Abstract
Octocog alfa, antihaemophilic factor, plasma/albumin free method (Advate®) is a recombinant, human, full-length coagulation factor VIII that does not contain human- or animal-derived plasma proteins. It is indicated for the control and prevention of bleeding episodes, for perioperative management and for routine prophylaxis in children and adults with haemophilia A. This article reviews the pharmacological properties, therapeutic efficacy and tolerability of Advate® in these patients.
In previously treated paediatric and adult patients with moderately severe or severe haemophilia A, Advate® administered prophylactically, on demand or during surgery was effective for the prevention and treatment of bleeding episodes in three pivotal, uncontrolled clinical trials. The haemostatic efficacy of Advate® in these trials was rated as ‘excellent’ or ‘good’ in most bleeding episodes, with the majority of episodes being managed with one infusion. These findings were supported by pooled analyses of clinical trials and routine clinical practice studies, including the Post-Authorization Safety Study.
Additionally, in a comparative study, routine prophylaxis with Advate® administered in a standard regimen or in a pharmacokinetic-tailored regimen was effective for the prevention of bleeding episodes in patients with moderately severe or severe haemophilia A, with no significant difference between the two regimens in terms of efficacy. Moreover, any routine prophylaxis with Advate® was found to be more effective in preventing bleeding episodes than on-demand therapy with Advate®.
Advate® was generally well tolerated in clinical trials and postmarketing studies, with the most common treatment-emergent adverse events being pyrexia and headache. Serious adverse events with Advate® therapy are development of high-titre factor VIII inhibitors (usually in previously untreated patients) and hypersensitivity reactions. As expected, the incidence of factor VIII inhibitors (any titre) appeared to be lower in previously treated patients (≤1.8%) than in previously untreated patients (≤20%).
There are no head-to-head comparative trials of Advate® and other factor VIII concentrates. Nevertheless, current evidence indicates that Advate® is an effective option for the management of paediatric and adult patients with haemophilia A.
1. Introduction
Haemophilia A is an X chromosome-linked bleeding disorder caused by a congenital deficiency or defect in coagulation factor VIII, resulting in insufficient coagulant activity and prolonged clotting times.[1] While normal plasma levels of factor VIII activity range between 50% and 150%, patients with mild, moderate and severe disease have factor VIII levels of 5–50%[2] (or 5–40%[3]), 1–5%[2,3] and <1%,[2,3] respectively. The proportion of patients with mild haemophilia varies between countries, as well as overtime in the same country.[4] In the US, of the patients with haemophilia, ≈15% have moderate and ≈60% have severe haemophilia,[2] whereas in a survey conducted in Sweden in 1980, 54%, 17% and 29% of patients were reported as having mild, moderate or severe haemophilia A, respectively.[5]
Patients with severe haemophilia A are at risk of uncontrolled, often spontaneous bleeding into joints, muscles or internal organs, or bleeding after injury, trauma or surgery.[1] In the absence of treatment, bleeding episodes may lead to serious complications, including permanent, disabling joint, muscle and nerve damage, loss of musculoskeletal function or death.[1] According to a global survey conducted by the World Federation of Haemophilia (WFH) in 106 countries, 125 049 people were affected by haemophilia A in 2010.[6]
Factor VIII replacement therapy has been the mainstay in the treatment of haemophilia A and has progressed over time from the use of blood transfusions prior to the 1940s to the use of plasma concentrates in the 1950s, cryoprecipitates in the mid 1960s, plasma-derived factor VIII concentrates in the late 1960s and recombinant products in the 1990s.[7,8] In the 1980s and 1990s plasma-derived factor VIII concentrates were linked with the transmission of blood-borne viruses, including HIV and hepatitis viruses.[1,9] However, the risk of viral transmission has been greatly reduced with the introduction of virus inactivation procedures and testing of donor populations, with no cases of viral transmission being documented since 1990.[9] Despite the improved safety of these products, the risk of viral transmission and potential transmission of prions (causative agent for classical or variant forms of Creutzfeldt-Jakob disease) have not been eliminated.[1,7]
Recombinant factor VIII concentrates were developed as safer alternatives to plasma-derived products. Three types of recombinant products are available, classified as the first-, second- and third-generation products based on the use of human- or animal-derived materials during manufacture and processing (section 7).[8,10] First-generation factor VIII concentrates require plasma proteins during manufacture and in the final formulation, second-generation products use plasma-derived proteins during manufacture, while third-generation products do not use any additional animal or human proteins during manufacture or in the final formulation,[1,8,10] thereby almost eliminating the risk of transmission of blood-borne pathogens.[10]
Octocog alfa, antihaemophilic factor (recombinant), plasma/albumin free method (Advate®) is one such third-generation factor VIII product.[1] This article reviews the pharmacological properties, therapeutic efficacy and tolerability of Advate® in paediatric and adult patients with haemophilia A. Factor VIII activity is expressed as percentage of normal throughout the article.
2. Pharmacodynamic Properties
Advate® contains the recombinant coagulation factor octocog alfa, a 2332 amino acid glycoprotein (≈280 kDa) that is biologically equivalent to the factor VIII glycoprotein found in human plasma.[11] Factor VIII is involved in the intrinsic pathway of blood coagulation.[12] It circulates in the plasma as a tightly bound complex with von Willebrand factor (VWF).[12] After activation by thrombin or activated factor X, it dissociates from the complex and serves as a cofactor for the activation of factor X by activated factor IX.[11,12] Activated factor X converts prothrombin to thrombin, which in turn converts fibrinogen to fibrin, leading to the formation of a blood clot.[11]
Advate® is synthesized by genetically engineered Chinese Hamster Ovary cells and is purified from the culture medium by chromatography.[13] No additives of human or animal origin are employed in the culture and purification processes.[13] Lyophilized Advate® was stable across a variety of conditions, including at 5°C for 30 months (92% residual factor VIII activity), at room temperature for 18 months (≈80% residual activity) and at 40°C for 3 months (84% residual activity).[14] A 2-week excursion at 40°C did not compromise subsequent stability of the lyophilized powder when stored at 5–30°C.[14] Reconstituted samples retained on average 92% of their activity after 24 hours at room temperature[14] and were stable under conditions usually encountered during continuous infusion.[15]
In vivo studies in animals showed that the haemostatic efficacy of Advate® was similar to that of its predecessor, Recombinate® (a first-generation recombinant factor VIII antihaemophilic factor).[16] In patients with haemophilia A and prolonged activated partial thromboplastin time (aPTT), treatment with Advate® normalized aPTT over the effective dosing period.[13]
Data from patients with severe haemophilia A (factor VIII <1%) receiving Advate® as prophylaxis in two pivotal clinical studies[17,18] (see section 4 for study design details) showed that increasing time with factor VIII levels <1% was significantly (p ≤ 0.02) associated with an increase in the incidence of total bleeding episodes and bleeding in the joints. For each additional hour spent with factor VIII levels <1%, the annual bleeding rate increased by 2.2% in patients aged 1–6 years (n = 44) and 1.4% in patients aged 10–65 years (n = 99).[19] Decreased adherence to the frequency of prophylaxis was also significantly (p < 0.0001) associated with an increase in the incidence of any bleeding or bleeding in the joints in both subgroups of patients.[19]
3. Pharmacokinetic Properties
Discussed are the pharmacokinetic properties of Advate® based on data from two pivotal trials[17,18] (see section 4 for study design details) and several pooled analyses.[19-21] These are supplemented with data from the US prescribing information.[13]
The pharmacokinetic properties of Advate® in paediatric and adult patients with moderately severe to severe haemophilia A are summarized in table1
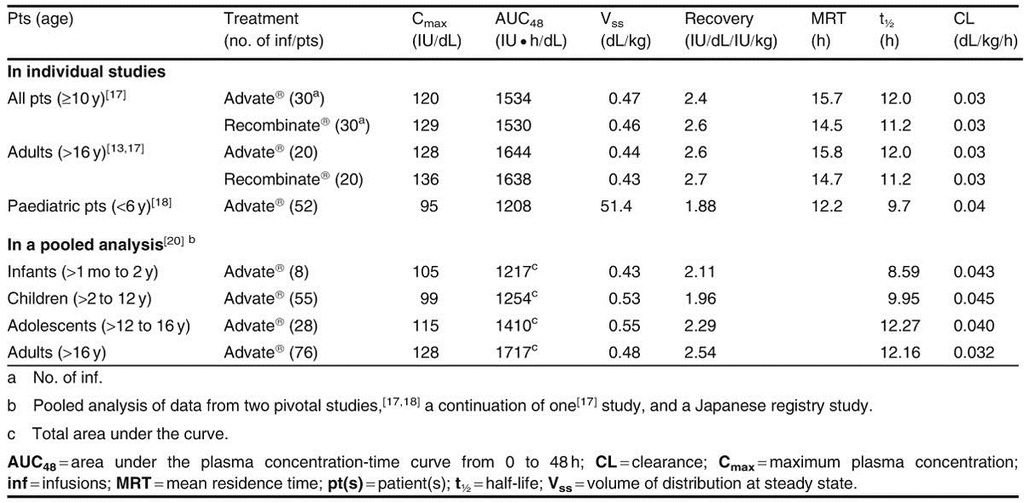
The area under the concentration-time curve (AUC), recovery and half-life (t½)of Advate® appeared to be higher, while its clearance appeared to be lower, in adolescents and adults than in infants and children (no statistical data were reported).[20]
Bioequivalence between Advate® (produced at pilot scale in Orth, Austria) and Recombinate® (table I) was demonstrated as the 90% confidence intervals of the mean of the logarithm of the ratios of AUC from 0 to 48 hours and adjusted recovery for Advate® and Recombinate® were within the predefined limits of 80% and 125%.[17] There was no significant change in the in vivo recovery of Advate® at the onset of treatment and after ≥75 exposure days.[13]
Advate® was also shown to be bioequivalent to Refacto® (a recombinant B-domain deleted factor VIII), as the 90% confidence intervals of the ratios of the geometric least squares mean of maximum plasma concentration (Cmax) and AUCs (from time 0 to infinity or until the last measurable concentration) of Refacto® to Advate® were within the predefined limits of 80% and 125%.[22]
A comparison of pharmacokinetic properties between children and adults showed that patients aged 1–6 years had lower in vivo recovery (between-group difference −0.58 IU/dL/IU/kg), higher weight-adjusted clearance (between-group difference 1.08 mL/h/kg) and shorter t½ (between-group difference −1.8 hours) of Advate® than patients aged 10–65 years.[21] However, it was estimated that up to approximately half of the total observed differences in Cmax, in vivo recovery and t½ were because of a reduced pharmacokinetic blood sampling schedule for children.[21]
Intrapatient variance (13–39%) in the pharmacokinetics of Advate® was less than the interpatient variance (61–87%) in a comparison between two infusions administered 72 hours to 30 days apart or ≥75 exposure days apart, which supports the use of pharmacokinetic measurements for tailoring of Advate® dosage.[21] Because of the variability in the pharmacokinetic response to Advate®, patients receiving the same dosage of the agent may vary markedly in the levels of factor VIII achieved. In patients with moderately severe or severe haemophilia A (n = 48 aged 1–6 years and 99 aged 10–65 years) who were receiving Advate® for prophylaxis, the t½ and dose frequency of Advate® were shown to have larger effects on trough factor VIII levels and the time per week with factor VIII <1% than its in vivo recovery or the infused dose per kg.[23] In patients aged 1–6 years, a decrease in the t½ of Advate® significantly (p = 0.01) increased the incidence of all bleeding episodes.[19]
Recently, a population pharmacokinetic, 2-compartment model has been developed that describes the relationship between the pharmacokinetics of Advate® and age and bodyweight, and may be useful for designing prophylactic schedules for individual patients.[24]
4. Therapeutic Efficacy
This section focuses on clinical trials (n > 50 patients) evaluating the efficacy of Advate® for prophylaxis and/or on-demand treatment of bleeding episodes (section 4.1) and for perioperative management (section 4.2) in patients with haemophilia A. Also discussed briefly are data from pooled analyses (section 4.3) and routine clinical practice studies (section 4.4).
Patients in all trials received Advate® regimens only; there were no comparisons with other factor VIII concentrates. Efficacy outcomes in these trials included the number of infusions required to manage bleeding episodes, the annual bleeding rates, and the patient's or investigator's efficacy ratings[17,18,25] (see table2 for definition of ratings, where reported).
4.1 For Prophylaxis and Treatment of Bleeding
Several trials in previously treated patients with moderately severe or severe haemophilia A[17,18,26] or in patients with mild to severe haemophilia A[25] assessed the efficacy of Advate® for prophylaxis and treatment of bleeding episodes (see table III for key eligibility criteria and study design details).
4.1.1 In Patients of Mixed Ages
An open-label, uncontrolled study in previously treated patients (aged 10–65 years) with moderately severe or severe haemophilia A showed that treatment with Advate® was effective for the prevention and treatment of bleeding episodes.[17] Following prophylactic treatment for ≥75 exposure days, the haemostatic efficacy of Advate® was rated as ‘excellent’ or ‘good’ in 86% of bleeding episodes, with the majority (81%) of episodes being managed with one infusion (table IV).[17]
A total of 510 bleeding episodes occurred during the entire study (including the pharmacokinetic phase, prophylactic phase [≥75 exposure days] and extended treatment); 45% of these were trauma related, 32% were spontaneous and the remainder were of indeterminate aetiology.[17] Of the 510 episodes, 274 occurred in 70% of patients during the prophylactic phase, while 30% of patients did not experience bleeding episodes during this period. The mean annual bleeding rate was 6.3 episodes/patient, with the rate significantly (p < 0.03) lower in patients who were adherent to prophylactic treatment than in those who were not adherent (4.4 vs 9.9 episodes/patient; post hoc analysis). Adherence was defined as treatment with Advate® dosages of ≥25–40 IU/kg per infusion for at least 80% of infusions and at least three or four infusions per week for at least 80% of weeks during the initial 75 exposure days.[17]
Results from this study were supported by those from an open-label continuation of this study (available from a review article[1]), during which patients (n = 81 evaluable) continued to receive Advate® as standard prophylaxis (25–40 IU/kg three or four times/week), modified prophylaxis (as determined by the investigator) or on-demand treatment. No bleeding episodes were reported in 11 of 81 patients, all of whom had been receiving prophylaxis for at least 90% of the prophylactic phase of the study. The remaining 70 patients experienced 822 bleeding episodes; 820 of these were treated with Advate®, with 88.5% of episodes being managed with one or two infusions. The annual bleeding rates in patients receiving modified prophylaxis (n = 53), standard prophylaxis (n = 54) and on-demand treatment (n = 9) were 4.8, 6.0 and 18.5 episodes/patient, respectively; the annual bleeding rate in patients who were compliant to therapy was 4.5 episodes/patient compared with 7.9 episodes/patient in those who were noncompliant to treatment.[1]
Advate® was also effective for the treatment of bleeding episodes when used as on-demand therapy in Chinese patients (aged 7–53 years) with mild to severe haemophilia A.[25] During a 6-month observation period, the efficacy of Advate® was rated as ‘excellent’ in 83% of bleeding episodes, with 96% of episodes treated with one or two infusions (table IV).[25]
4.1.2 In Paediatric Patients
An open-label, uncontrolled study showed that Advate® was effective for preventing and treating bleeding episodes in previously treated paediatric patients (aged <6 years) with moderately severe or severe haemophilia A.[18] Following prophylaxis or on-demand treatment for ≥50 exposure days, the haemostatic efficacy of Advate® was rated as ‘excellent’ or ‘good’ in 94% of bleeding episodes, with the majority (78%) of episodes being managed with one infusion (table IV).[18]
Of the 409 evaluable bleeding episodes, 48.4% (198 episodes) were trauma related, 17.8% (73) were spontaneous, 0.5% (2) were postoperative and the aetiology was uncertain for 33.0% (136) of episodes.[18] The median annual bleeding rates for all body sites with standard prophylaxis (n = 21 patients), modified prophylaxis (n = 37) and on-demand treatment (n = 5) were 4.0, 4.4 and 24.4 episodes/patient, respectively.[18]
There were 112 episodes of joint bleeds, which occurred in 33.3% (7 of 21) of patients receiving standard prophylaxis, 32.4% (12 of 37) of patients receiving modified prophylaxis and 80% (4 of 5) of patients receiving on-demand treatment.[18] The median annual bleeding rate for joint bleeds in patients receiving prophylaxis (standard or modified) was 0 (range 0–6.1) compared with 14.2 (range 0–34.5) in those receiving on-demand therapy, with most joint bleeds in the latter group attributed to non-traumatic causes (annual incidence of 13.0 vs 1.2 traumatic joint bleeding episodes/patient).[18]
4.1.3 Routine Prophylaxis
An open-label study in previously treated patients (aged >7 to <65 years) with moderately severe or severe haemophilia A compared the efficacy of Advate® administered as standard prophylaxis versus pharmacokinetic-tailored prophylaxis, and compared its efficacy as prophylaxis versus on-demand treatment.[26] Briefly, after 6 months of on-demand treatment with Advate®, patients were randomized to 12 months of standard prophylaxis (administered every second day) or pharmacokinetic-tailored prophylaxis (administered every third day) with Advate®.[26]
In terms of the prevention of bleeding episodes, the haemostatic efficacy of Advate® in a standard prophylaxis regimen did not differ significantly from that in a pharmacokinetic-tailored prophylaxis regimen, as assessed by the mean (1.6 vs 1.9 episodes/patient) or median (figure 1a) transformed annual bleeding rates (primary endpoint).[26] Moreover, any Advate® prophylaxis (i.e. standard or pharmacokinetic-tailored) was more effective in preventing bleeding episodes than Advate® on-demand treatment, as indicated by a 99.4% reduction in the median annual bleeding rates with any prophylaxis relative to on-demand therapy (figure 1b; secondary endpoint). Any prophylaxis was associated with significantly (p < 0.0001) lower annual bleeding rates for spontaneous (0 vs 32.2 episodes/patient) and traumatic (0 vs 11.5 episodes/patient) bleeding episodes, and joint (1.0 vs 38.3 episodes/patient) and non-joint (0 vs 3.9 episodes/patient) bleeding episodes than on-demand treatment.[26]
All patients receiving on-demand treatment had bleeding episodes (total of 1640 episodes over the 6-month treatment period). By contrast, 33.3% of patients receiving prophylaxis (13 of 32 receiving standard prophylaxis and 9 of 34 receiving pharmacokinetic-tailored prophylaxis) did not have bleeding episodes during the 12-month treatment period; 19 patients receiving standard prophylaxis and 25 patients receiving pharmacokinetic-tailored prophylaxis experienced 104 and 141 bleeding episodes, respectively.[26]
With regard to the treatment of bleeding episodes during standard prophylaxis, pharmacokinetic-tailored prophylaxis and on-demand therapy, most bleeding episodes (86%, 91% and 89%, respectively) were treated with one or two infusions of Advate® and most treatments (83%, 78% and 89%) were rated as ‘excellent’ or ‘good’ by the patients, regardless of the regimen.[26] The median annual consumption of Advate® in the standard and pharmacokinetic-tailored prophylactic regimens was 5768.2 and 5197.8 IU/kg; the median annual consumption of Advate® for on-demand therapy was significantly (p < 0.0001) less than that for any prophylaxis (2152.2 vs 5733.3 IU/kg), which was expected.[26]
In terms of health-related quality of life (HR-QOL; assessed in 57 patients aged ≥14 years), there were no significant differences in median Short Form-36 scores between patients receiving standard prophylaxis and those receiving pharmacokinetic-tailored prophylaxis.[26] However, the median scores for the bodily pain domain and physical component summary measure were clinically (larger than the estimated minimal important difference of 3 points) and significantly (p ≤ 0.0007) improved at the end of prophylaxis relative to the end of on-demand treatment.[26]
4.2 For Perioperative Management
An open-label, multicentre, uncontrolled study showed that Advate® was effective for perioperative haemostatic management in previously treated patients (aged ≥5 years) with moderately severe or severe haemophilia A (see table V for study design details).[28] After a loading dose administered prior to surgery, plasma factor VIII levels reached the target range within 10–30 minutes in 59 of the 63 procedures where levels were reported. In four procedures, postinfusion target factor VIII levels were not achieved, probably because of under dosing in one procedure and low recovery in two patients (50% and 65% of predicted within 30 minutes). However, there were no untoward clinical effects of below target factor VIII levels in these four patients and none of them required supplemental preoperative infusions, red blood cells or antifibrinolytic agents during or after surgery.[28]
The intraoperative and postoperative haemostatic efficacy of Advate® was rated as ‘excellent’ or ‘good’ in ≥91% of procedures (table V).[28] Similar results were observed when bolus and continuous infusions were compared, with the intraoperative and postoperative (at the time of discharge) efficacy of Advate® rated as ‘excellent’ or ‘good’ in 100% of procedures. Its postoperative efficacy at the time of drain removal was rated as ‘excellent’ or ‘good’ in 100% of procedures receiving continuous infusion and in 88% of those receiving bolus infusion; 12% of procedures were rated as ‘fair’ with the bolus infusion.[28] Actual blood loss was less than predicted in 27 of the 58 procedures that had data available; 28 procedures had blood loss within the predicted range.[28] Three procedures (total hip replacement, nevi excision and central venous catheter removal) had greater than predicted blood loss; however, the intraoperative efficacy of Advate® in these procedures was rated as excellent (one procedure [nevi excision]) or good (two procedures).[28]
Advate® also controlled bleeding episodes that occurred during home replacement therapy, with its efficacy rated as ‘excellent’ or ‘good’ in most episodes (table V).
In addition to this study, limited data suggested that Advate® was effective for the perioperative management of haemostasis in paediatric patients. Five of seven paediatric patients who underwent surgical procedures while participating in a previously discussed study[18] (section 4.1.2) were evaluable per-protocol; in the three patients who had efficacy assessments, the intraoperative and postoperative efficacy of Advate® was rated ‘excellent’ or ‘good’.[18] Additionally, in a pooled analysis (presented as an abstract)[29] of data from paediatric patients participating in two studies,[18,28] the intraoperative efficacy of Advate® was rated as ‘excellent’ or ‘good’ in 14 of 15 procedures and ‘fair’ in one of the procedures, while its postoperative efficacy was rated ‘excellent’ or ‘good’ in 17 of 17 procedures.
4.3 Pooled Analyses
A retrospective, pooled analysis[20] of data from five prospective studies in previously treated patients (n = 211; aged 1–65 years) and one study in previously untreated (n = 29; aged 0.6 months to 2.2 years) patients with moderately severe or severe haemophilia A supported the findings of the clinical studies (three[17,18,28] of the studies are fully published and have been discussed previously in sections 4.1 and 4.2). In total, 2100 bleeding episodes were reported in 171 patients, of which 58% were spontaneous or of unknown aetiology and 42% were trauma-related episodes.[20] The treatment efficacy of Advate® was rated as ‘excellent’ or ‘good’ in the majority (88%) of bleeding episodes, with most (90%) episodes being managed with one or two infusions.[20] With regard to the prevention of bleeding, patients receiving protocol- or investigator-defined prophylaxis with Advate® had 1.6- to 2-fold lower median annual bleeding rates than patients receiving on-demand treatment (3.0 and 3.9 vs 6.3 bleeding episodes/patient). Moreover, patients who complied with prophylaxis had significantly (p = 0.006) fewer bleeding episodes than patients who were noncompliant to therapy (annual bleeding rate 3.2 vs 4.2 episodes/patient); compliance was defined as having received at least the minimum prescribed dose of Advate® for at least 80% of infusions and adherence to treatment for at least 80% of the study weeks.[20]
Another retrospective, pooled analysis[30] analysed data from two pivotal studies in patients (aged 10–65[17] or <6 years;[18] see section 4.1.1) with moderately severe or severe haemophilia A who received Advate® as prophylaxis and/or on-demand therapy for bleeding episodes (total n = 145). Overall, 700 bleeding episodes were reported in 110 patients, all of whom were receiving prophylaxis. The annual incidences of bleeding in paediatric patients aged 1–6 years (n = 46), in those aged 10–17 years (n = 47) and in adult patients aged 18–65 years (n = 52) were 3.1, 3.3 and 2.1 episodes/patient, respectively. The number of joint bleeds increased with age, from 21% in patients aged 1–6 years to 50% in those aged 10–17 years and 62% in adult patients (both p < 0.01 vs children aged 1–6 years). Moreover, significantly (p < 0.01) more joint bleeds occurred in summer in patients aged 10–17 or 18–65 years than in those aged 1–6 years (43% and 46% vs 21%).[30]
4.4 Routine Clinical Practice
Routine clinical practice studies also supported the findings of the clinical trials and showed that Advate® was effective for the prevention and control of bleeding episodes and for perioperative haemostatic management in patients with haemophilia A.[27]
PASS (Post-Authorization Safety Study), which was conducted in the US and EU, monitored patients with differing haemophilia A severities and medical histories during 12 months of prophylaxis or on-demand therapy.[27] Of the 521 patients who were treated with one or more infusions of Advate®, ≈87% of patients had moderately severe or severe haemophilia A (baseline factor VIII level ≤2%). The majority (88%) of patients had received previous treatment with factor VIII concentrates (>50 exposure days); of the remaining patients, 12 had 0–3 prior exposure days, 17 had 4–50 prior exposure days and 32 had an estimated ≤50 prior exposure days to factor VIII concentrates. In the 408 evaluable patients who received on-demand therapy, the haemostatic efficacy of Advate® was rated as ‘excellent’ or ‘good’ in 386 (95%) patients. Similarly, in the 276 evaluable patients who received continuous prophylaxis during the study, the efficacy of Advate® was rated ‘excellent’ or ‘good’ in 255 (92%) patients, and in the 16 evaluable procedures where Advate® was administered for surgical prophylaxis, its efficacy was rated ‘excellent’ or ‘good’ in 100% of procedures.[27]
In the Japanese PASS, 383 patients with haemophilia A (82.6% with moderately severe or severe disease [factor VIII <2%]) were registered as of 20 December 2010, and case report forms were collected from 362 patients; at study entry, 321 patients had ≥51 days and 41 patients had <50 days of exposure to factor VIII concentrates (abstract presentation).[31] The efficacy of Advate® was rated as ‘excellent’ or ‘good’ in 94.7% of bleeding episodes during prophylaxis and 97.5% of bleeding episodes during on-demand therapy over a mean observation period of 15 months.[31] In previously untreated (≤3 exposure days at study entry; n = 66 as of 20 December 2010) patients with haemophilia A (81% severe, 14% moderate and 5% mild disease), 93.3% of bleeding episodes during prophylaxis and 96.5% of episodes during on-demand therapy were rated as ‘excellent’ or ‘good’ during a mean observation period of 13.3 months (abstract presentation).[32]
Another routine clinical practice study in previously treated patients with haemophilia A who switched to prophylaxis or on-demand treatment with Advate® showed that bleeding patterns and clotting factor consumption did not differ significantly before and after the treatment switch.[33] For example, during prophylaxis, the median consumption of Advate® was 2434 IU/kg/year versus 2252 IU/kg/year of other factor VIII concentrates, while the annual number of joint bleeds in the respective groups were 1.7 and 1.4 episodes/patient. The study included 82 patients with severe (71 patients), moderate (ten) or mild (one) haemophilia A (age range 0.5 to 67 years) who were required to have had ≥50 prior exposures to clotting factor concentrates and were to be followed-up for ≥1 year; data on 245 patient-years on Advate® and 165 patients-years on other products were analysed.[33]
5. Tolerability
Discussed is the tolerability of Advate® based on a pooled analysis[13,20] (of five clinical trials in previously treated patients and one clinical trial in previously untreated patients with moderately severe or severe haemophilia A), a routine prophylaxis trial[26] and postmarketing studies[27,31,32,34] (see section 4 for study design details). In the pooled analysis, as of March 2006, 234 patients had received a total of 44 926 infusions, the median duration of study participation was 370.5 (range 1–1256) days/patient and the median duration of exposure to Advate® was 128 (range 1–598) days/patient.[13,20] Treatment-emergent adverse events included those that occurred within 24 hours of the infusion, events that were considered related or possibly related to Advate®, or events for which causality was missing or indeterminate.[13]
Advate® as prophylaxis or on-demand treatment was generally well tolerated in clinical trials and postmarketing studies in patients with haemophilia A. Serious adverse events observed during Advate® therapy included hypersensitivity reactions and the development of high-titre factor VIII inhibitors (immunoglobulin G [IgG] antibodies that neutralize the function of factor VIII) necessitating alternative treatments.[13]
5.1 General Profile
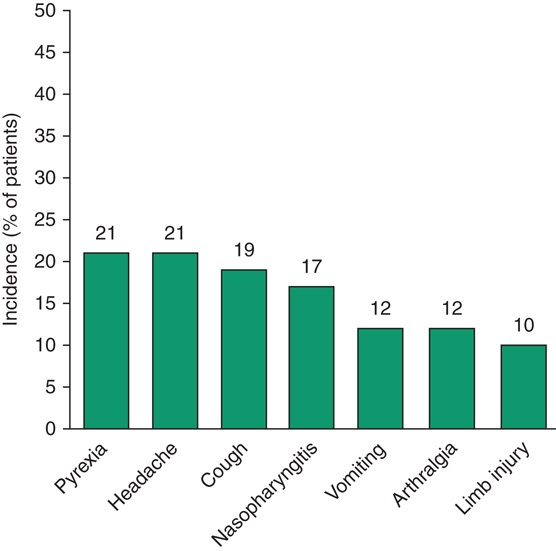
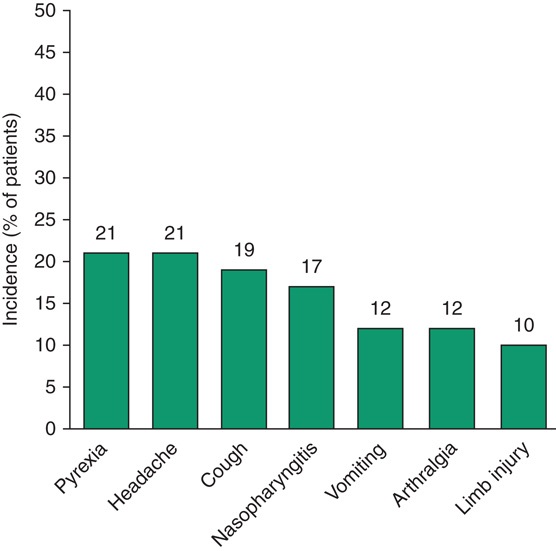
In the pooled analysis of six clinical trials, 2507 treatment-emergent adverse events occurred in 215 of 234 patients.[20] As would be expected during long-term treatment of haemophilia, the majority of these adverse events appeared to be related to trauma, intercurrent mild respiratory or gastrointestinal disease or complications of haemophilia.[20] The most common treatment-emergent adverse events associated with Advate® treatment were pyrexia and headache (figure 2).
Fifty-nine serious adverse events occurred in 37 patients, none of which resulted in death or serious sequelae or required withdrawal from therapy; five of these adverse events (development of factor VIII inhibitors in five previously untreated patients) were considered possibly treatment related (see section 5.2).[20] Of the nonserious adverse events, 51 events in 24 patients were considered treatment related, including headache, dysgeusia and pyrexia.[20]
In the routine prophylaxis trial,[26] 200 treatment-emergent adverse events were reported in 44 of 73 patients who received Advate® therapy (safety analysis set), of which 186 events in 41 patients were non-serious (19 events were considered treatment related) and 14 events in 11 patients were serious (one event was considered treatment related [a case of possible low-titre factor VIII inhibitor]). There were no deaths or withdrawals because of adverse events and no patient developed confirmed factor VIII inhibitors.[26]
5.2 Immunogenicity
In the Advate® clinical programme, which includes 270 previously treated patients who had ≥10 exposure days or a minimum study duration of 120 days, the incidence of factor VIII inhibitors was 0.37% (95% CI 0.02, 2.13).[26]
As noted earlier in section 5.1, 5 of 25 (20%) previously untreated patients (≤3 exposures to a factor VIII concentrate prior to study entry) developed factor VIII inhibitors following treatment with Advate®, which were detected at a median of 12 (range 1–80) exposure days.[20] Of these patients, four developed high-titre inhibitors (>5 Bethesda Units [BU]) and one developed low-titre inhibitors.[13] The 95% confidence intervals for the risk of developing any factor VIII inhibitors were 8.2%, 39.9%.[20]
Patients receiving Advate® were also assessed for the development of antibodies against heterologous proteins, including Chinese hamster ovary (CHO) cell protein, murine IgG, and human VWF.[13] Of the 182 patients assessed, upward trends in anti-CHO and anti-murine IgG antibody titres were observed in three and ten patients, respectively, while repeated but transient elevations in these antibodies were observed in four and two patients. Four patients with antibody (not specified) elevations reported isolated events of urticaria, pruritus, rash and slightly elevated eosinophil counts; however, these events did not reoccur during numerous repeat exposures to Advate® and the causal relationship with the agent has not been established. None of the 181 patients who were assessed for anti-VWF antibodies showed evidence of a positive serological response.[13]
5.3 Postmarketing Experience
There have been reports of serious hypersensitivity and anaphylactic reactions (e.g. dizziness, rash urticaria) and factor VIII inhibitor formation (usually in previously untreated patients) during postmarketing experience with Advate®.[13] Other adverse events reported were injection-site reactions, chills, fatigue/malaise, chest discomfort/pain and less than expected therapeutic effect. As these adverse events are reported voluntarily, their frequency and causal relationship with Advate® are uncertain.[13]
In the US and EU PASS, among haemophilia A patients who were receiving Advate®, ten nonserious treatment-related adverse events were reported in seven patients, of which two events (headache and fatigue in the same patient) were severe.[27] In addition, ten serious treatment-related adverse events were reported in ten patients (two patients had hypersensitivity reactions, one patient had reduced therapeutic effect of the agent and seven patients developed factor VIII inhibitors).
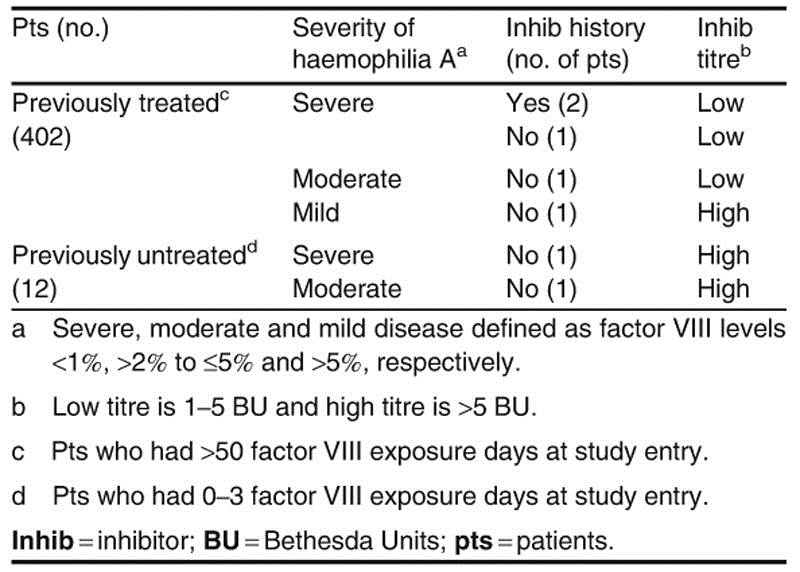
With regard to factor VIII inhibitor development, in the US and EU PASS, five previously treated patients (>50 exposure days at study entry) developed inhibitors (table VI) after 19–36 days of exposure to Advate® (data for exposure days was missing for two patients).[27] The incidence of inhibitor development in previously treated patients with factor VIII ≤2% of normal was 0.75% (95% CI 0.15, 2.17) and the incidence of inhibitor development in previously treated patients who had no inhibitor history was 0.29% (95% CI 0.01, 1.59).[27] In the Japanese PASS, among haemophilia A patients who were receiving Advate®, none of the previously treated patients with >51 exposure days at study entry developed factor VIII inhibitors (incidence 0% [95% CI 0, 1.31]); of the 328 patients without inhibitor history, two patients with <50 exposure days at study entry developed low-titre inhibitors (incidence 0.61% [95% CI 0.77, 2.19]).[abstract presentation].[31] No patient who switched from a first or second generation factor VIII concentrate to Advate® developed inhibitors.[31]
A retrospective review of case records of previously treated Irish haemophilia A patients (the majority [89%] of whom had severe haemophilia A [factor VIII <1%]) who switched to Advate® treatment in 2006, showed that 2 of 113 patients (1.8%) developed factor VIII inhibitors.[34] Of the 96 patients with no inhibitor history, a 1-year old child with three prior exposures to a factor VIII product developed low-titre inhibitors after three exposures to Advate® and of the 17 patients who had an inhibitor history, a 4-year-old child developed inhibitors following intense therapy for an acute bleed (maximum titre 32 BU).[34]
In previously untreated patients (1–3 exposure days at study entry) in the US and EU PASS, 2 of 12 (17%) patients developed high-titre factor VIII inhibitors after 16 and 26 days of exposure to Advate® (table VI).[27] In previously untreated patients (<3 exposure days at study entry) in the Japanese PASS, eight patients with severe haemophilia A developed factor VIII inhibitors (four each with low and high titres) following median 15 (range 8–93) days of exposure to Advate® (incidence 12.1% [95% CI 5.38, 22.5]).[abstract presentation].[32] Of these, four patients developed inhibitors after intracranial haemorrhage and intensive factor VIII treatment before 1 year of age, three patients had a family history of factor VIII inhibitors and two patients had two risk factors (intracranial haemorrhage and family history of inhibitors).[32]
6. Dosage and Administration
Intravenous Advate® is indicated for the control and prevention of bleeding episodes, for perioperative management and for routine prophylaxis in children (aged 0–16 years[13]) and adults with haemophilia A.[11,13] The dosage and duration of Advate® depend on the severity of factor VIII deficiency, the location and extent of bleeding and the patient's clinical condition.[11,13] The required dose of Advate® (IU) is determined by multiplying the desired increase in factor VIII (IU/dL or % of normal) by the patient's bodyweight (kg) multiplied by 0.5.[11,13] Advate® should be administered at a maximum infusion rate of 10 mL/min[11,13] over a period of ≤5 minutes.[13]
Table VII provides a guide for Advate® dosages in the treatment of bleeding episodes and perioperative management.[11,13] The dosage and duration of administration should be carefully controlled and factor VIII activity should not be allowed to fall below the required levels during the treatment period. The dosage and frequency of administration should be adapted based on clinical response, as individual patients may vary in their pharmacokinetic (e.g. t½ and in vivo recovery; section 3) and clinical responses. Appropriate determination of plasma factor VIII activity during the course of treatment is advisable, particularly during major surgical interventions.[11,13]
For routine prophylaxis, in the US, the recommended dosage of Advate® is 20–40 IU/kg every other day (three or four times weekly) or Advate® may be administered every third day at a dosage targeted to maintain trough factor VIII levels at ≥1%.[13] Dosage should be adjusted based on the patient's clinical response.[13] In the EU, the usual dosage of Advate® for long-term prophylaxis in patients with severe haemophilia A is 20–40 IU/kg every 2 or 3 days.[11] In patients aged <6 years, the recommended dosage of Advate® is 20–50 IU/kg three or four times weekly.[11]
Patients receiving Advate® should be monitored for the development of factor VIII inhibitors. Allergic-type hypersensitivity reactions, including anaphylaxis, may occur in patients being treated with Advate®.[11,13]
Local prescribing information should be consulted for comprehensive information on dosage and administration, contraindications, precautions and warnings.
7. Place of Advate® in the Management of Haemophilia A
As patients with haemophilia require lifelong treatment involving replacement of the missing clotting factor, the disease places a heavy burden on patients, caregivers and healthcare providers.[35] The burden of disease is greater in patients who develop inhibitors, as they are unresponsive to standard replacement therapy and may experience serious bleeding complications and severe arthropathy, resulting in increased morbidity and disability, greater impairment of HR-QOL and costs associated with treatment.[35,36]
A prospective, open-label study (PASS) showed that patients with haemophilia A had worse physical, but not mental, HR-QOL compared with age- and gender-adjusted US general population.[37] Compared with other chronic conditions, the physical burden of haemophilia A was equivalent to that of diabetes mellitus and rheumatoid arthritis and greater than that of chronic back pain.[37] The majority (≈80–90%) of bleeding episodes in patients with haemophilia occur in the musculoskeletal system (particularly the large synovial joints, e.g. knees and elbows), which may lead to synovial hypertrophy and cartilage damage and gradually destroy the joint.[38] Data from PASS showed that the presence of target joints was associated with worse HR-QOL in patients with haemophilia A, with the effect being more pronounced in patients aged >45 years.[37]
The goal of treatment in the management of haemophilia is to prevent bleeding.[39] For haemophilia A, factor VIII replacement therapy, administered prophylactically, on demand (episodic) or for perioperative management, is the mainstay of treatment.[40] Treatment for acute bleeds should be initiated as early as possible, preferably within 2 hours of the bleeding episode; prophylaxis can be started at an early age to prevent expected complications (primary prophylaxis) or administered for a short duration to prevent recurrence in patients who have repeated bleeding episodes (secondary prophylaxis).[39] Primary prophylaxis can be long-term treatment initiated after the first joint bleed and before the age of 2 years (type A) or initiated before the age of 2 years in the absence of clinically evident joint bleeds (type B).[40,41] Secondary prophylaxis can be long-term continuous treatment not fulfilling the criteria of primary prophylaxis (type A) or intermittent regular (i.e. short-term) treatment, generally started because of frequent bleeds (type B).[40,41]
Replacement therapy may be administered using recombinant or plasma-derived factor VIII concentrates. Treatment with cryoprecipitates is not recommended by the US National Haemophilia Foundation's Medical and Scientific Advisory Council (MASAC)[42] and the WFH supports the use of coagulation factor concentrates (recombinant or plasma-derived) over cryoprecipitates, fresh frozen plasma or cryo-poor plasma.[39] For patients with mild haemophilia A, other pharmacological options may be used, including desmopressin (a synthetic analogue of antidiuretic hormone that boosts plasma levels of factor VIII and VWF), tranexamic acid (an antifibrinolytic agent that competitively inhibits the inactivation of plasminogen to plasmin and promotes clot stability) or epsilon aminocaproic acid (a drug similar to tranexamic acid).[39] The MASAC guidelines recommend treatment with desmopressin whenever possible for patients with mild haemophilia A.[42]
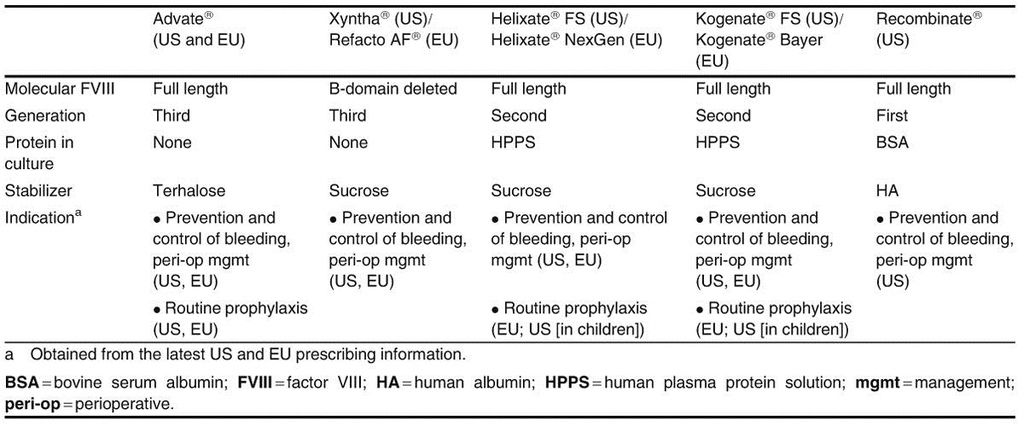
Several types of recombinant factor VIII concentrates are currently available (table VIII). Of these, Advate® is the only full-length, human, recombinant factor VIII product that is produced without animal- or human-derived proteins (table VIII). It is indicated for the prevention and control of bleeding episodes, perioperative management and routine prophylaxis in children and adults. In the US, Advate® is the only product currently approved for routine prophylaxis in adults (table VIII).
Advate® was bioequivalent to its predecessor, Recombinate®, in terms of its pharmacokinetic properties (section 3) and animal studies showed that both products were similar in terms of haemostatic efficacy.[16]
In two pivotal clinical trials, Advate® administered prophylactically or on demand for ≥50 exposure days was effective for the prevention and treatment of bleeding episodes in previously treated paediatric and adult patients with moderately severe or severe haemophilia A (sections 4.1.1 and 4.1.2). The haemostatic efficacy of Advate® was rated as ‘excellent’ or ‘good’ in ≥86% of patients, with the majority (>78%) of patients being managed with one infusion. Advate® was also shown to be effective for perioperative management in a similar patient population, with its haemostatic efficacy rated as ‘excellent’ or ‘good’ in ≥91% of procedures intraoperatively and postoperatively, and in 75% of bleeding episodes during home replacement therapy (section 4.2). These findings were supported by data from pooled analyses of clinical trials (section 4.3) and routine clinical practice studies, such as PASS (section 4.4). There are no direct head-to-head efficacy comparisons of Advate® with other factor VIII concentrates.
Several studies have shown the benefit of prophylactic treatment in paediatric patients, with evidence supporting early initiation of therapy to preserve joint integrity.[41] For example, in a 10-year study in children (aged 1–7 years) with severe haemophilia A (factor VIII <1%; n = 45), patients receiving prophylaxis had significantly (p < 0.02) fewer haemarthrosis than those receiving on-demand therapy (patients received Recombinate® initially and were then switched to Advate®).[43] Moreover, prophylaxis was significantly (p < 0.05) more effective when started at a younger age (≤3 years of age) than at an older age, as indicated by radiological signs of arthropathy.[43] Another study in children with moderately severe or severe haemophilia A (factor VIII ≤2%) showed that significantly (p = 0.006) more patients receiving prophylaxis had normal index-joint structure than those receiving on-demand therapy (65 children aged <30 months were enrolled and received Kogenate® or Kogenate® FS; assessments were conducted at the age of 6 years).[44]
Prophylaxis may also be beneficial in adults.[45,46] For example, in one study, patients (aged 30–45 years; n = 20) with severe haemophilia A (factor VIII <1%) who were switched from 6 months of on-demand therapy to 7 months of prophylaxis experienced significantly (p < 0.001) fewer haemarthrosis with prophylaxis than on-demand therapy with Kogenate FS.[47] Prophylactic treatment with Advate® was also more effective than on-demand therapy, as indicated by a significant reduction in the annual bleeding rate in previously treated patients aged >7 to <65 years with moderately severe or severe haemophilia A (section 4.1.3).
Guidelines by the United Kingdom Haemophilia Centre Doctors' Organization (UKHCDO)[48] and MASAC[49] recommend that patients with severe haemophilia A should receive prophylactic infusions of factor VIII to prevent haemarthrosis and other bleeding episodes. Treatment should be initiated at an early age in children, prior to the onset of significant bleeding[49] or by the second joint bleed or significant soft tissue bleed.[48] The MASAC guidelines recommend factor VIII dosages of 25–50 IU/kg three times per week or every other day for prophylaxis in patients with severe haemophilia A, and state that patients may continue to benefit from prophylaxis throughout their life.[49] According to the UKHCDO, prophylaxis may be initiated by administering factor VIII concentrate once weekly, but treatment should be rapidly escalated to more frequent administration, venous access permitting.[48] Ideally, factor VIII concentrate (25–50 IU/kg) should be administered every 48 hours, circumstances permitting; the minimum dosage of factor VIII that prevents breakthrough bleeds should be used.[48] The UKHCDO also recommends that adolescent and adult patients with severe haemophilia A should be encouraged to continue regular prophylaxis at least until they have reached physical maturity; the dosage and frequency of infusions should be adjusted based on bleeding phenotype and the patient's pharmacokinetic response.[48]
Data from clinical trials showed that the incidence of bleeding in patients receiving factor VIII replacement therapy was associated with the length of time an individual spent with factor VIII levels less than <1% (section 2), which in turn was influenced by the pharmacokinetic response of the individual. Of the pharmacokinetic parameters, the t½ and dose frequency of Advate® were found to affect trough plasma levels of factor VIII and the time spent per week with levels <1% to the greatest extent (section 3). Moreover, the pharmacokinetic response to Advate® varied, to some extent, with age (section 3).
As these factors contribute to considerable inter-individual variance in the pharmacokinetic response to Advate®, tailoring of Advate® dosages, based on an individual's response to treatment, may help to provide more cost-effective therapy and potentially increase access to factor VIII concentrates in regions with limited healthcare resources.[50] A comparative study showed that during routine prophylaxis in previously treated patients with moderately severe or severe haemophilia A, the haemostatic efficacy of Advate® in a pharmacokinetic-tailored regimen (administered every third day) did not differ significantly from that in a standard regimen (administered every second day).[section 4.1.3].[26] These results suggest that treatment with similar amounts of factor VIII, but fewer infusions, may be a viable treatment option, which could have implications in terms of adherence to therapy, particularly in children.[26] The need for frequent venous access is a problem in children and while central venous access devices aid home treatment and prophylaxis, complications (e.g. infections, occlusions) associated with their use are a major concern in the management of haemophilia.[41] Factors that improve adherence (e.g. modifying the prophylaxis schedule, decreased infusion volumes, easier administration techniques, factor VIII concentrates with longer t½) could potentially improve treatment outcomes.[51] Indeed, a pooled analysis showed that compliance to prophylaxis was associated with lower bleeding rates (section 4.3).
Advate® was generally well tolerated in clinical trials and postmarketing studies in patients with haemophilia A, with the most common treatment-emergent adverse events being pyrexia and headache (section 5). Serious adverse events observed during Advate® therapy included hypersensitivity reactions and the development of high-titre factor VIII inhibitors (section 5).
Development of factor VIII inhibitors is one of the major challenges of haemophilia A treatment. A systematic review found that up to 39% of patients could be at risk of inhibitor development (usually during the first 20 exposure days), with the incidence and prevalence of inhibitors being higher in patients with severe haemophilia A.[52] The incidence of factor VIII inhibitors in Advate® clinical (section 5.2) and postmarketing (section 5.3) studies ranged between 0% and 20%, with higher incidences observed in patients who were previously untreated with factor VIII concentrates. Factor VIII inhibitors are thought to develop as a result of an interplay between genetic factors (factor VIII genotype, family history of inhibitors, ethnicity, immuno-genotype) and non-genetic factors (presence of danger conditions, e.g. trauma, surgery) at the time of replacement therapy.[53] With regard to the non-genetic or modifiable factors, studies suggested that intensive treatment periods (e.g. surgical procedures) were associated with an increased risk of inhibitor development,[54] whereas early and regular prophylaxis may protect patients against the development of inhibitors.[55,56]
In patients with low-titre inhibitors (generally <5 BU/mL), bleeding can usually be resolved with normal or increased quantities of exogenous factor VIII.[8] However, patients with high-titre (≥5 BU/mL) inhibitors require treatment with bypassing agents (e.g. recombinant activated factor VII, activated prothrombin complexes) or immune tolerance induction (ITI) therapy to control bleeding.[8,57] The WFH guidelines state that ITI therapy may often eradicate inhibitors in patients with haemophilia A[39] and guidelines by the UKHCDO recommend ITI therapy for patients with severe congenital haemophilia A and confirmed factor VIII inhibitor.[58] However, the optimal dosage and frequency of dosage for ITI therapy and the role of different types of factor VIII concentrates in ITI are currently not known.[39,40,58] Large international trials[59-61] are currently ongoing, which are expected to resolve some of these issues. ITI using Advate® was shown to be successful in 73–77% of patients in a case-series,[62] a prospective study (PUP-ITI) and a retrospective chart review (PRE-PAIR).[63] It was also generally well tolerated when used for ITI, with no serious treatment-related adverse events and six nonserious adverse events being reported in PUP-ITI, PRE-PAIR and an ongoing prospective Advate® ITI registry (PAIR).[63]
In conclusion, prophylaxis or on-demand therapy with Advate® was effective for the prevention and treatment of bleeding episodes and for perioperative management in paediatric and adult patients with haemophilia A. Routine prophylaxis with Advate® was also effective for the prevention of bleeding episodes in patients with haemophilia A, with no significant difference between a standard and a pharmacokinetic-tailored regimen in terms of efficacy. Moreover, any routine prophylaxis with Advate® was more effective in preventing bleeding episodes than on-demand therapy with the concentrate. Advate® was generally well tolerated in clinical trials and postmarketing studies. Serious adverse events with Advate® therapy are development of high-titre factor VIII inhibitors (usually in previously untreated patients) and hypersensitivity reactions. There are no comparative trials of Advate® and other factor VIII concentrates. Nevertheless, current evidence indicates that Advate® is an effective option for the management of paediatric and adult patients with haemophilia A.
Disclosure
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from comments received were made by the author on the basis of scientific and editorial merit.
References1. Shapiro AD. Anti-hemophilic factor (recombinant), plasma/albumin-free method (octocog-alpha; ADVATE®) in the management of haemophilia A. Vasc Health Risk Manag 2007; 3 (5): 555-65
2. National Haemophilia Foundation. Haemophilia A (factor VIII deficiency).[online]. Available from URL: http://www.haemophilia.org [Accessed 2012 Feb 14]
3. White GC2nd, Rosendaal F, Aledort LM, et al. Definitions in haemophilia: recommendation of the scientific subcommittee on factor VIII and factor IX of the scientific and standardization committee of the International Society on Thrombosis and Haemostasis. Thromb Haemost 2001 Mar; 85 (3): 560
4. Schulman S. Treatment of haemophilia [online]. Available from URL: http://www.wfh.org/2/docs/Publications/Diagnosis_and_Treatment/TOH-41_Mild-Haemophilia.pdf [Accessed 2012 Mar 29]
5. Larsson SA, Nilsson IM, Blomback M. Current status of Swedish haemophiliacs. I. A demographic survey. Acta Med Scand 1982; 212 (4): 195-200
6. World Federation of Haemophilia. Report on the annual global survey 2010 [online]. Available from URL: http://www.wfh.org/2/docs/Publications/Statistics/2010_WFH_Global_Survey_Report.pdf [Accessed 2012 Feb 13]
7. Ananyeva N, Khrenov A, Darr F, et al. Treating haemophilia A with recombinant blood factors: a comparison. Expert Opin Pharmacother 2004 May; 5 (5): 1061-70
8. Wong T, Recht M. Current options and new developments in the treatment of haemophilia. Drugs 2011; 71 (3): 305-20
9. Powell JS. Recombinant factor VIII in the management of haemophilia A: current use and future promise. Ther Clin Risk Manag 2009 Apr; 5 (2): 391-402
10. Franchini M, Lippi G. Recombinant factor VIII concentrates. Semin Thromb Hemost 2010 Jul; 36 (5): 493-7
11. European Medicines Agency. Advate: summary of product characteistics [online]. Available from URL: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000520/WC500022467.pdf [Accessed 2012 Feb 8]
12. Klinge J, Ananyeva NM, Hauser CA, et al. Haemophilia A: from basic science to clinical practice. Semin Thromb Hemost 2002 Jun; 28 (3): 309-22
13. Baxter Healthcare Corp. Antihaemophilic factor (recombinant), plasma/albumin-free method (Advate): US prescribing information [online]. Available from URL: http://advate.com/pdf/advate_iri_pi.pdf [Accessed 2012 Feb 1]
14. Parti R, Schoppmann A, Lee H, et al. Stability of lyophilized and reconstituted plasma/albumin-free recombinant human factor VIII (ADVATE rAHF-PFM). Haemophilia 2005 Sep; 11 (5): 492-6
15. Fernandez M, Yu T, Bjornson E, et al. Stability of ADVATE, antihaemophilic factor (recombinant) plasma/albumin-free method, during simulated continuous infusion. Blood Coagul Fibrinolysis 2006 Apr; 17 (3): 165-71
16. European Medicines Agency. Scientific discussion [online]. Available from URL: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/human/000520/WC500022464.pdf [Accessed 2012 Feb 8]
17. Tarantino MD, Collins PW, Hay CR, et al. Clinical evaluation of an advanced category antihaemophilic factor prepared using a plasma/albumin-free method: pharmacokinetics, efficacy, and safety in previously treated patients with haemophilia A. Haemophilia 2004 Sep; 10 (5): 428-37
18. Blanchette VS, Shapiro AD, Liesner RJ, et al., on behalf of the rAHF-PFM Clinical Study Group. Plasma and albumin-free recombinant factor VIII: pharmacokinetics, efficacy and safety in previously treated paediatric patients. J Thromb Haemost 2008 Aug; 6 (8): 1319-26
19. Collins PW, Blanchette VS, Fischer K, et al. Break-through bleeding in relation to predicted factor VIII levels in patients receiving prophylactic treatment for severe haemophilia A. J Thromb Haemost 2009 Mar; 7 (3): 413-20
20. Shapiro A, Gruppo R, Pabinger I, et al. Integrated analysis of safety and efficacy of a plasma- and albumin-free recombinant factor VIII (rAHF-PFM) from six clinical studies in patients with haemophilia A. Expert Opin Biol Ther 2009 Mar; 9 (3): 273-83
21. Björkman S, Blanchette VS, Fischer K, et al. Comparative pharmacokinetics of plasma- and albumin-free recombinant factor VIII in children and adults: the influence of blood sampling schedule on observed age-related differences and implications for dose tailoring. J Thromb Haemost 2010 Apr; 8 (4): 730-6
22. Di Paola J, Smith MP, Klamroth R, et al. ReFacto® and Advate®: a single-dose, randomized, two-period crossover pharmacokinetics study in subjects with haemophilia A. Haemophilia 2007 Mar; 13 (2): 124-30
23. Collins PW, Bjorkman S, Fischer K, et al. Factor VIII requirement to maintain a target plasma level in the prophylactic treatment of severe haemophilia A: influences of variance in pharmacokinetics and treatment regimens. J Thromb Haemost 2010 Feb; 8 (2): 269-75
24. Bjorkman S, Oh M, Spotts G, et al. Population pharmacokinetics of recombinant factor VIII: the relationships of pharmacokinetics to age and body weight. Blood 2012 Jan 12; 119 (2): 612-8
25. Zhang L, Zhao Y, Sun J, et al. Clinical observation on safety and efficacy of a plasma- and albumin-free recombinant factor VIII for on-demand treatment of Chinese patients with haemophilia A. Haemophilia 2011 Mar; 17 (2): 191-5
26. Valentino LA, Mamonov V, Hellmann A, et al. A randomized comparison of two prophylaxis regimens and a paired comparison of on-demand and prophylaxis treatments in hemophilia A management. J Thromb Haemost 2012 Mar; 10 (3): 359-67
27. Oldenburg J, Goudemand J, Valentino L, et al. Postauthorization safety surveillance of ADVATE [antihaemophilic factor (recombinant), plasma/albumin-free method] demonstrates efficacy, safety and low-risk for immunogenicity in routine clinical practice. Haemophilia 2010 Nov; 16 (6): 866-77
28. Négrier C, Shapiro A, Berntorp E, et al. Surgical evaluation of a recombinant factor VIII prepared using a plasma/albumin-free method: efficacy and safety of Advate in previously treated patients. Thromb Haemost 2008 Aug; 100 (2): 217-23
29. Shapiro A, Abshire T, Hernadez F, et al. Efficacy and safety of advate rAHF-PFM for perioperative management of hemostasis in previously treated children with haemophilia A. XXVIIth International Congress of the World Federation of Haemophilia; 2006 May 21-25; Vancouver (BC)
30. Fischer K, Collins P, Bjorkman S, et al. Trends in bleeding patterns during prophylaxis for severe haemophilia: observations from a series of prospective clinical trials. Haemophilia 2011 May; 17 (3): 433-8
31. Fukutake K, Hanabusa H, Taki M, et al. The Post-Authorization Safety Surveillance (PASS) program confirmed actual clinical safety and efficacy of recombinant plasma/albumin-free method factor VIII in JPN [abstract no. P-TU-502]. J Thromb Haemost 2011; 9 Suppl. 2: 466
32. Taki M, Hanabusa H, Fukutake K, et al. Clinical experience of previously untreated patients with antihaemophilic factor (recombinant), plasma/albumin-free method from post-authorization safety surveillance in JPN [abstract no. P-TU-510]. J Thromb Haemost 2011; 9 Suppl. 2: 468
33. Den Uijl I, Mauser-Bunschoten EP, Roosendaal G, et al. Efficacy assessment of a new clotting factor concentrate in haemophilia A patients, including prophylactic treatment. Haemophilia 2009 Nov; 15 (6): 1215-8
34. Bacon CL, Singleton E, Brady B, et al. Low risk of inhibitor formation in haemophilia A patients following en masse switch in treatment to a third generation full length plasma and albumin-free recombinant factor VIII product (ADVATE®). Haemophilia 2011 May; 17 (3): 407-11
35. Escobar MA. Health economics in haemophilia: a review from the clinician's perspective. Haemophilia 2010 May; 16 Suppl. 3: 29-34
36. Scalone L, Mantovani LG, Mannucci PM, et al. Quality of life is associated to the orthopaedic status in haemophilic patients with inhibitors. Haemophilia 2006 Mar; 12 (2): 154-62
37. Klamroth R, Pollmann H, Hermans C, et al. The relative burden of haemophilia A and the impact of target joint development on health-related quality of life: results from the ADVATE post-authorization safety surveillance (PASS) study. Haemophilia 2011 May; 17 (3): 412-21
38. Rodriguez-Merchan EC, Jimenez-Yuste V, Aznar JA, et al. Joint protection in haemophilia. Haemophilia 2011 Sep; 17 Suppl. 2: 1-23
39. World Federation of Haemophilia. Guidelines for the management of haemophilia [online]. Available from URL: http://www.wfh.org/2/docs/Publications/Diagnosis_and_Treatment/Guidelines_Mng_Haemophilia.pdf [Accessed 2012 Feb 16]
40. Coppola A, Di Capua M, Di Minno MN, et al. Treatment of haemophilia: a review of current advances and ongoing issues. J Blood Med 2010; 1: 183-95
41. Coppola A, Tagliaferri A, Di Capua M, et al. Prophylaxis in children with haemophilia: evidence-based achievements, old and new challenges. Semin Thromb Hemost 2012 Feb; 38 (1): 79-94
42. US National Haemophilia Foundation. MASAC recommendations concerning products licensed for the treatment of haemophilia and other bleeding disorders (revised Nov 2011).[online]. Available from URL: http://www.haemophilia.org/NHFWeb/Resource/StaticPages/menu0/menu5/menu57/masac190.pdf [Accessed 2010 Feb 17]
43. Gringeri A, Lundin B, von Mackensen S, et al. A randomized clinical trial of prophylaxis in children with haemophilia A (the ESPRIT study). J Thromb Haemost 2011 Apr; 9 (4): 700-10
44. Manco-Johnson MJ, Abshire TC, Shapiro AD, et al. Prophylaxis versus episodic treatment to prevent joint disease in boys with severe haemophilia. N Engl J Med 2007 Aug 9; 357 (6): 535-44
45. Blanchette VS. Prophylaxis in the haemophilia population. Haemophilia 2010 Jul; 16 Suppl. 5: 181-8
46. Hay CR. Prophylaxis in adults with haemophilia. Haemophilia 2007 Sep; 13 Suppl. 2: 10-5
47. Collins P, Faradji A, Morfini M, et al. Efficacy and safety of secondary prophylactic vs. on-demand sucrose-formulated recombinant factor VIII treatment in adults with severe haemophilia A: results from a 13-month crossover study. J Thromb Haemost 2010 Jan; 8 (1): 83-9
48. Richards M, Williams M, Chalmers E, et al. A United Kingdom Haemophilia Centre Doctors' Organization guideline approved by the British Committee for Standards in Haematology: guideline on the use of prophylactic factor VIII concentrate in children and adults with severe haemophilia A. Br J Haematol 2010 May; 149 (4): 498-507
49. National Haemophilia Foundation. MASAC recommendation concerning prophylaxis (regular administration of clotting factor concentrate to prevent bleeding).[document #179; online]. Available from URL: http://www.haemophilia.org [Accessed 2012 Feb 21]
50. Collins PW, Fischer K, Morfini M, et al., on behalf of the International Prophylaxis Study Group Pharmacokinetics Expert Working Group. Implications of coagulation factor VIII and IX pharmacokinetics in the prophylactic treatment of haemophilia. Haemophilia 2011 Jan; 17 (1): 2-10
51. Thornburg CD. Prophylactic factor infusions for patients with haemophilia: challenges with treatment adherence. J Coagul Dis 2009; 2 (1)
52. Wight J, Paisley S. The epidemiology of inhibitors in haemophilia A: a systematic review. Haemophilia 2003 Jul; 9 (4): 418-35
53. Coppola A, Santoro C, Tagliaferri A, et al. Understanding inhibitor development in haemophilia A: towards clinical prediction and prevention strategies. Haemophilia 2010 Jan; 16 Suppl. 1: 13-9
54. Gouw SC, van den Berg HM, LE Cessie S, et al. Treatment characteristics and the risk of inhibitor development: a multicenter cohort study among previously untreated patients with severe haemophilia A. J Thromb Haemost 2007 Jul; 5 (7): 1383-90
55. Gouw SC, van der Bom JG, Marijke van den Berg H. Treatment-related risk factors of inhibitor development in previously untreated patients with haemophilia A: the CANAL cohort study. Blood 2007 Jun 1; 109 (11): 4648-54
56. Santagostino E, Mancuso ME, Rocino A, et al. Environmental risk factors for inhibitor development in children with haemophilia A: a case-control study. Br J Haematol 2005 Aug; 130 (3): 422-7
57. Franchini M, Lippi G. Immune tolerance induction for patients with severe haemophilia A: a critical literature review. J Thromb Thrombolysis 2011 Nov; 32 (4): 439-47
58. Hay CR, Brown S, Collins PW, et al. The diagnosis and management of factor VIII and IX inhibitors: a guideline from the United Kingdom Haemophilia Centre Doctors Organisation. Br J Haematol 2006 Jun; 133 (6): 591-605
59. City of Hope Medical Center. Study of first time immunotolerance induction in severe haemophilia A patients with inhibitor at high risk of failure: comparison with FVIII concentrates with or without von Willebrand Factor -RES.I.S.T. Naive (RESIST NAIVE).[ClinicalTrials.gov identifier NCT01051544]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2012 Feb 9]
60. City of Hope Medical Center. Rescue immunotolerance study in induction of immune tolerance (ITI)-experienced patients (RES.I.S.T. Experienced) (RESIST EXP).[ClinicalTrials.gov identifier NCT01051076]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://clinicaltrials.gov [Accessed 2012 Feb 9]
61. Dimichele DM, Hay CR. The international immune tolerance study: a multicenter prospective randomized trial in progress [letter]. J Thromb Haemost 2006 Oct; 4 (10): 2271-3
62. Valentino LA, Recht M, Dipaola J, et al. Experience with a third generation recombinant factor VIII concentrate (Advate) for immune tolerance induction in patients with haemophilia A. Haemophilia 2009; 15 (3): 718-26
63. Spotts G, Abbuehl BE, Luu HD, et al. Safety and efficacy profile of rAHF-PFM for immune tolerance induction as assessed in 3 clinical trials (PUP ITI, PRE-PAIR, and PAIR).[abstract no. 3670]. Blood (ASH Annual Meeting Abstracts) 2010; 116 (21)
|






 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 