By Yael Waknine
Medscape Medical News
February 3, 2010 — The US Food and Drug Administration (FDA) has approved collagenase clostridium histolyticum injection (Xiaflex, Auxilium Pharmaceuticals, Inc) as the first treatment for a progressive hand disease known as Dupuytren's contracture. The company expects to launch the first-in-class, orphan-designated biologic in late March 2010.
Dupuytren's is a debilitating condition caused by an abnormal buildup of collagen in the connective tissue of the palms and hands, forming thick rope-like cords that cause the fingers to curl. As the contractures increase, it may be difficult to use affected fingers to grasp objects. The condition affects an estimated 240,000 individuals across the United States and Europe and is most common among whites and men older than 50 years.
"Before the FDA approved Xiaflex, the only effective treatment for this hand disorder was surgery, which sometimes meant a long recovery and the need for physical therapy for patients," said Bob Rappaport, MD, director, Division of Anesthesiology, Analgesia, and Rheumatology of the FDA's Center for Drug Evaluation and Research, in an agency news release. "Since there are no other non-surgical alternatives for Dupuytren's contracture, Xiaflex will be an important advance in the management of this disabling condition."
The product, which contains 2 microbial collagenases derived fromClostridium histolyticum bacteria, is administered intralesionally into affected areas of the hand to soften and ultimately lyse the cords, restoring flexibility.
Its approval was based in part on data from a 306-patient study, showing that 64% of those treated with the biologic achieved finger mobility compared with 7% of those receiving placebo. In a smaller study (n = 66), the success rate was 44% (vs 5% for placebo).
Adverse events most commonly reported in the studies (incidence ? 25%) included peripheral edema, contusion, injection-site reaction, injection-site hemorrhage, and pain in the injected extremity. Although no serious allergic reactions were observed, the presence of foreign proteins poses a risk for immune response.
"With the safety and effectiveness of Xiaflex demonstrated across multiple clinical trials, physicians can now use Xiaflex to treat any symptomatic cords in patients with Dupuytren's contracture," said Larry Hurst, MD, study investigator and professor and chair, Department of Orthopedics at State University of New York Stony Brook in a company news release. "I believe that Xiaflex, as a new nonsurgical treatment, could potentially become the standard of care for Dupuytren's contracture."
Because of the potential for tendon ruptures, the FDA is requiring implementation of a risk evaluation and mitigation strategy program that will inform clinicians about how to properly perform injections and finger extensions. Access to the collagenase product will be limited to clinicians who have completed the training program, which can be obtained as a video or written manual by calling 1-877-XIAFLEX.
February 3, 2010 — 美國食品藥物管理局(FDA)核准溶組織芽胞梭菌膠原酵素注射劑(商品名Xiaflex,Auxilium Pharmaceuticals藥廠)用於一種漸進性手部疾病、Dupuytren氏孿縮症(Dupuytren's contracture)的治療,該公司預計在2010年3月底發售此類別中的第一種罕見疾病生物製劑。
Dupuytren氏孿縮症是一種手掌和手連結組織中之膠原異常建立而引起的令人衰弱的狀況,形成厚繩狀的韌帶而使得手指捲曲。隨著攣縮程度增加,而難以使用這些發病的手指抓取物品,在歐美,約有240,000人受到此狀況影響,其中以白人與50歲以上男性較常見。
FDA疾病評估與研究中心麻醉、止痛和風濕製劑小組主任Bob Rappaport醫師在該局的新聞稿中表示,在FDA核准Xiaflex之前,這種手部疾病的唯一有效治療方式就是手術,有時候需要很長的恢復期、病患也需要物理治療。因為Dupuytren氏孿縮症沒有其他非手術的替代治療方式,Xiaflex將是此一失能狀況之處置的重要進步。
該產品包括了衍生自溶組織芽胞梭菌的兩個微生物膠原酵素,注射入手之患部病灶內,軟化與分解這些韌帶,重建彈性。
這項核准是根據一個有306名病患之研究的部份資料,顯示那些以這個生物製劑治療的病患有64%達到手指可動,使用安慰劑者只有7%。在比較小型的研究中(n = 66人),成功率為44% (安慰劑組為5%)。
這些研究中,最常報告的副作用(發生率≧25%)包括週邊水腫、挫傷、注射部位反應、注射部位出血、注射四肢疼痛。雖然沒有觀察到嚴重的過敏反應,出現外來蛋白質仍有可能引起免疫反應的風險。
紐約州立大學石溪校區骨科部主任Larry Hurst醫師在藥廠的新聞稿中表示,經過多個臨床試驗顯示Xiaflex的安全性和效果,醫師們現在可以使用Xiaflex來治療Dupuytren氏孿縮症病患之任何有症狀的韌帶。我相信,Xiaflex這種新的非手術治療方式,可以變成Dupuytren氏孿縮症的標準照護。
因為可能會造成肌腱破裂,FDA要求進行風險評估以及緩解策略計畫,告知醫師們有關如何適當進行注射以及手指伸展。僅限有受過完整訓練計畫的醫師們使用膠原酵素製劑,請來電1-877-XIAFLEX索取影片或書面手冊。
出處:WebMD醫學新聞
除了藥物照片以外,另外附上一些廠商提供的治療圖片

下面這張是注射前
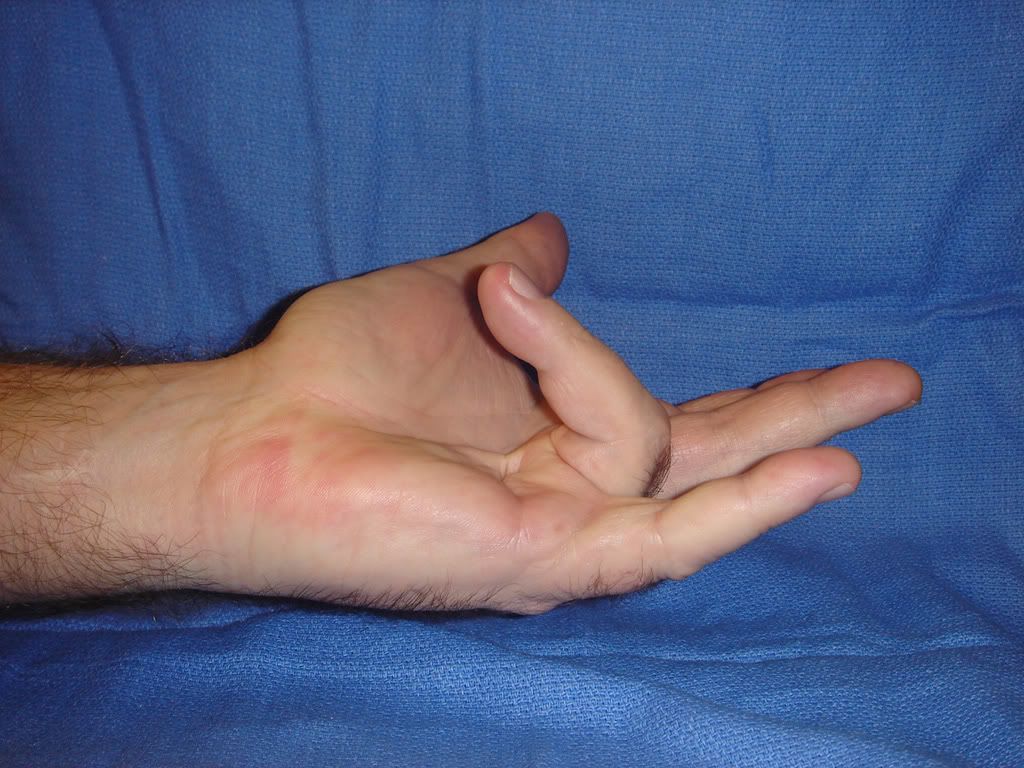
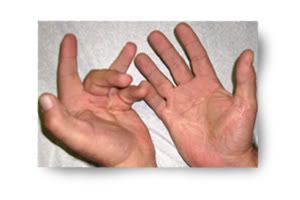
相信你可以從圖片中看到他攣縮的很嚴重,接著是給藥之後的照片
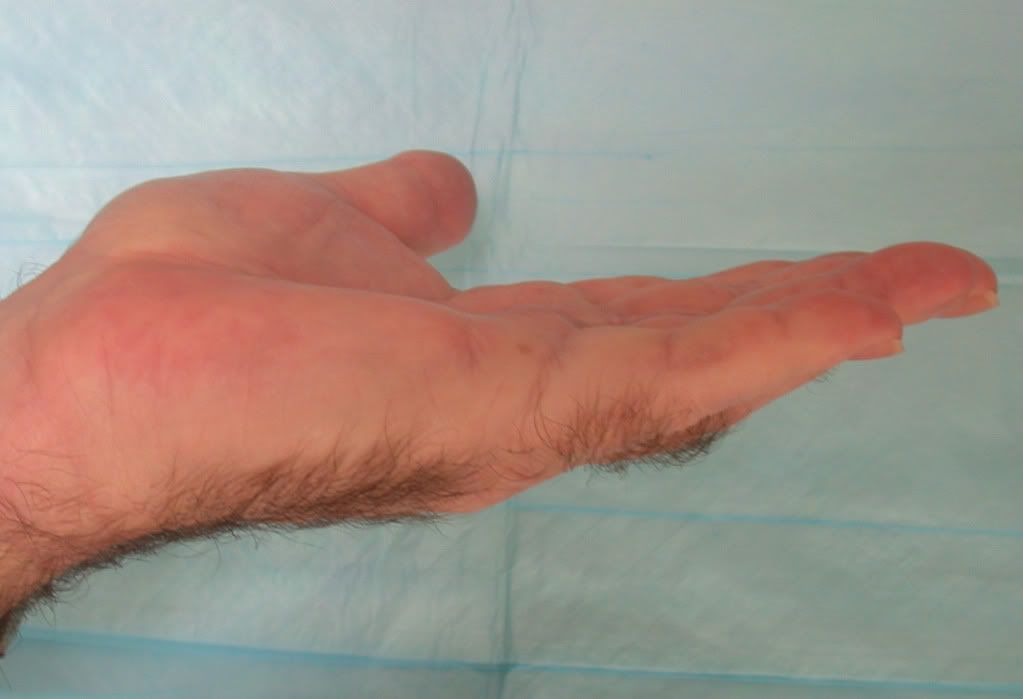
Before receiving XIAFLEX, tell your healthcare provider if you have had an allergic reaction to a previous XIAFLEX injection, or have a bleeding problem or any other medical conditions. Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Be sure to tell them if you use blood thinners such as aspirin, clopidogrel (Plavix®), prasugrel hydrochloride (Effient®), or warfarin sodium (Coumadin®).





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 