in both SWISH (9% in the safety set) and in Jones and colleagues' study (9%) compared with the 8 week incidence in BOLERO-2 (27%). Several ongoing clinical studies are assessing the effect of preventive and therapeutic rinses on oral mucositis or stomatitis in patients receiving everolimus and exemestane (NCT02229136, NCT02376985, and NCT01265810). These data will shed further light on the role of steroid-based mouthwash in the prevention of stomatitis.4
We recognise that our study has some limitations. This single-arm study does not have a control population; an indirect comparison with historical controls was used. Patients from BOLERO-2 served as a benchmark for the 8 week incidence of stomatitis, an endpoint that was valid because most events were observed within this timeperiod.3SWISH patients seemed to be less heavily pretreated than those in BOLERO-2, which might affect the risk of stomatitis. For the efficacy analyses in SWISH, grade of stomatitis was assessed as a composite score based on investigator confirmation of stomatitis, visual analogue scale, and Normalcy of Diet Scale, whereas it was assessed with CTCAE 3.0 criteria in BOLERO-2. A phase 2, non-randomised trial design for SWISH was chosen because of both ethical and technical issues. For example, it would be unethical to randomly assign patients to best supportive care because this approach has been shown to have a minimal prophylactic effect.19 Non-randomised, single-arm studies might have limited generalisability to populations not included in the study or might not be valid to compare with other studies.20 Confounding variables and unknown factors can bias results,21 including the use of video instructions on stomatitis and good oral hygiene in this trial, which would have improved adherence and outcomes compared with a real-world situation. However, the minimal change from baseline for oral hygiene scores suggests at most a modest impact. Potential bias was minimised by analysis of multiple datasets of stomatitis in patients treated with everolimus and exemestane.
The SWISH trial showed that prophylactic use of a commercially available, inexpensive oral dexamethasone mouthwash resulted in a more than ten-times reduction in the incidence of grade 2 or worse stomatitis at 8 weeks compared with BOLERO-2. Steroid prophylaxis should be considered a new standard of oral care for postmenopausal patients who are receiving everolimus and exemestane for treatment of hormone receptor-positive, HER2-negative advanced breast cancer and metastatic breast cancer, especially in the first 8 weeks of treatment and as needed thereafter. In the future, steroid prophylaxis might be considered as a treatment option in other diseases for which everolimus is indicated (advanced renal cell carcinoma, subependymal giant cell astrocytoma associated with tuberous sclerosis complex, and advanced neuroendocrine tumours).
References
- 1
-
J Lauring, BH Park, AC WolffThe phosphoinositide-3-kinase-Akt-mTOR pathway as a therapeutic target in breast cancerJ Natl Compr Canc Netw, 11 (2013), pp. 670-678
- 2
-
Afinitor (everolimus) prescribing information, Novartis Pharmaceuticals Corp, East Hanover, NJ (2016)
- 3
-
J Divers, J O'ShaughnessyStomatitis associated with use of mTOR inhibitors: implications for patients with invasive breast cancerClin J Oncol Nurs, 19 (2015), pp. 468-474
- 4
-
HS Rugo, KI Pritchard, M Gnant, et al.Incidence and time course of everolimus-related adverse events in postmenopausal women with hormone receptor-positive advanced breast cancer: insights from BOLERO-2Ann Oncol, 25 (2014), pp. 808-815
- 5
-
HS Rugo, GN Hortobagyi, J Yao, et al.Meta-analysis of stomatitis in clinical studies of everolimus: incidence and relationship with efficacyAnn Oncol, 27 (2016), pp. 519-525
- 6
-
R Shameem, M Lacouture, S WuIncidence and risk of high-grade stomatitis with mTOR inhibitors in cancer patientsCancer Invest, 33 (2015), pp. 70-77
- 7
-
DA Yardley, S Noguchi, KI Pritchard, et al.Everolimus plus exemestane in postmenopausal patients with HR(+) breast cancer: BOLERO-2 final progression-free survival analysisAdv Ther, 30 (2013), pp. 870-884
- 8
-
AP Pilotte, MB Hohos, KM Polson, TM Huftalen, N TreisterManaging stomatitis in patients treated with Mammalian target of rapamycin inhibitorsClin J Oncol Nurs, 15 (2011), pp. e83-e89
- 9
-
S Sonis, N Treister, S Chawla, G Demetri, F HaluskaPreliminary characterization of oral lesions associated with inhibitors of mammalian target of rapamycin in cancer patientsCancer, 116 (2010), pp. 210-215
- 10
-
O Nicolatou-Galitis, A Nikolaidi, I Athanassiadis, E Papadopoulou, SSonisOral ulcers in patients with advanced breast cancer receiving everolimus: a case series report on clinical presentation and managementOral Surg Oral Med Oral Pathol Oral Radiol, 116 (2013), pp. e110-e116
- 11
- Rugo HS, Gnant M, Geberth M, et al. Everolimus-related adverse events: safety insight from BOLERO-2. St Gallen International Breast Cancer Conference; St Gallen, Switzerland; March 13–16, 2013. Poster 274.
- 12
-
MA List, LL D'Antonio, DF Cella, et al.The performance status scale for head and neck cancer patients and the functional assessment of cancer therapy-head and neck scale. A study of utility and validityCancer, 77 (1996), pp. 2294-2301
- 13
-
MA List, C Ritter-Sterr, SB LanskyA performance status scale for head and neck cancer patientsCancer, 66 (1990), pp. 564-569
- 14
-
D Gould, D Kelly, L Goldstone, J GammonExamining the validity of pressure ulcer risk assessment scales: developing and using illustrated patient simulations to collect the dataJ Clin Nurs, 10 (2001), pp. 697-706
- 15
-
ST Sonis, JP Eilers, JB Epstein, et al.Validation of a new scoring system for the assessment of clinical trial research of oral mucositis induced by radiation or chemotherapy. Mucositis Study GroupCancer, 85 (1999), pp. 2103-2113
- 16
-
M Royce, C Villaneuva, M Ozguroglu, et al.BOLERO-4: phase 2 trial of first-line everolimus (EVE) plus letrozole (LET) in estrogen receptor–positive (ER+), human epidermal growth factor receptor 2–negative (HER2−) advanced breast cancer (BC)Ann Oncol, 27 (2016), pp. 68-69
- 17
-
MA de Oliveira, EMF Martins, Q Wang, et al.Clinical presentation and management of mTOR inhibitor-associated stomatitisOral Oncol, 47 (2011), pp. 998-1003
- 18
-
VL Jones, LL Jensen, KJ McIntyre, et al.Evaluation of miracle mouthwash (MMW) plus hydrocortisone versus prednisolone mouth rinses as prophylaxis for everolimus-associated stomatitis: preliminary results of a randomized phase II studyCancer Res, 76 (2016)P1-15-06-P1-15-06.
- 19
-
F Schütz, E-M Grischke, T Decker, et al.Stomatitis in patients treated with everolimus and exemestane—results of the third interim analysis of the non-interventional trial BRAWOCancer Res, 76 (2016)P4-13-08-P4-13-08.
- 20
-
L Zheng, SL Rosenkranz, B Taiwo, MF Para, JJ Eron Jr, MD HughesThe design of single-arm clinical trials of combination antiretroviral regimens for treatment-naive HIV-infected patientsAIDS Res Hum Retroviruses, 29 (2013), pp. 652-657
- 21
-
SR EvansClinical trial structuresJ Exp Stroke Transl Med, 3 (2010), pp. 8-18






 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 