Roerig, James L.; Steffen, Kristine J.; Mitchell, James E.; Zunker, Christie
Abstract
Laxatives have been used for health purposes for over 2000 years, and for much of that time abuse or misuse of laxatives has occurred. Individuals who abuse laxatives can generally be categorized as falling into one of four groups. By far the largest group is made up of individuals suffering from an eating disorder such as anorexia or bulimia nervosa. The prevalence of laxative abuse has been reported to range from approximately 10% to 60% of individuals in this group. The second group consists of individuals who are generally middle aged or older who begin using laxatives when constipated but continue to overuse them. This pattern may be promulgated on certain beliefs that daily bowel movements are necessary for good health. The third group includes individuals engaged in certain types of athletic training, including sports with set weight limits. The fourth group contains surreptitious laxative abusers who use the drugs to cause factitious diarrhoea and may have a factitious disorder.
Normal bowel function consists of the absorption of nutrients, electrolytes and water from the gut. Most nutrients are absorbed in the small intestine, while the large bowel absorbs primarily water. There are several types of laxatives available, including stimulant agents, saline and osmotic products, bulking agents and surfactants. The most frequently abused group of laxatives are of the stimulant class. This may be related to the quick action of stimulants, particularly in individuals with eating disorders as they may erroneously believe that they can avoid the absorption of calories via the resulting diarrhoea.
Medical problems associated with laxative abuse include electrolyte and acid/base changes that can involve the renal and cardiovascular systems and may become life threatening. The renin-aldosterone system becomes activated due to the loss of fluid, which leads to oedema and acute weight gain when the laxative is discontinued. This can result in reinforcing further laxative abuse when a patient feels bloated and has gained weight.
Treatment begins with a high level of suspicion, particularly when a patient presents with alternating diarrhoea and constipation as well as other gastrointestinal complaints. Checking serum electrolytes and the acid/base status can identify individuals who may need medical stabilization and confirm the severity of the abuse. The first step in treating laxative misuse once it is identified is to determine what may be promoting the behaviour, such as an eating disorder or use based on misinformation regarding what constitutes a healthy bowel habit. The first intervention would be to stop the stimulant laxatives and replace them with fibre/osmotic supplements utilized to establish normal bowel movements. Education and further treatment may be required to maintain a healthy bowel programme. In the case of an eating disorder, referral for psychiatric treatment is essential to lessen the reliance on laxatives as a method to alter weight and shape.
1. Introduction
For more than 2000 years, purgative use has been an important element in medical therapy. However, laxatives are increasingly used as a method of weight control.[1] Individuals who abuse laxatives can generally be categorized as falling into one of four groups. The first includes those with eating disorders, a group of patients who are well known to have a high prevalence of laxative abuse.[2] In light of the high prevalence of laxative abuse in this group, reported to range from approximately 10% to 60%, we review it in detail. The second group consists of individuals who are generally middle aged or older who begin using laxatives when constipated, but who continue to overuse them to the point that their bowel becomes relatively refractory to the laxatives.[3] Considering that normal stool frequency on a Western diet is at least three times a week, excessive use may begin with the belief that daily bowel evacuation is necessary for good health.[4] It can also be the result of increased constipation associated with a variety of causes including poor diet, decreased mobility or concomitant drug therapy. As these patients become dependent on laxatives, it becomes increasingly hard to intervene. Both of these use patterns are associated with medical complications that may have a significant impact on the health of the patient, and highlight the need for early detection and intervention. The third group includes individuals engaged in certain types of athletic training, including sports with set weight limits. The subgroup here that has perhaps been best characterized includes wrestlers, who take laxatives as a way to drop weight.[5] The fourth group contains surreptitious laxative abusers who use the drugs to cause factitious diarrhoea and may have a factitious disorder.[6,7]
Eating disorder patients constitute the largest group of individuals who abuse laxatives and are certainly the best characterized. These patients may initially take laxatives in response to constipation. This is particularly true of patients with anorexia nervosa who, because of low food intake and dehydration, frequently have problems with constipation. However, most eating disorder patients take laxatives to induce diarrhoea in order to feel thinner, and in an attempt to get rid of unwanted calories and lose weight. Often, laxatives are misused following eating binges, when the individual mistakenly believes that the laxatives will work to rush food and calories through the gut before they can be absorbed and, thus, will prevent calorie absorption and weight gain.[8] Obviously, these individuals are very concerned about weight and shape, and see laxative abuse as one method of controlling their bodyweight. This abuse sets up a vicious cycle in which they become dehydrated because of fluid loss, retain fluid because of the renin-angiotensen response to dehydration, gain fluid weight and then need to use the laxatives again to dehydrate themselves. Also, because their bowels become relatively refractory to laxatives, they need to escalate the dose to get the same effect, and eventually develop patterns of using large amounts of laxatives.[9] The goal of this review is to acquaint the practitioner with the epidemiology, presentation and management of laxative abuse.
2. Gastrointestinal Functioning
The main function of the gastrointestinal (GI) tract is the extraction of fluid and nutrients from the contents present in the lumen. Nutrients are absorbed mainly by the small intestine and the right colon absorbs mainly water and electrolytes. As faecal matter moves into the left colon, it becomes more formed.[10] Control of the GI tract involves intrinsic and extrinsic innervation. The intrinsic innervation involves the enteric nervous system,[11-13] and comprises myenteric, submucosal and mucosal neuronal layers. Normal functioning of these layers involves interneurons and utilizes neurotransmitter amines and/or peptides, including acetylcholine, vasoactive intestinal peptide, opioids, noradrenaline (norepinephrine), serotonin, adenosine triphosphate and nitric oxide. The myenteric plexus regulates smooth-muscle function. The submucosal plexus regulates secretion, fluid transport and vascular flow.[14,15] The extrinsic innervation of the GI tract involves the parasympathetic autonomic nervous system which modulates motor and secretory functions. The excitatory neurotransmitters controlling motor function are acetylcholine and the tachykinins, such as substance P.
Ultimately, fluid content is important in the determination of stool consistency and volume, with water accounting for 70–85% of total stool weight. A balance exists between the ingestion and secretion of water and electrolytes into the GI tract and the absorption over its length. Eight to ten litres of fluid enter the small intestine daily. Due to osmotic gradients, absorption of water in the small intestine reduces the fluid presented to the large bowel to only 1–5 L. The colon extracts most of the remaining fluid, leaving about 100 mL of faecal water daily. This process can be altered by neurohumoral mechanisms, pathogens and drugs. The extent of water absorption is also influenced by the GI transit time. Colon motility is made up of two types of contractions: nonpropulsive contractions provide a mixing function and propulsive contractions move the contents of the bowel toward the rectum. Slowed GI motility results in a slower GI transit time, allowing more water to be absorbed, which can lead to constipation. Rapid transit time results in less water absorption, with the potential for resulting diarrhoea.[14,15]
Various drugs can affect this system. The use of anticholinergic drugs such as benztropine or diphenhydramine results in a slowing of the GI tract with the potential for constipation. On the other hand, a variety of drugs have a prokinetic effect on the bowel. These include cholinergic agents (bethanechol), acetylcholinesterase inhibitors (neostigmine or donepezil), dopamine-receptor antagonists (metoclopramide), serotonin enhancing drugs such as selective serotonin reuptake inhibitors, and motilin receptor agonists (erythromycin and other macrolide antibiotics).[16-18]
3. Epidemiology
3.1 Eating Disorders
Studies investigating the prevalence of laxative abuse are complicated. Virtually all investigations rely on self report and many have used different criteria to define laxative abuse. Overall, the lifetime occurrence of laxative abuse has been reported to be 4.18% in the general population.[19] However, the authors reported that the rates are substantially higher in people who suffer from an eating disorder. The lifetime occurrence among individuals with bulimia nervosa was 14.94%, greater than a 3-fold increase over the prevalence in the general population. Other studies have reported that laxative abuse among patients with bulimia nervosa range from 18% to 75%.[20-22] In a consecutive series of 100 patients with bulimia nervosa, Mitchell et al.[23] reported finding that 36% had abused laxatives for weight control purposes during the month prior to evaluation. More recently, Steffen et al.[24] reported that laxatives had been used at some point to control weight or “get rid of food” by 67% of 39 eating disorder patients surveyed. Of these, 31% reported abuse of laxatives during the month prior to evaluation. Phelps et al.[1] studied the prevalence of laxative abuse over time in adolescent females. They surveyed students on three occasions (1984, 1989 and 1992). Prevalence of laxative use for weight control remained relatively stable over the time period, ranging from 3.2% to 5.5% of those in high school and 0–1.8% of those in middle school (ages 13–15 years). Recently, Steffen and colleagues[25] explored the use of herbal laxatives and found that of 100 participants with eating disorder symptoms, 26% reported having used a herbal laxative at some point in time.
A difference in the prevalence of laxative abuse between eating disorder diagnoses may exist. A study compared subjects with bulimia nervosa – purging subtype (BN-P) and eating disorders not otherwise specified – purging only (EDNOS-P) with controls.[26] Laxative abuse was utilized significantly more frequently in the EDNOS-P group than by BN-P subjects (62% vs 27%; p < 0.04) as their purging method, while vomiting was more frequently used in the BN-P group than by EDNOS-P subjects (86% vs 38%; p < 0.001). A study in adolescents diagnosed with anorexia nervosa examined laxative use via self-report and biochemical laboratory evaluation, which included serum electrolytes, calcium, magnesium and phosphate levels, and urinary laxative screening for the stimulant laxatives bisacodyl, phenolphthalein and rhein.[27] Caution should be utilized when monitoring laxative use via laboratory assessment; difficulties with assays for senna and bisacodyl have recently been reported.[9] The frequency of laxative use from self-report alone was 12% and when combined with urine screening was 19%. With prospective follow-up, the frequency of laxative use increased to 32%. By all accounts, laxative abuse is not uncommon in patients with eating disorders.
3.2 Habitual Laxative Abuse
Unfortunately, the prevalence of chronic laxative use is difficult to quantify in light of the condition often being overlooked or the diagnosis being made only after extensive investigations have proven negative.[28,29] Studies suggest that the prevalence of constipation is as high as 50% in the elderly, which increases to 74% of nursing home residents using daily laxatives.[30-32] However, this probably under-represents the true impact of constipation and subsequent medical use, as many people either do not seek medical attention or use over-the-counter remedies (either alone or in combination with those prescribed by their physician).
The elderly are a group of individuals with unique health challenges and different medical needs than their younger counterparts. The high frequency of laxative use in the elderly may be caused, in part, by underestimation of stool frequency.[30] This can lead them to plan their schedules around their bowel movements. Unfortunately, laxative treatments in these patients often precipitate loose stools and incontinence. Subsequently, they may present to their healthcare provider with diarrhoea of unknown origin. Habitually used laxatives constitute an important cause of chronic diarrhoea in these patients.[33] Extensive medical investigations and an increase in the lengths of hospital stays are associated with diarrhoea of unknown origin. However, there are many people who are well served by chronic laxative use, such as patients with chronic constipation due to slow colonic transit or pelvic floor dysfunction.
4. Presentation/Diagnosis
The most important factor in making the diagnosis of laxative abuse is a high index of suspicion on the part of the clinician. If possible, objective evidence for laxative abuse should be obtained. However, laxative use is often carried out in secret and patients may be less than forthcoming regarding self report. The only sign that has been found to be suggestive of chronic laxative use is melanosis coli, a non-pathological, reversible, pigmentation of the colon associated with anthraquinone use.[34] A pattern of diarrhoea alternating with constipation, as well as other GI symptoms, may be reported. A variety of laboratory tests are helpful in determining the diagnosis. Serum hypokalaemia in eating disorder patients has been suggested to be definitive evidence of purging, including by laxative use.[35] In the presence of culture-negative diarrhoea, faecal electrolytes can aid in diagnosis.[33] This analysis includes quantification of the osmolality, the electrolyte composition and the pH level of stool water, with the calculation of the osmotic gap.[36-38] Diarrhoea caused by stimulant laxatives (such as bisacodyl), is associated with an osmotic gap that is small; in patients with elevated magnesium levels, the gap is much greater. Faecal magnesium concentration testing can aid in determining the presence of abuse with a magnesium-containing laxative. Fine and colleagues[39] reported that the upper limits of faecal output of soluble magnesium and faecal magnesium concentration in healthy volunteers were 14.6 mmol/day and 45.2 mmol/L, respectively. They defined a concentration of magnesium above 50 mmol as diarrhoeogenic. For pure magnesium-induced diarrhoea (without additional laxatives such as phenolphthalein), stool magnesium concentration in excess of 100 mmol indicate the use of a magnesium-containing laxative such as milk of magnesia. Laboratory tests for the presence of laxatives themselves are found in table I.
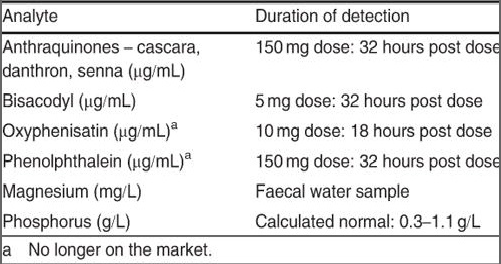
Many patients report that the feeling of having emptied themselves is associated not only with gratification resulting from the apparent weight loss but also with a sense of purification.[41] Eating disorder patients also believe that laxatives can reduce nutrient absorption. However, most nutrient absorption occurs in the small intestine.[42] Nutrient absorption has been reported to be reduced by only 12% by laxative use.[43] The patient's weight may be reduced slightly by the expulsion of water leading to a temporary mood-elevating effect further reinforcing the laxative use.[8]
Several publications have suggested that laxative abuse may characterize a more ill subgroup of patients with eating disorders, and a number of investigations support this observation in patients diagnosed with anorexia nervosa. A group of anorexia nervosa patients who abuse laxatives have been reported to have higher scores on histrionic personality assessments.[44] Kovacs and Palmer[41] assessed 117 patients diagnosed with anorexia nervosa. They found that 22 (18.8%) used laxatives for weight control. Subjects abusing laxatives were rated higher on the Ineffectiveness, Body Dissatisfaction and Drive for Thinness subscales of the Eating Disorder Inventory, and on the depression, somatization and global scores on the Symptom Checklist-90-Revised. They also scored higher on the Rosenberg Self-Esteem Scale (RSES),[45] suggesting lower self-esteem. In this series of adults, logistic regression revealed that in subjects with anorexia nervosa, body dissatisfaction (p < 0.002) and RSES scores (p < 0.033) were significant predictors of laxative abuse. Furthermore, the association of lower ‘self-liking’ in subjects suffering from anorexia nervosa and laxative abuse and self-induced vomiting has recently been reported.[46] A multiple regression was then performed to determine the relative contributions of each of these purging methods. Laxative abuse was determined to be the sole contributor to lower self-liking. These results suggest the possibility that laxative abuse is distinct from other types of purging by its association with self-liking rather than self-competence. This is in line with data indicating a significant association between laxative abuse and lower self-esteem.[41] The direction of this association has yet to be elucidated.
In patients diagnosed with bulimia nervosa, there is ample data associating more severe symptomology with laxative use. Bryant-Waugh and colleagues[47] examined laxative use in 201 consecutive patients in an outpatient eating disorder population. Those who misused laxatives were compared with those who did not. Fifty-three (26.4%) patients had misused laxatives in the month before assessment. Those who misused laxatives scored higher on measures of anorectic behaviours and cognitions, restraint, and weight and shape concerns. A history of use was associated with anorectic behaviours, depression and an increase in clinical severity, regardless of diagnosis. In a study of 23 bulimia nervosa patients who purged with laxatives and 17 who purged by vomiting, women who abused laxatives had higher levels of state anxiety than non-laxative-abusing women after laxative use was discontinued.[48] This study also found that the patients who abused laxatives were more likely to be treated with an antianxiety medication. Fifty-nine percent of a sample of 280 bulimia nervosa patients were found to abuse laxatives.[44] In this sample, patients with a laxative abuse history were found to demonstrate greater perfectionism and avoidant personality features. Subsequently, 40 ‘multi-impulsive’ bulimia nervosa patients were compared with 177 non-impulsive subjects.[49] A significantly greater likelihood of laxative abuse was found in the impulsive group. Another study evaluated the association of impulsivity and compulsivity in 125 patients with bulimia nervosa.[50] Laxative abuse was demonstrated to be associated with the impulsive dimension. Thus, impulsivity appears to be a personality characteristic that is frequently found in patients who abuse laxatives.
In a large community sample of young adult women (n = 5255), the 39 individuals who misused laxatives were found to be older, perceived themselves to be in poorer physical health and were less likely to have sought treatment specifically for a problem with eating than those who engaged in self-induced vomiting.[51] However, they found little evidence that young adult women who engage in self-induced vomiting differ from those who misuse laxatives with respect to levels of eating disorder psychopathology, health-related quality of life and general psychological distress.
In contrast, recent findings suggest that measures of greater illness were associated with the number of purging methods used and not associated with the type of purging method.[52] In this study, the authors reported similar levels of pathology between subjects who used a single method of purging (vomiting or laxative abuse) and greater eating pathology in those who used both methods. Behavioural co-morbidity found in laxative-abusing individuals may include affective disorders,[8,53,54] substance abuse,[22,55-57] self-destructive behaviours,[54] collateral purge methods[22] and general life impairments.[58]
A group of 43 adolescents diagnosed with anorexia nervosa was investigated in terms of eating symptomology.[27] The results showed that laxative use was associated with a longer duration of disease and with higher scores on the Eating Concern subscale of the Eating Disorders Examination (EDE). Laxative abuse has also been shown to predict lower quality of life in eating disordered samples,[59] and to be associated with a number of other variables suggestive of greater severity, including increased rates of self-harm and suicidal behaviours and increased rates of borderline personality disorder (BPD).[2,60] Analysis of symptoms revealed that specific features of BPD, including suicidality and self-harm, and feelings of emptiness and anger, were most strongly associated with laxative abuse.[2]
While urinary screening for laxatives may increase the detection of laxative use, so can monitoring for medical complications such as hypokalaemia. Monitoring serum electrolytes as well as faecal electrolytes, such as magnesium, may be more cost effective.[61] Turner and colleagues[27] reported performing 144 screens for laxatives, which yielded only 26 positive results.
In summary, the abuse of laxatives appears to be a marker for high rates of co-morbidity and other problematic behaviours among individuals with eating disorders. These patients will often not wish to reveal their laxative abuse and it falls on the clinician to investigate the possibility of this condition. Serum and faecal electrolytes represent an effective screening tool in patients who present with diarrhoea and additional GI complaints. Urine analysis for individual laxatives may be best reserved for more difficult cases. As indicated earlier, caution should be utilized when monitoring laxative use via laboratory assessment in light of recent difficulties with assays for senna and bisacodyl.[9]
5. Types of Laxatives
Table II lists commonly used laxatives, their onset of effect and daily dose. Laxatives generally act in one of the following ways: (i) enhancing retention of fluid by hydrophilic or osmotic mechanisms; (ii) decreasing net absorption of fluid by effects on small and large bowel fluid and electrolyte transport; or (iii) altering motility by either inhibiting segmenting (nonpropulsive) contractions or stimulating propulsive contractions.[64] All of the agents, with the exception of docusate calcium, reduce the transit time through the small bowel. Most of the agents enhance propulsive contractions in the large bowel and variably increase the amount of stool water. Of the laxative classes, stimulant types appear to be the most commonly abused agents.[42,65] This preference for stimulant agents may be related to the rapid, high-volume faecal discharge associated with these products. Of the 248 products surveyed by Steffen et al.,[24] 89 (36%) contained a nonprescription stimulant laxative (not including aloe-containing compounds). In their sample of treatment-seeking patients with bulimia nervosa, ex-lax® was the most commonly used laxative. This agent currently contains sennosides.

5.1 Stimulant Laxatives
There are two pharmacological classes of stimulant laxatives: diphenylmethane derivatives (bisacodyl) and anthraquinones (senna and cascara). Some authors also include castor oil in the stimulant class.
5.1.1 Diphenylmethane Derivatives
Previously, many of the over-the-counter laxative preparations utilized phenolphthalein as the active ingredient; however, concerns over carcinogenicity led the US FDA to ban it from laxative preparations in 1997. Ironically, two subsequent case-controlled studies could not identify an association of phenolphthalein and ovarian cancer.[66,67] These discrepant findings may be explained by the dosage (3–1000 times the human dose) used in the animal studies upon which the FDA based its ruling.[68,69] Currently, the only diphenylmethane agent in use is bisacodyl. This agent stimulates peristalsis by directly irritating the smooth muscle of the intestine. It also alters water and electrolyte secretion, producing net intestinal fluid accumulation. Bisacodyl is well tolerated; adverse effects include mild abdominal cramps, nausea, vomiting and rectal burning. The tablets are manufactured with a pH-sensitive coating so the agent will not dissolve in the stomach or upper GI tract. If taken with dairy products or antacids, the coating will dissolve while the dose is in the stomach and upper GI tract; any action of bisacodyl in these areas will produce cramping.
5.1.2 Anthraquinone Derivatives
Anthraquinones (senna and cascara) are metabolized by gut bacteria to active agents. A distinguishing effect of these compounds is a colouring of the bowel referred to as pseudomelanosis coli or melanosis coli. Its presence is associated with 9–12 months of anthraquinone use and can be used to identify abuse of these agents.[70] A controversial link between pseudomelanosis coli and colorectal cancer has been reported.[71] In fact, the anthraquinone laxative danthron was withdrawn from the market because of tumour formation in laboratory animals. Both agents are well tolerated. Senna can cause abdominal cramps, diarrhoea, nausea and vomiting. Cascara has similar adverse effects and can discolour the urine (reddish, pink or brown).
5.1.3 Castor Oil
Castor oil is derived from the bean of the castor plant, Ricinus communis. It contains two noxious ingredients: an extremely toxic protein, ricin, and an oil composed chiefly of the triglyceride of ricinoleic acid. These agents can produce a strong laxative effect with accompanying abdominal pain.
5.1.4 Colorectal Cancer
Concerns have been voiced regarding the association of long-term use of stimulant laxatives and the development of colorectal cancer. Long-term administration of bisacodyl has been reported to be toxic to the GI tract. Disturbed cytoplasmic and nuclear structure have been demonstrated in rat small-intestinal enterocytes.[72] A meta-analysis did find an association between colorectal cancer and constipation and cathartic use.[73] However, it appears that these data were confounded by the underlying dietary habits of the subjects. A case-controlled study involving middle-aged adults[74] found an association between constipation and laxative use and an increased risk of colorectal cancer. However, the association with laxatives disappeared when the data were adjusted for constipation. The association remained for constipation when adjusted for laxative use. Thus, it appears that the factor increasing the risk of colorectal cancer is constipation and not the laxative abuse. Additional studies question the colorectal cancer association. An investigation by Nusko and colleagues[75] found no association between colorectal cancer and melanosis coli or laxative use. The results of a 26-week study in the transgenic mice strain p53+/− revealed neither drug-related neoplasm nor micronuclei in polychromatic erythrocytes, and did not induce transformations in the in vitro Syrian hamster embryo assay.[76] Subsequent studies have also not found an association;[73,74,77-81] hence, the FDA has categorized bisacodyl as category I (safe and effective). The human evidence appears not to support an association between stimulant cathartics and colorectal cancer. Various authors also question the toxic potential of stimulant laxatives,[82,83] particularly neuronal toxicity leading to cathartic colon.
5.2 Bulk-Forming Laxatives
Bulk, softness and hydration of faeces depend on the fibre content of the diet. Fibre is the part of food that does not undergo enzymatic digestion. It reaches the colon largely unchanged. Colonic bacteria ferment fibre to varying degrees, depending on its chemistry and water solubility. Most of the bulk laxative formulations include a derivative of semi-synthetic fibre (methylcellulose), psyllium or polycarbophil (a hydrophilic resin). Others contain fibre or bran products. These agents are usually manufactured as a powder to be mixed with fluids (water or juice). They absorb water in the intestine to form a viscous liquid that promotes peristalsis and reduces transit time. Insoluble, poorly fermentable fibres have the greatest effect on increasing bulk.
Bulk-forming laxatives soften the faeces.[15,84] Bile acid binding such as with psyllium may reduce the production of low-density lipoprotein.[85-87] Bulk-forming laxatives have a slow onset of action (between 12 and 72 hours) and substantial relief from constipation may take several months of continued use. Due to the longer onset of action, eating disorder patients find these agents inadequate to control weight. Adverse effects with these agents are generally mild and include bloating, allergic reactions and flatulence.[71] Specific contraindications exist, including use in patients with obstructive symptoms and in those with megacolon or megarectum.[15]
5.3 Saline Laxatives
Many saline laxatives contain magnesium salts. Their use results in an increase in osmotic pressure in the bowel, which aids in water retention. In addition, magnesium salts may stimulate the secretion of cholecystokinin, which increases intestinal secretion and motility.[15] The onset of action is dose dependent, with lower doses having an onset of effect in 6–8 hours. The onset of effect for higher doses can be less than 3 hours, such as with the use of magnesium citrate to evacuate the bowel prior to surgical and diagnostic procedures.[15] Adverse effects of these agents include hypotension, hypermagnesaemia, abdominal cramps, diarrhoea, gas formation and respiratory depression (see section 6).
5.4 Osmotic Laxatives
An increase in osmotic pressure in the gut lumen is also caused by osmotic laxatives. They remain unabsorbed and are not digested in the small intestine. Lactulose is metabolized to form fructose and galactose then converted to lactate, acetate and formate. Lactulose helps to reduce the intestinal absorption of ammonia. It has frequently been used to manage hepatic encephalopathy associated with hepatic failure, although its efficacy for this purpose is controversial.[88] This agent is generally well tolerated, and adverse effects including flatulence, cramping, abdominal discomfort, nausea and vomiting. Larger doses can cause diarrhoea, fluid loss, hypokalaemia and hypernatraemia.
Polyethylene glycol (PEG) is an osmotic laxative supported by good-quality evidence that has demonstrated efficacy and safety in the treatment of patients with chronic constipation. While lactulose is more effective in relieving constipation than placebo, it is less effective than PEG. PEG is used prior to procedures such as a colonoscopy. Patients are instructed to drink 4 L of a PEG solution, which results in a watery bowel evacuation. Fluid and electrolyte status is not altered. Adverse effects include urticaria, abdominal bloating, cramping, diarrhoea, flatulence and nausea.[89]
5.5 Surfactants
The docusate compounds are often used as stool-softening agents. They may be combined with other laxatives such as the stimulant compounds. Docusate calcium reduces the surface tension of the oil-water interface of the stool. This results in the incorporation of water and fat, with resultant stool softening. These agents have limited, if any, efficacy in most cases of constipation.[15] Adverse effects of these compounds are mild and include abdominal cramps, rashes and nausea.
5.6 Lubricants
Mineral oil acts as a laxative by decreasing water absorption in the colon, as well as lubricating the intestine. Onset of action is approximately 6–8 hours if administered orally and 2–5 minutes with rectal administration. This agent is not recommended for use in the elderly as aspiration may occur with resulting lipid pneumonitis.
Glycerin is a trihydroxy alcohol that acts as a hygroscopic agent and lubricant when given rectally as a suppository, and results in defecation in about 30 minutes. Adverse reactions associated with glycerin suppositories include local discomfort, burning or hyperaemia, and (minimal) bleeding. Some glycerin suppositories contain sodium stearate, which also can cause local irritation.
6. Medical Complications
Medical complications stemming from laxative ingestion depend on the severity of abuse, as well as the frequency, duration and type of agent used. Problems may be confined to the GI tract or involve systemic features. The most frequent complication of laxative abuse is obviously diarrhoea. GI cramping and pain may present with the diarrhoea due to stool volume and a direct effect of the laxative on intestinal motility. Frequency of stools may reach 15–20 per day.[3] Medical complications can be divided into disturbances involving electrolytes, metabolic issues, bowel, kidney and miscellaneous effects.
6.1 Electrolyte Disturbances
Medical complications of laxative abuse are often a result of chronic diarrhoea and the associated severe electrolyte disturbances. Potassium is the primary electrolyte in stool water (70–90 mmol/L), with lower concentrations of sodium and chloride (30–40 and 15 mmol/L, respectively). With the development of hypokalaemia, the patient may present with generalized muscle weakness and lassitude.[90] Additionally, the presentation may include skeletal muscle paralysis, or rhabdomyolysis with renal impairment, and nerve palsies. More severe hypokalaemia can result in cardiac arrhythmias with an increased risk of sudden death.[3,91] Severe hypokalaemia has been associated with distal renal tubular acidosis in laxative abuse.[92,93] With the expulsion of an added volume of stool water, dehydration, hypotension, tachycardia, postural dizziness and syncope may occur. Renin secretion with secondary hyperaldosteronism can develop with chronic dehydration. Hypermagnesaemia associated with large doses of magnesium-containing laxatives has presented with quadriparesis and neuromuscular junction defects.[94] Recently, it has been reported that patients with congestive heart failure (CHF) but normal renal function who presented with hypermagnesaemia had a lower rate of 3-year survival than patients with normal magnesium levels. These subjects also had a greater antacid/laxative use profile: 82.7% versus 24.8% (p < 0.0001).[95] Patients with pre-existing renal dysfunction are at an increased risk of adverse effects with saline laxatives. However, acute phosphate nephropathy is an accepted complication of the use of phosphate preparations in patients about to undergo to colonoscopy. Patients' CHF may be exacerbated due to the sodium content.
6.2 Metabolic Disturbances
Acid-base disturbances are possible with laxative use. Metabolic alkalosis is the most common acid-base disturbance associated with laxative use, and is related to hypokalaemia, volume contraction and secondary hyperaldosteronism. Concomitant purging by vomiting may increase the risk of developing metabolic alkalosis. Additional acid-base disturbances include hypochloraemic metabolic alkalosis and secondary complications such as increased renal ammoniagenesis,[96] which promotes bicarbonate reabsorption in the renal tubule. Laxative abuse has also been reported to cause GI dysfunction, particularly pancreatic damage.[97] In a small study, Brown et al.[98] compared 18 recovered anorexia nervosa patients with age- and weight-matched controls. Ten of the 18 anorexia nervosa patients had a history of laxative abuse; these subjects showed a more gradual increase and decrease in insulin secretion in response to a standard meal, but no differences in glucose response or hunger ratings. The authors concluded that the difference in insulin response is due to changes in the enteroinsular axis induced by chronic laxative abuse.
6.3 Bowel Disturbances
Bowel dysfunctions including colonic mucosa inflammation and ulceration, ileocaecal sphincter dilation, colonic neuropathy, steatorrhoea and protein-losing gastroenteropathy have been reported with laxative abuse.[99-106] Other presentations include GI bleeding[107] and dehydration with various electrolyte abnormalities.[108] Diarrhoea may alternate with periods of constipation, which causes the patient to enter a vicious cycle alternating between the two.
Direct toxicity to the small intestinal mucosa may be a GI complication of laxative use that leads to steatorrhoea. Rarely, fat-soluble vitamin malabsorption leads to osteomalacia and pseudofractures.[3] Progressive laxative dosing observed in some laxative users may be attributed to hypofunctioning in bowel processes, loss of intrinsic innervation action and laxative tolerance effects.[100] However, controversy exists regarding the effect of senna laxatives on bowel innervation and function. The adverse effects and toxicity of stimulant-type laxatives often include constipation[102] and cathartic colon, which is defined as a loss of colonic myenteric neurons, atrophy of smooth muscle, loss of haustral markings, increased submucosal fat, fibrosis and hypertrophy of the muscularis mucosae.[3,103,109] Barium enema procedures have shown the terminal ileum to be smooth, without a normal mucosal pattern.[8] However, contrary to the widely held belief, stimulant laxatives, which promote intestinal motility, do not seem to lead to bowel injury.[83,109] Recently, Morales and colleagues[110] concluded that there is no convincing evidence that the long-term use of senna causes a structural and/or functional alteration of the enteric nerves or the intestinal smooth muscle. Supporting these findings is the absence of reports of cathartic colon since the removal of phenolpthalein from clinical use. They also report that current evidence does not show that there is a genotoxic risk for patients who take laxatives containing senna extracts or sennosides.
6.4 Kidney Disturbances
Prolonged laxative abuse is also associated with chronic kidney disease and can lead to renal failure.[111] Renal function is reduced by a combination of several factors, including severe hypokalaemia, volume depletion, rhabdomyolysis and hyperuricaemia. In addition, some laxatives are nephrotoxic and cause renal tubular damage.[3] Urolithiasis has also been reported in conjunction with laxative abuse.[112] Wright and DuVal[92] reported five cases of laxative-associated renal damage with electrolyte abnormalities.
6.5 Miscellaneous Disturbances
Recently, laxative use has been reported as a complication in four of seven patients with rectal prolapse and the diagnosis of bulimia nervosa.[113] Causality in these cases was uncertain. A patient intermittently abusing cascara over 2 years was reported to have developed gastric melanosis.[114] Recently, paraesthesias and fascicular and ventricular tachycardia were reported to be associated with an Ayurveda bowel regimen.[115] The preparation included substrates from the Aconitum species, Aconitum heterophyllum (atvish), Aegle marmelos (bilwa), Pavonia adorata (suganda bala), Cyperus rotundus (musta), Picrorrhiza kurrooa (kutki) and Holarrhena antidysenterica (vatsaka). A. heterophyllum (atvish), also known as aconite, monkshood or wolfsbane, was thought to be the most likely offending agent in this combination. Aconite roots contain aconitine, mesaconitine, hypaconitine and other aconitum alkaloids, which are known cardiotoxins and neurotoxins.[115,116]
7. Treatment
The best technique for withdrawing individuals from laxatives has not been systematically ascertained. Whether a patient is habitually abusing laxatives or using them surreptitiously, the main goals of treatment are to stop the laxative use and maintain healthy GI function. However, as a first step, the patient's beliefs regarding the laxative use have to be determined. If the patient is expressing erroneous beliefs regarding the normal number and frequency of bowel movements, education is essential. If the person is suffering from an eating disorder, an appropriate treatment plan for the particular eating disorder should be enacted. In addition, eating disorder patients have been reported to have difficulties with increasing anxiety with laxative discontinuation.[48] This may be related to the retention of fluid with a subsequent increase in weight. The patient may also experience further constipation, which may result in a strong drive to continue to take laxatives. Close supervision is required during this time to ensure that the laxative withdrawal is successful.
Another risk that needs to be appreciated in laxative withdrawal is the development of CHF. This problem is illustrated by the case of a 60-year-old woman who presented with hypokalaemia and weakness.[117] She was consuming a laxative combination of phenolphthalein and rhubarb in doses above the package recommendation. On presentation her potassium was 2.6 mmol/L. She had no signs of CHF at that time. The laxatives were withdrawn and oral potassium supplements started. Over the next 10 days she began to experience oedema, a weight gain of 15 kg and breathlessness. By day 10, her chest radiograph showed cardiomegaly and bilateral pleural effusions. Furosimide and captopril were initiated. She recovered over the next 2 weeks. The depletion of sodium and water through diarrhoea leads to an increase in renin secretion with secondary hyperaldosteronism. Water retention develops with oedema and, in this case, the development of CHF. Oedema following laxative withdrawal is not uncommon, but usually subsides over a period of weeks as sodium balance becomes re-established.
Several protocols have been published regarding the intervention methods designed to reduce and eliminate the use of laxatives. Harper et al.[118] reported a 6-month prospective trial evaluating a pharmacist-supervised, blinded withdrawal protocol. Ten eating disorder patients were enrolled in the trial and seven completed the study. Five of the seven reduced their laxative intake by at least 50%. Three of the seven withdrew completely from laxative use. Their protocol called for discontinuation of all stimulant laxatives with the substitution of docusate calcium, psyllium and fruit lax, a mixture of natural ingredients (senna leaves, pitted prunes, figs, pitted dates, dark raisins), as tolerated. In addition, a combination of 30 mL of Magnolax® (formula per mL: magnesium hydroxide 60 mg and mineral oil 0.25 mL) and 30 mL of cascara was initiated in a dose that was based on the patient's previous dose of bisacodyl or an equivalent. The liquid was reduced by 5 mL every 3–7 days until no longer needed (see table III). The withdrawal protocol included patient education concerning normal eating and bowel habits.
Colton et al.[119] reported on the results of their laxative withdrawal protocol at 3 and 20 months' follow-up. Subjects included all patients admitted for laxative withdrawal in their eating disorders programme between 1993 and 1995. They were assessed with a shortened version of the EDE.[120] Their programme consists of the following components: initial contact, which includes abrupt discontinuation of the laxative; an immediate withdrawal stage (before the first normal bowel movement), involving the provision of non-laxative aids to normal bowel function and psychoeducation; and, lastly, the desensitization stage (after the first normal bowel movement). The final stage initially includes visiting a pharmacy with a staff member and no money with which to buy laxatives, and progresses through to visiting the pharmacy alone with money and not purchasing laxatives. Aids to normal bowel function progress over the month and initially include bulk-forming agents with fluids, stool softeners and glycerin suppositories. In the second half of the month, if need be, they progress to a small clear-water enema and, ultimately, the addition of an irrigation agent. This programme resulted in 57% of patients being abstinent from laxatives at follow-up and significant reductions in laxative-related symptom variables.

8. Summary and Conclusion
Abuse of laxatives is not an uncommon occurrence in the general population and is quite frequent among certain groups, such as in those with eating disorders. Both patients with anorexia nervosa and bulimia nervosa utilize laxatives in an attempt to reduce weight and remove unwanted calories, neither of which is possible with laxatives. Other groups that may utilize laxatives in an unhealthy way include athletes, middle-aged adults and the elderly. The motivation in the elderly group is often based on rigid and sometimes false conceptions about the need for daily bowel movements. This type of use can be difficult to identify as the individual may start out with legitimate constipation related to other disease states or concomitant medication use. However, when the frequency and duration of use exceeds the need, medical complications may occur.
Stimulant laxatives are the more frequent type of laxative used in the eating disorder population. Also, the presence of laxative abuse has been reported to be associated with greater psychopathology by some, but not all, investigators. Certainly, the use of such a drastic purging method places these patients at greater risk than a person who does not purge.
In severe cases, the individual may present with electrolyte and acid/base changes that can involve the renal and cardiovascular systems and may become life threatening. Due to the loss of fluid, the renin-aldosterone system becomes activated, which leads to oedema and acute weight gain when the laxative is discontinued.
Treatment begins with a high level of suspicion, particularly when a patient presents with alternating diarrhoea and constipation as well as other GI complaints. Checking serum electrolytes and the acid/base status can identify individuals who may need medical stabilization. At times, it may also be helpful to check the stool water for electrolytes, particularly magnesium, which may be associated with abuse of magnesium-containing laxatives. Once stable, any stimulant laxatives should be discontinued and a fibre/osmotic supplement utilized to establish normal bowel movements. Health education is of paramount importance in order to enable patients to alter their diet and bowel habits so as to avoid the need for future laxative use. In the case of a suspected or confirmed eating disorder, the patient should be referred for psychiatric treatment to lessen the reliance on laxatives as a method to alter weight and shape.
Acknowledgements
No sources of funding were used to assist in the preparation of this review. James Mitchell has received a research grant from GlaxoSmithKline to study orlistat (Alli®). The other authors have no conflicts of interest that are directly relevant to the content of this review.
References
1. Phelps L, Andrea R, Rizzo FG, et al. Prevalence of self- induced vomiting and laxative/medication abuse among female adolescents: a longitudinal study. Int J Eat Disord 1993; 14: 375-8
2. Tozzi F, Thornton LM, Mitchell JE, et al., on behalf of the Price Foundation Collaborative Group. Features associated with laxative abuse in individuals with eating disorders. Psychosom Med 2006; 68: 470-7
Cited Here... | View Full Text | PubMed | CrossRef
3. Baker EH, Sandle GI. Complications of laxative abuse. Annu Rev Med 1996; 47: 127-34
Cited Here... | PubMed | CrossRef
4. Wills S. Drugs and substance misuse: over-the-counter products. Pharm J 1993; 251: 807-10
5. Marquart LF, Sobal J. Weight loss beliefs, practices and support systems for high school wrestlers. J Adolesc Health 1994; 15: 410-5
Cited Here... | PubMed | CrossRef
6. Shelton JH, Santa Ana CA, Thompson DR, et al. Factitious diarrhea induced by stimulant laxatives: accuracy of diagnosis by a clinical reference laboratory using thin layer chromatography. Clin Chem 2007; 53: 85-90
Cited Here... | PubMed | CrossRef
7. Keswani RJ, Sauk J, Kane SV. Factitious diarrhea masquerading as refractory celiac disease. South Med J 2006; 99: 293-5
Cited Here... | View Full Text | PubMed | CrossRef
8. Vanin JR, Saylor KE. Laxative abuse: a hazardous habit for weight control. J Am Coll Health 1989; 37: 227-30
9. Pomeroy C, Mitchell JE, Roerig J, et al. Medical complications of psychiatric illness. Washington, DC: American Psychiatric Publishing, Inc., 2002
10. Seow-Choen F. The physiology of colonic hydrotherapy. Colorectal Dis 2009; 11: 686-8
Cited Here... | View Full Text | PubMed | CrossRef
11. Furness JB, Sanger GJ. Intrinsic nerve circuits of the gastrointestinal tract: identification of drug targets. Curr Opinion Pharmacol 2002; 2: 612-22
12. Galligan J. Pharmacology of synaptic transmission in the enteric nervous system. Curr Opin Pharmacol 2002; 2: 623-9
Cited Here... | PubMed | CrossRef
13. Hansen MB. The enteric nervous system I: organization and classification. Pharmacol Toxicol 2003; 92: 105-13
Cited Here... | View Full Text | PubMed | CrossRef
14. Camilleri M, Murray JA. Chapter 40: diarrhea and constipation. In: Fauci AS, Braunwald E, Kasper DL, et al., editors. Harrison's principles of internal medicine. 17th ed. New York: The McGraw-Hill Companies, 2008: 245-54
15. Pasricha PJ. Chapter 37: treatment of disorders of bowel motility and water flux; antiemetics; agents used in biliary and pancreatic disease. In: Brunton L, Lazo J, Parker K, editors. Goodman & Gilman's the pharmacological basis of therapeutics. 11th ed. New York: McGraw-Hill Professional, 2006: 983-1008
16. Tonini M, Cipollina L, Poluzzi E, et al. Review article: clinical implications of enteric and central D2 receptor blockade by antidopaminergic gastrointestinal prokinetics. Aliment Pharmacol Ther 2004; 19: 379-90
Cited Here... | View Full Text | PubMed | CrossRef
17. Talley NJ. Serotoninergic neuroenteric modulators. Lancet 2001; 358: 2061-8
Cited Here... | PubMed | CrossRef
18. Nguyen NQ, Chapman M, Fraser RJ, et al. Prokinetic therapy for feed intolerance in critical illness: one drug or two? Crit Care Med 2007; 35: 2561-7
Cited Here... | View Full Text | PubMed | CrossRef
19. Neims DM, McNeill J, Giles TR, et al. Incidence of laxative abuse in community and bulimic populations: a descriptive review. Int J Eat Disord 1995; 17: 211-28
Cited Here... | PubMed | CrossRef
20. Abraham SF, Beumont PJ. How patients describe bulimia or binge eating. Psychol Med 1982; 12: 625-35
Cited Here... | PubMed | CrossRef
21. Cooper PJ, Fairburn CG. Cognitive behaviour therapy for anorexia nervosa: some preliminary findings. J Psychosom Res 1984; 28: 493-9
Cited Here... | PubMed | CrossRef
22. Mitchell JE, Boutacoff LL, Hatsukami D, et al. Characteristics of 275 patients with bulimia. Am J Psychiatry 1985; 142: 462-85
23. Mitchell JE, Pomeroy C, Seppala M, et al. Diuretic use as a marker for eating problems and affective disorders among women. J Clin Psychiatry 1988; 49: 267-70
24. Steffen KJ, Mitchell JE, Roerig JL. The eating disorders medicine cabinet revisited: a clinician's guide to ipecac and laxatives. Int J Eat Disord 2007; 40: 360-8
Cited Here... | PubMed | CrossRef
25. Steffen KJ, Roerig JL, Mitchell JE, et al. A survey of herbal and alternative medication use among participants with eating disorder symptoms. Int J Eat Disord 2006; 39: 741-6
Cited Here... | PubMed | CrossRef
26. Wade TD. A retrospective comparison of purging type disorders: eating disorder not otherwise specified and bulimia nervosa. Int J Eat Disord 2007; 40: 1-6
Cited Here... | PubMed | CrossRef
27. Turner J, Batik M, Palmer LJ, et al. Detection and importance of laxative use in adolescents with anorexia nervosa. J Am Acad Child Adolesc Psychiatry 2000 Mar; 39 (3): 378-85
Cited Here... | View Full Text | PubMed | CrossRef
28. Bytzer P, Stokholm M, Andersen I, et al. Prevalence of surreptitious laxative abuse in patients with diarrhea of uncertain origin: a cost benefit analysis of a screening procedure. Gut 1989; 30: 1379-84
Cited Here... | PubMed | CrossRef
29. Duncan A, Morris AJ, Cameron A, et al. Laxative induced diarrhea: a neglected diagnosis. J R Soc Med 1992; 85: 203-5
30. Harari D, Gurwitz JH, Avorn J, et al. Constipation: assessment and management in an institutionalized elderly population. J Am Geriatr Soc 1994; 42: 947-52
31. Talley NJ. Definitions, epidemiology, and impact of chronic constipation. Rev Gastroenterol Disord 2004; 4 Suppl. 2: S3-10
32. Primrose WR, Capewell AE, Simpson GK. Prescribing patterns observed in registered nursing homes and long-stay geriatric wards. Age Ageing 1987; 16: 25-8
Cited Here... | PubMed | CrossRef
33. Read NW, Krejs GJ, Read MG, et al. Chronic diarrhea of unknown origin. Gastroenterology 1980; 78: 264-71
34. Harris A, Buchanan GN. Melanosis coli is reversible. Colorectal Dis 2009; 11: 788-9
Cited Here... | View Full Text | PubMed | CrossRef
35. Greenfeld D, Mickley D, Quinlan DM, et al. Hypokalemia in outpatients with eating disorders. Am J Psychiatry 1995; 152: 60-3
36. Eherer AJ, Fordtran JS. Fecal osmotic gap and pH in experimental diarrhea of various causes. Gastroenterology 1992; 103: 545-51
37. Phillips S, Donaldson L, Geisler K, et al. Stool composition in factitial diarrhea: a 6-year experience with stool analysis. Ann Intern Med 1995; 123: 97-100
38. Duncan A. Screening for surreptitious laxative abuse. Ann Clin Biochem 2000; 37: 1-8
Cited Here... | PubMed | CrossRef
39. Fine KD, Santa Ana CA, Fordtran JS. Diagnosis of magnesium-induced diarrhea. N Engl J Med 1991; 324: 1012-7
Cited Here... | PubMed | CrossRef
40. Mayo Foundation for Medical Education and Research. Mayo 2010 test catalog. Rochester (MN): Mayo Foundation for Medical Education and Research, 2010
41. Kovacs D, Palmer RL. The associations between laxative abuse and other symptoms among adults with anorexia nervosa. Int J Eat Disord 2004; 36: 224-8
Cited Here... | PubMed | CrossRef
42. Lacey JH, Gibson E. Does laxative abuse control body weight? A comparative study of purging and vomiting bulimics. Hum Nutr Appl Nutr 1985; 39: 36-42
43. Bo-Linn GW, Santa Ana CA, Morawski SG, et al. Purging and calorie absorption in bulimic patients and normal women. Ann Intern Med 1983; 99: 14-7
44. Pryor T, Wiederman MW, McGilley B. Laxative abuse among women with eating disorders: an indication of psychopathology? Int J Eat Disord 1996; 20: 13-8
Cited Here... | PubMed | CrossRef
45. Rosenberg M. Society and the adolescent self-image. Princeton (NJ): Princeton University Press, 1965
46. Surgenor LJ, Maguire S, Russell J, et al. Self-liking and self-competence: relationship to symptoms of anorexia nervosa. Eur Eat Disord Rev 2007; 15: 139-45
Cited Here... | PubMed | CrossRef
47. Bryant-Waugh R, Turner H, East P, et al. Misuse of laxatives among adult outpatients with eating disorders: prevalence and profiles. Int J Eat Disord 2006; 39: 404-9
Cited Here... | PubMed | CrossRef
48. Weltzin TE, Bulik CM, McConaha CW, et al. Laxative withdrawal and anxiety in bulimia nervosa. Int J Eat Disord 1995; 17: 141-6
Cited Here... | PubMed | CrossRef
49. Wiederman MW, Pryor T. Multi-impulsivity among women with bulimia nervosa. Int J Eat Disord 1996; 20: 359-65
Cited Here... | PubMed | CrossRef
50. Favaro A, Santonastaso P. Impulsive and compulsive self-injurious behavior in bulimia nervosa: prevalence and psychological correlates. J Nerv Ment Dis 1998; 186: 157-65
Cited Here... | View Full Text | PubMed | CrossRef
51. Mond JM, Hay PJ, Rodgers B, et al. Correlates of self-induced vomiting and laxative misuse in a community sample of women. J Nerv Ment Dis 2006; 194: 40-6
Cited Here... | View Full Text | PubMed | CrossRef
52. Haedt AA, Edler C, Heatherton TF, et al. Importance of multiple purging methods in the classification of eating disorder subtypes. Int J Eat Disord 2006; 39: 648-54
Cited Here... | PubMed | CrossRef
53. Hatsukami D, Eckert E, Mitchell JE, et al. Affective disorders and substance abuse in women with bulimia. Psychol Med 1984; 14: 701-4
Cited Here... | PubMed | CrossRef
54. Mitchell JE, Hatsukami D, Pyle RL, et al. Bulimia with and without a family history of depressive illness. Compr Psychiatry 1986; 27: 215-9
Cited Here... | PubMed | CrossRef
55. Cance JD, Ashley OS, Penne MA. Unhealthy weight control behaviors and MDMA ecstasy use among adolescent females. J Adolesc Health 2005; 37: 409
Cited Here... | PubMed | CrossRef
56. Hatsukami D, Mitchell JE, Eckert ED, et al. Characteristics of patients with bulimia only, bulimia with affective disorders, and bulimia with substance abuse problems. Addict Behav 1986; 11: 399-406
Cited Here... | PubMed | CrossRef
57. Jonas JM, Gold MS, Sweeney D, et al. Eating disorders and cocaine abuse: a survey of 259 cocaine abusers. J Clin Psychiatry 1987; 48: 47-50
58. Johnson CL, Love SQ. Bulimia: multivariate predictors of life impairment. J Psychiatr Res 1985; 19: 343-7
Cited Here... | PubMed | CrossRef
59. Latner JD, Vallance JK, Buckett G. Health-related quality of life in women with eating disorders: association with subjective and objective binge eating. J Clin Psychol Med Settings 2008; 15: 148-53
Cited Here... | PubMed | CrossRef
60. Anderson CB, Carter FA, McIntosh VV, et al. Self-harm and suicide attempts in individuals with bulimia nervosa. Eat Disord 2002; 10: 227-43
61. Duncan A, Forest JAH. Surreptitious abuse of magnesium laxatives as a cause of chronic diarrhea. Eur J Gastroenterol Hepatol 2001; 13: 599-601
Cited Here... | View Full Text | PubMed | CrossRef
62. McQuaid KR. Chapter 15: gastrointestinal disorders. In: McPhee SJ, Papadakis MA, editors. CURRENT Medical Diagnosis & Treatment 2010 [online]. Available from URL: http://www.accessmedicine.com.ezproxy.undmedlibrary.org/content.aspx?aID=6395 [Accessed 2010 Jan 15]
63. Kreek MJ. Constipation syndromes. In: Lewis JH, editor. A pharmacological approach to gastrointestinal disorders. Baltimore (MD): Williams & Wilkins, 1994: 179-208
64. Izzo AA, Gaginella TS, Mascolo N, et al. Recent findings on the mode of action of laxatives: the role of platelet activating factor and nitric oxide. Trends Pharmacol Sci 1998; 19: 403-5
Cited Here... | PubMed | CrossRef
65. Willard SG, Winstead DK, Anding R, et al. Laxative detoxification in bulimia nervosa. In: Johnson WG, editor. Advances in eating disorders: bulimia nervosa: perspectives on clinical research and therapy. Greenwich (CT): JAI Press Inc., 1989
66. Coogan PF, Rosenberg L, Palmer JR, et al. Phenolphthalein laxatives and risk of cancer. J Natl Cancer Inst 2000; 23: 1943-4
Cited Here... | PubMed | CrossRef
67. Cooper GS, Longnecker MP, Peters RK. Ovarian cancer risk and use of phenolphthalein-containing laxatives. Pharmacoepidemiol Drug Saf 2004; 13: 35-9
Cited Here... | PubMed | CrossRef
68. US Food and Drug Administration. Laxative drug products for over-the-counter human use; proposed amendment to the tentative final monograph. Fed Regist 1997 Sep 2; 62: 46223-7
69. Dunnick JK, Hardisty JF, Herbert RA, et al. Phenolphthalein induces thymic lymphomas accompanied by loss of the p53 wild type allele in heterozygous p53-deficient +/− mice. Toxicol Pathol 1997; 25: 533-40
Cited Here... | PubMed | CrossRef
70. Siegers CP, von Hertzberg-Lottin E, Otte M, et al. Anthranoid laxative abuse: a risk for colorectal cancer? Gut 1993; 34: 1099-101
Cited Here... | PubMed | CrossRef
71. Xing JH, Soffer EE. Adverse effects of laxatives. Dis Colon Rectum 2001; 44: 1201-9
Cited Here... | View Full Text | PubMed | CrossRef
72. Saunders DR, Sillery J, Rachmilewitz D, et al. Effect of bisacodyl on structure and function of rodent and human intestine. Gastroenterology 1977; 72: 849-56
73. Sonnenberg A, Müller AD. Constipation and cathartics as risk factors of colorectal cancer: a meta-analysis. Pharmacology 1993; 47 Suppl. 1: 224-33
Cited Here... | PubMed | CrossRef
74. Jacobs EJ, White E. Constipation, laxative use, and colon cancer among middle-aged adults. Epidemiology 1998; 9: 385-91
Cited Here... | View Full Text | PubMed | CrossRef
75. Nusko G, Schneider B, Ernst H, et al. Melanosis coli: a harmless pigmentation or a precancerous condition? Z Gastroenterol 1997; 35: 313-8
76. Stoll RE, Blanchard KT, Stoltz JH, et al. Phenolphthalein and bisacodyl: assessment of genotoxic and carcinogenic responses in heterozygous p53 (+/−) mice and Syrian hamster embryo (SHE) assay. Toxicol Sci 2006; 90: 440-50
Cited Here... | PubMed | CrossRef
77. Nusko G, Schneider B, Schneider I, et al. Anthranoid laxative use is not a risk factor for colorectal neoplasia: results of a prospective case control study. Gut 2000; 46: 651-5
Cited Here... | View Full Text | PubMed | CrossRef
78. Dukas L, Willett WC, Colditz GA, et al. Prospective study of bowel movement, laxative use, and risk of colorectal cancer among women. Am J Epidemiol 2000; 151: 958-64
Cited Here... | View Full Text | PubMed
79. Kune GA, Kune S, Field B, et al. The role of chronic constipation, diarrhea, and laxative use in the etiology of large-bowel cancer: data from the Melbourne Colorectal Cancer Study. Dis Colon Rectum 1988; 31: 507-12
Cited Here... | View Full Text | PubMed
80. Nascimbeni R, Donato F, Ghirardi M, et al. Constipation, anthranoid laxatives, melanosis coli, and colon cancer: a risk assessment using aberrant crypt foci. Cancer Epidemiol Biomarkers Prev 2002; 11: 753-7
81. Roberts MC, Millikan RC, Galanko JA, et al. Constipation, laxative use, and colon cancer in a North Carolina population. Am J Gastroenterol 2003; 98: 857-64
Cited Here... | PubMed | CrossRef
82. Fioramonti J, Bueno L. Toxicity of laxatives: how to discriminate between myth and fact? Eur J Gastroenterol Hepatol 1995; 7: 5-7
83. Muller-Lissner SA, Kamm MA, Scarpignato C, et al. Myths and misconceptions about chronic constipation. Am J Gastroenterol 2005; 100: 232-42
Cited Here... | PubMed | CrossRef
84. Drug facts and comparisons 2006. 60th ed. Philadelphia (PA): Lippincott, Williams & Wilkins, 2006
85. Anderson JW, Zettwoch N, Feldman T, et al. Cholesterol-lowering effects of psyllium hydrophilic mucilloid for hypercholesterolemic men. Arch Intern Med 1988; 148: 292-6
Cited Here... | PubMed | CrossRef
86. Bell LP, Hectorne K, Reynolds H, et al. Cholesterol-lowering effects of psyllium hydrophilic mucilloid: adjunct therapy to a prudent diet for patients with mild to moderate hypercholesterolemia. JAMA 1989; 261: 3419-23
Cited Here... | PubMed | CrossRef
87. Levin EG, Miller VT, Muesing RA, et al. Comparison of psyllium hydrophilic mucilloid and cellulose as adjuncts to a prudent diet in the treatment of mild to moderate hypercholesterolemia. Arch Intern Med 1990; 150: 1822-7
Cited Here... | PubMed | CrossRef
88. Als-Nielsen B, Gluud LL, Gluud C. Nonabsorbable disaccharides for hepatic encephalopathy. Cochrane Database Syst Rev 2004; 2: CD003044
89. Loening-Baucke V, Pashankar DS. A randomized, prospective, comparison study of polyethylene glycol 3350 without electrolytes and milk of magnesia for children with constipation and fecal incontinence. Pediatrics 2006; 118: 528-35
Cited Here... | PubMed | CrossRef
90. Slugg PH, Carey WD. Clinical features and follow-up of surreptitious laxative users. Cleveland Clin Q 1984; 51: 167-71
91. Krahn LE, Lee J, Richardson JW, et al. Hypokalemia leading to torsades de pointes: Munchausen's disorder or bulimia nervosa? Gen Hosp Psychiatry 1997; 19: 370-7
Cited Here... | PubMed | CrossRef
92. Wright LF, DuVal Jr JW. Renal injury associated with laxative abuse. South Med J 1987; 80: 1304-6
Cited Here... | View Full Text | PubMed | CrossRef
93. Vanderperren B, Rizzo M, Angenot L, et al. Acute liver failure with renal impairment related to the abuse of senna anthraquinone glycosides. Ann Pharmacother 2005; 39: 1353-7. Epub 2005 Jun 14
94. Qureshi T, Melonakos TK. Acute hypermagnesemia after laxative use. Ann Emerg Med 1996; 28: 552-5
Cited Here... | PubMed | CrossRef
95. Corbi G, Acanfora D, Iannuzzi GL, et al. Hypermagnesemia predicts mortality in elderly with congestive heart disease: relationship with laxative and antacid use. Rejuvenation Res 2008; 11: 129-38
96. Sebastian AN, Hulter HN, Rector FC. Metabolic alkalosis. In: Brenner BM, Stein JH, editor. Contemporary issues in nephrology, acid-base and potassium homeostasis. New York: Churchill-Livingstone, 1978: 101-36
97. Kobayashi N, Tamai H, Uehata S, et al. Pancreatic abnormalities in patients with eating disorders. Psychosom Med 1988; 50: 607-14
98. Brown NW, Treasure JL, Campbell IC. Evidence for long-term pancreatic damage caused by laxative abuse in subjects recovered from anorexia nervosa. Int J Eat Disord 2001; 29: 236-8
Cited Here... | PubMed | CrossRef
99. Cummings JH. Laxative abuse. Gut 1974; 15: 758-66
Cited Here... | PubMed | CrossRef
100. Cummings JH, Sladen GE, James OF, et al. Laxative-induced diarrhoea: a continuing clinical problem. BMJ 1974; 23: 537-41
Cited Here... | PubMed | CrossRef
101. LaRusso NF, McGill DB. Surreptitious laxative ingestion. Delayed recognition of a serious condition: a case report. Mayo Clin Proc 1975; 50: 706-8
102. Cooke WT. Laxative abuse. Clin Gastroenterol 1977; 6: 659-73
103. Kim SK, Gerle RD, Rozanski R. Cathartic colitis. AJR Am J Roentgenol 1978; 131: 1079-81
104. Sansone RA. Complications of hazardous weight-loss methods. Am Fam Physician 1984; 30: 141-6
105. Cuellar RE, Van Thiel DH. Gastrointestinal consequences of the eating disorders: anorexia nervosa and bulimia. Am J Gastroenterol 1986; 81: 1113-24
106. Mitchell JE, Seim HC, Colon E, et al. Medical complications and medical management of bulimia. Ann Intern Med 1987; 107: 71-7
107. Weiss BD, Wood GA. Laxative abuse causing gastrointestinal bleeding. J Fam Pract 1982; 15: 177-81
108. Mitchell JE, Pyle RL, Eckert ED, et al. Electrolyte and other physiological abnormalities in patients with bulimia. Psychol Med 1983; 13: 273-8
Cited Here... | PubMed | CrossRef
109. Wald A. Is chronic use of stimulant laxatives harmful to the colon? J Clin Gastroenterol 2003; 36: 386-9
Cited Here... | View Full Text | PubMed | CrossRef
110. Morales MA, Hernández D, Bustamante S, et al. Is senna laxative use associated to cathartic colon, genotoxicity, or carcinogenicity? J Toxicol 2009; 2009: 287247
111. Copeland PM. Renal failure associated with laxative abuse. Psychother Psychosom 1994; 62: 200-2
Cited Here... | PubMed | CrossRef
112. Wu WJ, Huang CH, Chiang CP, et al. Urolithiasis related to laxative abuse. J Formos Med Assoc 1993; 92: 1004-6
113. Malik M, Stratton J, Sweeney WB. Rectal prolapse associated with bulimia nervosa: report of seven cases. Dis Colon Rectum 1997; 40: 1382-5
Cited Here... | View Full Text | PubMed | CrossRef
114. Mitty RD, Wolfe GRZ, Cosman M. Initial description of gastric melanosis in a laxative-abusing patient. Am J Gastroenterol 1997; 92: 707-8
115. Chan TY. Aconite poisoning. Clin Toxicol 2009; 47: 279-85
116. Lin CC, Chan TY, Deng JF. Clinical features and management of herb-induced aconitine poisoning. Ann Emerg Med 2004; 43: 574-9
Cited Here... | PubMed | CrossRef
117. Riley JA, Brown AR, Walker BE. Congestive cardiac failure following laxative withdrawal. Postgrad Med J 1996; 72: 491-2
Cited Here... | PubMed | CrossRef
118. Harper J, Leung M, Birmingham CL. A blinded laxative taper for patients with eating disorders. Eat Weight Disord 2004; 9: 147-50
119. Colton P, Woodside DB, Kaplan AS. Laxative withdrawal in eating disorders: treatment protocol and 3 to 20-month follow-up. Int J Eat Disord 1999; 25: 311-7
Cited Here... | PubMed | CrossRef
120. Cooper PJ, Fairburn CG. The eating disorders examination: a semi-structured interview for the assessment of the specific psychopathology of the eating disorders. Int J Eat Disord 1987; 6: 1-8
Cited Here... | PubMed | CrossRef
請先 登入 以發表留言。