
Sanford, Mark
Abstract
Roflumilast is an orally administered, selective phosphodiesterase 4 inhibitor that is a novel treatment for patients with severe chronic obstructive pulmonary disease (COPD).
Across four randomized, double-blind, multinational trials in patients with moderate to severe or severe COPD, roflumilast 500 μg/day produced significantly greater improvements from baseline than placebo in pre- and post-bronchodilator forced expiratory volume in 1 second (FEV1). While mean increases in FEV1 in roflumilast groups were small, they were in marked contrast to the decreases or negligible increase in FEV1 in the placebo groups. In three of four trials, roflumilast significantly reduced the COPD exacerbation rate; in a pooled analysis of two 52-week trials, roflumilast was associated with a 17% reduction over placebo.
Further randomized, double-blind, multinational trials compared roflumilast 500 μg plus salmeterol with placebo plus salmeterol and roflumilast 500 μg plus tiotropium bromide with placebo plus tiotropium bromide. In both trials, roflumilast plus long-acting bronchodilator produced significantly greater increases in pre- and post-bronchodilator FEV1 than placebo plus long-acting bronchodilator.
Roflumilast had an acceptable tolerability profile. In a pooled analysis of two trials, 19% and 22% of roflumilast and placebo recipients had serious adverse events; in the corresponding groups, 14% and 11% discontinued medications because of adverse events.


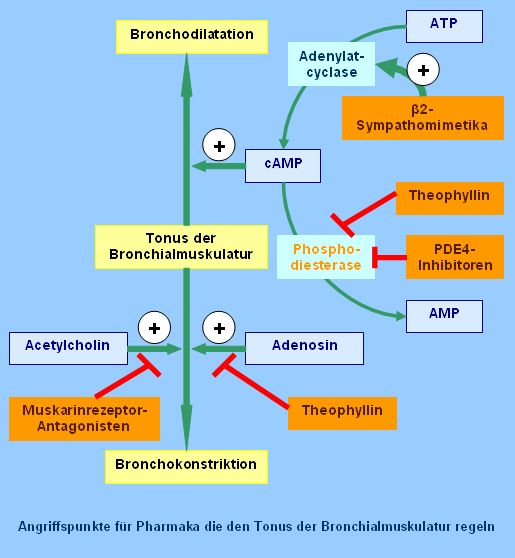
Chronic obstructive pulmonary disease (COPD) is a progressive inflammatory disease of the lung associated with persistent obstruction of lung air flow.[1] COPD responds poorly to bronchodilators and worsens over time.[1] Worldwide, it is a major cause of morbidity and mortality, as it is currently the sixth most frequent cause of death, with patients also suffering from poor quality of life and from the effects of co-morbid disorders.[1] The key clinical features of COPD according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) are dyspnoea, chronic cough, chronic sputum production and a history of exposure to tobacco smoke, occupational dusts and chemicals or smoke from home cooking and heating, with the diagnosis confirmed by standardized spirometry wherever possible.[2] COPD severity is graded I–IV (mild, moderate, severe and very severe) according to airflow limitation based on spirometry findings, symptoms, complications and exacerbations.[2]
COPD exacerbations are associated with lower respiratory tract bacterial and viral infections and environmental pollution, and become more severe and frequent in association with worsening of the disease.[3,4] During exacerbations, there is increased inflammation, both systemically and in upper and lower airways, which manifests as bronchospasm, airways oedema, increased production of sputum, further limitation of airflow and lung hyperinflation.[3,5] It appears that one exacerbation increases susceptibility to subsequent exacerbations,[3] and possibly speeds up the loss of lung function.[4] Therefore, although there is controversy regarding the definition of exacerbation,[4] the prevention of exacerbations is recognized as an important treatment goal, alongside relieving symptoms, improving health status, improving functioning and reducing mortality.[1-3]
In COPD, inflammation results in increased mucus production and chronic bronchitis in large airways, obstructive bronchiolitis stemming from fibrosis of bronchioles in association with small airway remodelling, and centrilobular emphysema arising from damage to third order bronchioles, alveoli and lung interstitium.[4] A variety of inflammatory cells are involved in lung inflammation, including T lymphocytes, neutrophils and macrophages, and these are further increased during exacerbations, as are various cytokines, chemokines and proteases.[4] As systemic manifestations of inflammation may worsen co-morbid conditions, targeting systemic inflammation may also prove to be an important component of COPD treatment.[6]
In mild COPD, short-acting bronchodilators in combination with strategies to reduce risk factors (chiefly avoidance of tobacco smoke) are the key therapy strategies.[2] In moderate COPD, long-acting bronchodilators (β2-adrenergic receptor agonists and muscarinic receptor antagonists) are recommended for use as needed, while in patients with severe COPD with repeated exacerbations, long-acting bronchodilators used concomitantly with inhaled corticosteroids are recommended,[2] as they significantly reduce exacerbations[1] and may protect against decline in lung function and reduce COPD mortality.[1,7] Response to treatment is assessed by ongoing clinical evaluation and spirometry.[2] However, change in forced expiratory volume in 1 second (FEV1) with treatment is not a strong predictor of change in other clinical endpoints, such as improvement in exercise capacity, dyspnoea or perceived breathlessness.[8,9] Small changes in FEV1 can be associated with other changes in lung function that contribute to reduced perceived breathlessness.[9] Thus, reliance on FEV1 can lead to an underestimate of true clinical benefit from treatment.[8]
New anti-inflammatory drugs are required that are effective for the prevention and treatment of exacerbations.[4] Roflumilast (Daxas®) is an orally administered, selective phosphodiesterase 4 (PDE4) inhibitor with anti-inflammatory properties.[10] The European Medicines Agency has recently authorized roflumilast maintenance treatment, as an add-on to bronchodilator therapy, in adult patients with severe COPD (post-bronchodilator FEV1 <50% predicted) associated with chronic bronchitis and a history of frequent exacerbations,[11] making it the first drug in its class that is available for use in COPD. The US FDA advisory panel (April 2010) voted against approval of roflumilast at this time and have requested additional information and analyses of existing trials for their continuing review.[12]
This review focuses on the efficacy and tolerability of roflumilast in patients with moderate to severe or severe COPD, as the primary analyses in roflumilast clinical trials include data from these populations. The review also overviews the pharmacological properties of roflumilast. Medical literature on the use of roflumilast in COPD was identified using MEDLINE and EMBASE, supplemented by AdisBase (a proprietary database). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug. Searches were last updated 23 July 2010.
1. Pharmacodynamic Profile
This section reports information from a comprehensive review of roflumilast pharmacology,[13] and fully published preclinical[14-16] and clinical studies of roflumilast in patients with COPD.[17,18]
Mechanism of Action
* Roflumilast is a selective PDE4 inhibitor that has no PDE4 subtype selectivity.[13] PDE4 is present in human tissues, including cells of haematopoietic origin, and is a catalyst of cyclic adenosine monophosphate (cAMP) degradation. As such, it is a key second messenger system of cellular signalling, with a broad range of effects that include the regulation and enhancement of cAMP activity in proinflammatory cells and of the remodelling functions of these cells. Thus, by inhibiting PDE4, roflumilast exerts broad anti-inflammatory effects by interfering in the functioning of proinflammatory cells.[13]
Preclinical Studies
* In vitro, roflumilast is a potent inhibitor of all PDE4 isoenzymes, although with slightly less potent inhibition of the PDE4C subtype; roflumilast does not affect any other PDE isoenzymes.[13] The roflumilast concentrations that produced 50% inhibition (IC50) of PDE catalysis were <1 nmol/L for PDE4A, PDE4B and PDE4D isoenzymes (range 0.2–0.9 nmol/L) and ≥3 for PDE4C isoenzymes (range 3–4.3 nmol/L).[13] Nonselectivity for PDE4 isoenzymes may advantage roflumilast over the PDE4 inhibitor cilomilast.[13] Cilomilast shows some selectivity for PDE4D (IC50 of ≤0.03 nmol/L) and PDE4D inhibition may be linked to emesis and/or adverse cardiovascular effects in patients who are at risk of heart failure.[13]
* The anti-inflammatory effects of roflumilast, have been fully documented in cellular and animal models of COPD.[13] These include inhibition of neutrophil influx into airways after short-term exposure to tobacco; marked reduction in the influx of cells characterizing the innate and adaptive immune response (including neutrophils, macrophages and lymphocytes) in response to chronic exposure to tobacco smoke; and reduction of bleomycin-induced inflammatory responses that are associated with oxidative stress. Oxidative stress may be important in tobacco smoke-induced lung inflammation and in the development of emphysema.[13] In animals, roflumilast also inhibits fibrotic remodelling of lungs and vascular remodelling, which can lead to pulmonary hypertension.[13]
* The therapeutic effects of roflumilast in COPD may be related in part to enhanced ciliary activity, resulting in clearance of mucous from small airways.[14] In vitro, roflumilast and the PDE4 inhibitor rolipram significantly increased ciliary beat frequency in central and lateral airways in rat lung (all p < 0.001 at 60 minutes vs baseline), whereas the β2-adrenergic receptor agonist terbutaline and colforsin (forskolin) increased ciliary beat frequency in central airways (p < 0.001 vs baseline), but not in lateral airways.[14]
* In mice chronically exposed to cigarette smoke, roflumilast prevented destruction of lung parenchyma.[15] Animals who were exposed to three cigarettes per day for 7 months and who were untreated or received oral roflumilast 1 mg/kg/day developed foci of emphysema throughout lung parenchyma, whereas there was no difference between the lungs of cigarette-exposed animals who received roflumilast 5 mg/kg/day and animals who were not exposed to cigarette smoke.[15]
* In healthy nonsmokers, oral roflumilast reduced the recruitment of inflammatory cells into airways after a bronchoalveolar endotoxin challenge.[16] In a randomized, double-blind, single-centre trial, participants (n = 37) were treated with roflumilast 500 μg or placebo once daily for 28 days, after which saline and endotoxin was installed in different lung segments during bronchoscopy. Compared with placebo recipients, roflumilast recipients had a ≈36% reduction in total cells in a bronchoalveolar lavage performed 24 hours after the endotoxin challenge (p = 0.02), chiefly because of significant reductions in eosinophils (−74% vs placebo; p = 0.01) and neutrophils (−39% vs placebo; p = 0.02). There were no significant between-group differences in soluble inflammatory markers in the lavage fluid.[16]
Clinical Studies
* In patients with COPD, oral roflumilast reduced inflammatory cells in the sputum.[17] In a randomized, placebo-controlled, crossover trial, patients with COPD who at baseline had no exacerbation, no upper respiratory tract infection and sputum neutrophilia with ≥45% nonsquamous cells (n = 38 in analyses) were randomized to roflumilast 500 μg or placebo once daily for 4 weeks, with a 4- to 6-week washout period between treatments. Roflumilast treatment led to a reduction in total sputum inflammatory cell count, whereas placebo was associated with an increase in cell count (mean between-group difference 34%; p = 0.002). Compared with placebo, roflumilast treatment significantly reduced the absolute number of neutrophils (p = 0.002) [primary endpoint], eosinophils (p < 0.001) and lymphocytes (p = 0.022) in sputum, while there were no significant between-group differences in sputum weight, macrophages or differential cell counts.[17]
* During roflumilast treatment, soluble markers of neutrophil (interleukin-8 and neutrophil elastase) and eosinophil (eosinophil cationic protein) activity were also significantly reduced (all p < 0.05 vs placebo), as were sputum levels of α2-macroglobulin (a marker of microvascular leakage) [p < 0.001 vs placebo].[17] Sputum lactoferrin, a marker of neutrophil activity that is also produced by submucosal glands, was not significantly lowered by roflumilast. Ex vivo stimulation of whole blood cultures by lipopolysaccharide was associated with a significant reduction in tumour necrosis factor-α (p = 0.047), but not in E-selectin.[17]
* In two large placebo-controlled trials in patients with severe COPD treated with oral roflumilast 500 μg once daily for 52 weeks (M2-124 [n = 1385 in analysis] and M2-125 [n = 1376 in analysis[18]]; see section 3 for trial details), there was no significant change from baseline to the last post-randomization visit in least squares mean C-reactive protein levels in roflumilast or placebo groups, and there were no significant differences between roflumilast and placebo recipients in C-reactive protein levels with treatment.[18]
2. Pharmacokinetic Profile
The pharmacokinetics of roflumilast were evaluated in randomized, open-label, crossover studies,[19,20] and in a population pharmacokinetic study.[21] Supplemental data are from the EU summary of product characteristics.[11]
In a study of single and repeated doses of roflumilast, healthy adults (n = 15 in analyses) were randomized to 250 or 500 μg of roflumilast administered orally on the morning of day 1, with the same dose repeated once daily from days 5 to 12; participants subsequently crossed over to the second dose condition.[19] The effect of food on roflumilast pharmacokinetics was evaluated in healthy adults (n = 12) who received oral roflumilast 500 μg after an overnight fast and 5 minutes after a high-fat breakfast in a crossover manner.[20] The population pharmacokinetic study modelled data from 26 phase I studies in healthy adults and two phase II and III trials in patients with COPD.[21] Total PDE4 inhibitory activity was based on a composite measure that incorporated the in vivo exposure, free plasma fraction and intrinsic activity of roflumilast and of its metabolite roflumilast N-oxide.[21] Roflumilast N-oxide pharmacokinetic parameters are clinically relevant as this metabolite is clinically active and, on average, its systemic exposure is approximately 10-fold that of roflumilast.[11]
Absorption and Distribution
* In healthy adults, oral roflumilast was rapidly absorbed and metabolized to its active metabolite, roflumilast N-oxide; roflumilast and roflumilast N-oxide plasma exposures were dose-proportional with increased dose when given in single or repeated doses over a range of 250 to 1000 μg.[11] After a single oral dose of roflumilast 500 μg, the geometric mean maximum plasma concentration (Cmax) and area under the plasma concentration-time curve (AUC) from time zero to infinity (AUC∞) values of roflumilast and its metabolite roflumilast N-oxide were 5.3 and 9.4 μg/L and 35.0 and 351.3 μg · h/L. The median times to roflumilast and roflumilast N-oxide Cmax (tmax) were 1.3 and 11.0 hours[19] (corresponding median values of ≈1 and ≈8 hours are cited in the summary of product characteristics).[11]
* Steady-state plasma concentrations of roflumilast and roflumilast N-oxide were reached within 7 days after repeated administration of roflumilast 500 μg once daily.[19] The roflumilast and roflumilast N-oxide Cmax, AUC∞ and tmax values were 6.0 and 21.7 μg/L, 33.7 and 375.4 μg · h/L, and 1.0 and 4.0 hours, respectively.[19] The roflumilast N-oxide Cmax at steady-state was approximately 2-fold that of the single dose. There was no significant change in the tmax with a higher dose (250 vs 500 μg) or with repeated doses (tmax ranges for roflumilast and roflumilast N-oxide for all conditions were 0.5–2.0 and 1.0–12.0 hours).[19]
* Ingestion of food with roflumilast had no clinically important effect on roflumilast pharmacokinetics.[20] After a high-fat meal, there was an estimated ≈40% reduction in the geometric mean roflumilast Cmax, a modest increase in roflumilast AUC values, and the mean tmax was 2.0 hours (vs 1 hour after fasting). Roflumilast N-oxide Cmax, AUC and tmax values were unchanged.[20]
* The absolute bioavailability after administration of a roflumilast tablet was ≈80%.[11] The roflumilast volume of distribution after a dose of roflumilast 500 μg was ≈2.9 L/kg.[11] Roflumilast and roflumilast N-oxide are highly protein bound, but binding was nonsaturable at clinically relevant concentrations.[19]
Metabolism and Elimination
* Roflumilast is metabolized by hepatic cytochrome P450 (CYP) enzymes, predominantly CYP3A4 and CYP1A2, to roflumilast N-oxide, which is active with a potency 2- to 3-fold less than roflumilast and has the same selectivity for PDE4 isoenzymes.[13] Roflumilast N-oxide is thought to be the main contributor to total PDE4 inhibitory activity.[11]
* After a short-term intravenous infusion of roflumilast, the roflumilast plasma clearance was ≈9.6 L/hour.[11] After administration of radiolabelled roflumilast, approximately 70% of radioactivity is detected in the urine as inactive metabolites of roflumilast and roflumilast N-oxide.[21] After oral administration of roflumilast 500 μg once daily, the geometric mean apparent terminal plasma half-life values for roflumilast and roflumilast N-oxide were 18.0 and 21.2 hours[19] (corresponding median values of ≈17 and ≈30 hours after an oral dose are cited in the summary of product characteristics).[11]
* In the population pharmacokinetic study, a two-compartment model that included first-order absorption and a lag time was adequate in characterizing roflumilast pharmacokinetics; a one-compartment model with zero order absorption was adequate to characterize roflumilast N-oxide pharmacokinetics.[21]
Special Populations
* In patients with mild (Child-Pugh A) and moderate liver cirrhosis (Child-Pugh B) [n = 8 each], total PDE4 inhibitory activity was increased by ≈20% and ≈90%.[11] Roflumilast is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) and should be used with caution in patients with mild hepatic impairment (Child-Pugh A).[11]
* In patients with severe renal impairment (creatinine clearance 10–30 mL/min [0.6–1.8 L/h]), total PDE4 inhibitory activity was decreased by 9%, indicating that no dosage adjustment is necessary for patients with renal impairment.[11]
* In the population pharmacokinetic study, total PDE4 inhibitory activity following administration of roflumilast was positively associated with common adverse events, such as diarrhoea, nausea and headache.[21] Various covariates, including COPD, cigarette smoking, sex, age and race, influenced roflumilast and roflumilast N-oxide total PDE4 inhibitory activity.[21] Total PDE4 inhibitory activity was increased in elderly persons, females and non-Caucasians, and slightly decreased in smokers. These changes were not considered to be clinically important, although a combination of these factors could increase roflumilast exposure.[11]
Drug Interactions
* Interactions with drugs that are substrates, inducers or inhibitors of various CYP enzymes are possible.[11] Coadministration of roflumilast with fluvoxamine (a CYP1A2 inhibitor), cimetidine and enoxacin (CYP3A4/CYP1A2 inhibitors) increased total PDE4 inhibitory activity by 59%, 47% and 25%, respectively. There was a 9% increase in total PDE4 inhibitory activity when roflumilast was coadministered with erythromycin and ketoconazole (CYP3A4 inhibitors). Roflumilast coadministered with any of these substances might lead to increased roflumilast exposure and ongoing intolerability, under which circumstances roflumilast treatment would need reassessment.[11]
* In vitro studies indicate that roflumilast and roflumilast N-oxide have no inhibitory effects on CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4, CYP3A5, CYP4A9 or CYP4A11 and is unlikely to have clinically relevant interactions with drugs that are substrates of these enzymes.[11] Roflumilast was a weak inducer of CYP2B6, did not induce CYP1A2, CYP2A6, CYP2C9, CYP2C19, CYP3A4 or CYP3A5 and is unlikely to effect the metabolism of drugs that are substrates of these enzymes.[11]
* When roflumilast was coadministered with rifampicin (rifampin) [an inducer of several CYP enzymes], total PDE4 inhibitory activity was reduced by ≈60%.[11] The efficacy of roflumilast could be reduced if coadministered with potent CYP enzyme inducers, such as rifampicin, phenytoin, phenobarbitone and carbemazepine.[11]
* There was an 8% increase in total PDE4 inhibitory activity when roflumilast was coadministered with theophylline.[11]
* There were no clinically important interactions when roflumilast was coadministered with salbutamol, digoxin, oral montelukast, budesonide, formoterol, warfarin, midazolam and sildenafil.[11]
* Roflumilast and N-oxide roflumilast absorption and pharmacokinetics were not altered when roflumilast was administered with magnesium hydroxide or aluminium hydroxide-containing antacids.[11]
3. Therapeutic Efficacy
Randomized, double-blind, multinational trials in patients with COPD evaluated the efficacy of oral roflumilast (M2-107,[22] M2-112,[23] M2-124,[18] M2-125[18]) and oral roflumilast used concomitantly with inhaled long-acting bronchodilators, the β2-adrenergic receptor agonist salmeterol (M2-127)[24] and the muscarinic receptor antagonist tiotropium bromide (M2-128).[24] Fully published data from these trials are presented in this section.
Included patients were aged ≥40 years and current or former smokers (≥10[22-24] or ≥20[18] pack-years [1 pack-year was defined as 20 cigarettes per day for 1 year]). Patients were required to have moderate to severe (M2-107,[22] M2-127 and M2-128[24]) or severe (M2-112,[23] M2-124 and M2-125[18]) COPD. Spirometry inclusion criteria were FEV1 ≤80% predicted and FEV1/forced vital capacity (FEV1/FVC) <0.70 for moderate to severe COPD, and FEV1 ≤50% predicted and FEV1/FVC <0.70 for severe COPD. Across trials, patients were also required to have stable disease[22-24] or no current unresolved COPD exacerbation.[18] M2-124 and M2-125 also required chronic cough and sputum production and at least one recorded exacerbation requiring hospitalization or systemic corticosteroids in the previous year.[18] M2-128 required chronic cough and sputum production and frequent use of short-acting β2-adrenergic receptor agonists.[24]
Exclusion criteria included COPD exacerbation in the prior 4 weeks,[18,22-24] regular use of more than eight puffs of rescue medication per day,[22] need for long-term oxygen therapy,[22,23] asthma or other relevant lung disease,[18,22,24] known α-1-antitrypsin deficiency[18,22-24] and other relevant medical conditions or treatments unrelated to COPD.[18,23,24]
Patients were randomized to roflumilast 250 μg, roflumilast 500 μg or placebo once daily for 24 weeks (M2-107),[22] roflumilast 500 μg or placebo once daily for 52 weeks (M2-112,[23] M2-124,[18] M2-125[18]), roflumilast 500 μg plus salmeterol or placebo plus salmeterol for 24 weeks (M2-127[24]), and roflumilast 500 μg plus tiotropium bromide or placebo plus tiotropium bromide for 24 weeks (M2-128[24]). Before randomization, patients were required to meet the following criteria during a 4-week placebo run-in: clinically stable;[18,23] no moderate or severe COPD exacerbation;[24] post-bronchodilator FEV1 30–80% predicted;[22] total cough and sputum scores 1 week before randomization of >14;[18] negative faecal haemoccult test;[18] and compliant,[23] or took ≥80%[18,24] or >80% and <125%[22] of placebo, during the run-in.
Across trials, patients were able to receive concomitant short-acting β2-adrenergic receptor agonists as a rescue medication, and systemic corticosteroids if required for exacerbations.[18,22-24] Patients could also continue respiratory drugs at stable daily dosages as follows: M2-107 allowed short-acting anticholinergics;[22] M2-112 allowed short-acting anticholinergics and inhaled corticosteroids;[23] and M2-124 and M2-125 allowed short-acting anticholinergics and long-acting β2-adrenergic receptor agonists.[18] M2-127 and M2-128 did not allow concomitant respiratory drugs other than rescue medications.[24]
The primary endpoints were mean changes from baseline in the post-bronchodilator FEV1 at 24 (M2-107[22]) or 52 (M2-112[23]) weeks, mean change in pre-bronchodilator FEV1 from baseline to each post-randomization visit (M2-124,[18] M2-125,[18] M2-127,[24] M2-128[24]), health-related quality of life (HR-QOL) as measured by the St George's Respiratory Questionnaire[25] total score at 24 weeks (M2-107[22]), and mean rate of moderate or severe exacerbations per patient per year (M2-112,[23] M2-124,[18] M2-125[18]). Secondary endpoints included further spirometry outcomes,[18,22-24] time to exacerbations,[18] time to death,[18] mean Transition Dyspnoea Index[26] focal score (a measure of change in dyspnoea)[18,24] and mean change in the Shortness of Breath Questionnaire[27] score.[24] As similar results were observed for the FEV1 and other spirometry endpoints,[18,22-24] other spirometry findings are not discussed.
Primary efficacy analyses were performed in modified intent-to-treat populations that included all patients who received at least one dose of randomized treatment (M2-124,[18] M2-125,[18] M2-127,[24] M2-128,[24] M2-107,[22] M2-112[23]) and who had at least one post-baseline assessment (M2-107,[22] M2-112[23]), using last-observation-carried-forward imputation (M2-107,[22] M2-112[23]). A pooled analysis was also performed using data from the M2-124 and M2-125 trials.[18] Sensitivity analyses to test the robustness of results were conducted in M2-124,[18] M2-125,[18] M2-127[24] and M2-128.[24]
In the roflumilast and placebo groups, the mean/median age of patients ranged from 63 to 65 years, 64–81% were male and the mean number of cigarette pack-years ranged from 42 to 49.[18,22-24] See table I for baseline FEV1 values. Roflumilast versus Placebo

* In all trials, oral roflumilast 500 μg once daily produced significantly greater improvements in pre- and post-bronchodilator FEV1 than placebo (table I).[18,22,23] The mean increase in pre-bronchodilator and post-bronchodilator FEV1 with roflumilast ranged from 9 to 49 mL and 12 to 57 mL, whereas in all instances placebo recipients had a decrease or a negligible 8 mL increase in FEV1 over the trial period (table I). In the pooled analysis (M2-124 and M2-125 trials), the mean changes in pre-bronchodilator FEV1 were significantly higher in roflumilast than placebo groups (40 vs −9 mL; p < 0.0001).[18] Where performed, sensitivity analyses showed the findings for primary endpoints to be robust with regard to missing data and dropouts.[18]
* Prespecified subgroup analyses indicated that the effects of roflumilast on lung function were not related to concomitant corticosteroid treatment or smoking status (current vs former smoker).[23]
* In the M2-107, M2-124 and M2-125 trials, roflumilast groups had significantly lower exacerbation rates than placebo groups over 24[22] and 52[18] weeks of roflumilast treatment when compared with placebo (table I). In the pooled analysis of the M2-124 and M2-125 trials, there was a 17% lower COPD exacerbation rate in roflumilast than placebo groups (p = 0.0003).[18]
* In the M2-112 trial, there was no significant difference between treatment groups in the exacerbation rate in the total population (table I), but in the GOLD stage IV subpopulation, roflumilast recipients had a significantly lower exacerbation rate than placebo recipients (1.01 vs 1.59 per patient per year; p = 0.024).[23]
* There were no significant differences between treatments in the median time to the first moderate or severe exacerbation in the M2-124 or M2-125 trials.[18] However, the median time to the second exacerbation was significantly longer in roflumilast than placebo recipients (172 vs 159 days, p = 0.0290, in M2-124, and 188 vs 144 days, p = 0.0214, in M2-125).[18] In the pooled analysis of the M2-124 and M2-125 trials, the median times to moderate or severe exacerbations were significantly longer in roflumilast than placebo recipients (80 vs 71 days to the first exacerbation, p = 0.0185; 177 vs 148 days to the second exacerbation, p = 0.0014).[18]
* Dyspnoea improved in roflumilast recipients over placebo recipients.[18] There was a significant (p < 0.05) but small difference between the roflumilast and placebo groups in the change from baseline in Transition Dyspnoea Index focus scores in the M2-124 and M2-125 trials.[18]
* In both the roflumilast and placebo groups in the M2-107 and M2-112 trials, there were significant (p < 0.05) improvements from baseline in HR-QOL, as measured by the change in the St George's Respiratory Questionnaire total score, although there was no significant difference between roflumilast and placebo groups.[22,23]
* Where reported, there was no significant difference in mortality between roflumilast and placebo groups.[18,23] In the M2-124 and M2-125 trials, there was no difference in time to death or in mortality rates, which were 2% (both groups) in M2-124 and 3% (both groups) in M2-125.[18] In the M2-112 trial, 1.6% of roflumilast and 2.6% of placebo recipients died during the study.[23]
Roflumilast Used Concomitantly with Long-Acting Bronchodilator versus Placebo and Long-Acting Bronchodilator
* Roflumilast was efficacious when used concomitantly with salmeterol or tiotropium bromide over a 6-month treatment period, indicating that the benefits of roflumilast plus long-acting bronchodilator were over and above those achieved with a long-acting bronchodilator alone (table I). In both trials, roflumilast plus long-acting bronchodilator recipients had significantly greater improvements in pre-bronchodilator (primary endpoint) and post-bronchodilator FEV1 than placebo plus long-acting bronchodilator recipients.[24]
* The mean increases in pre- and post-bronchodilator FEV1 with roflumilast plus long-acting bronchodilator groups were 39 mL and 68 mL in the M2-127 trial and 65 mL and 74 mL in the M2-128 trial, while the corresponding changes in placebo plus long-acting bronchodilator groups were −10 mL and 8 mL and −16 mL and −7 mL (table I). Sensitivity analyses showed the findings for primary endpoints to be robust with regard to missing data and dropouts.[24]
* There were no significant differences between treatment groups in exacerbation rates over the 24-week treatment period in either trial (table I).
* Findings on other exacerbation endpoints, however, provide further evidence that roflumilast plus long-acting bronchodilator can reduce exacerbations.[24] In the M2-127 trial, roflumilast plus salmeterol recipients had a significantly longer median time to the first moderate or severe exacerbation than placebo plus salmeterol recipients (83 vs 71 days; p = 0.0067) and a significantly lower proportion of patients with a moderate or severe exacerbation (11% vs 18%; p = 0.0015) or a mild, moderate or severe exacerbation (28% vs 34%; p = 0.0419). There was no difference between treatments in median time to first mild, moderate or severe exacerbation.[24]
* In the M2-128 trial, roflumilast plus tiotropium bromide recipients had a significantly longer median time to the first mild, moderate or severe exacerbation than placebo plus tiotropium bromide recipients (50 vs 37 days; p = 0.0264) and a significantly lower proportion of patients with a mild, moderate or severe exacerbation (22% vs 30%; p = 0.0169).[24] There was no difference between treatment groups in median time to first moderate or severe exacerbation, or in the proportion of patients with a moderate or severe exacerbation.[24]
* Dyspnoea improved in roflumilast recipients when it was used concomitantly with tiotropium bromide, but not when used concomitantly with salmeterol.[24] Compared with tiotropium bromide plus placebo, in the roflumilast plus tiotropium bromide group there were significant (p < 0.05) improvements in the Transition Dyspnoea Index score, Shortness of Breath Questionnaire score and in use of rescue medication in the M2-128 trial.[24] There were no significant differences between treatment groups on these endpoints in the M2-127 trial.[24]
Pharmacoeconomic Considerations
The M2-112,[23] M2-124[18] and M2-125[18] trials also collected healthcare resource use, and employment and health utility data for subsequent pharmacoeconomic analyses. Results of the M2-122 pharmacoeconomic analysis are reported; the M2-124 and M2-125 pharmacoeconomic analyses are not yet available.[28]
The prospective cost-effectiveness analysis from the M2-112 trial[28] used data collected over 52 weeks and considered cost effectiveness from both UK societal and National Health System (NHS) perspectives. In this trial, there was a relatively low exacerbation rate that was not significantly different between treatment groups. Costs were based on 2004 costing. In the societal analysis, costs included those of study drugs, concomitant and rescue medications, costs associated with hospitalization, emergency room visits, contacts with care providers and ambulance transport, and COPD-related productivity costs. Analysis from the perspective of the NHS excluded productivity costs.[28]
* From a UK societal perspective, the annual COPD-related costs in the roflumilast and placebo groups were €1637 and €1401, while from a UK NHS perspective these costs were €1418 and €1242.[28] The incremental cost of roflumilast per moderate or severe exacerbation avoided was €2356 when considered from a societal perspective and €1755 from a NHS perspective; corresponding values for roflumilast per additional patient with a relevant improvement in St George's Respiratory questionnaire total score were €4712 and €3510. At a willingness to pay to prevent an exacerbation of €0, €5000 and €50 000, the probabilities that roflumilast would be cost effective were 10%, >70% and 90%, respectively.[28] In patients with very severe COPD, a subgroup in which roflumilast significantly lowered the exacerbation rate, roflumilast dominated placebo for cost per moderate or severe exacerbation avoided and cost per additional patient with relevant improvement on the St George's Respiratory Questionnaire.[28]
* In sensitivity analyses, the findings were generally robust with respect to model assumptions regarding healthcare and productivity costs and whether or not the analyses were restricted to trial completers.[28]
* This data is based on a single multinational trial that assigned UK unit costs to health outcomes and resource use across countries, and, thus, is unlikely to reflect between-country differences in cost frameworks and healthcare systems. The study is also subject to other limitations common to pharmacoeconomic analyses, such as lack of long-term efficacy data and the effect of missing data.[28]
4. Tolerability
The tolerability of oral roflumilast was evaluated in the randomized, trials described in section 3. The findings reported in this section are chiefly from the M2-124 trial and from a pooled analysis of the M2-124 and M2-125 trials, in which roflumilast was administered for 52 weeks.[18] The findings from these trials were consistent with those of the other roflumilast randomized trials, including trials of roflumilast coadministered with long-acting bronchodilators.[22-24] Data are fully published[18,24] or available as abstracts.[29,30] Supplemental data are from the EU summary of product characteristics.[11]
* In the pooled analysis of the M2-124 and M2-125 trials, 67% and 62% of roflumilast and placebo recipients reported treatment-emergent adverse events; in the corresponding groups, 19% and 22% had serious adverse events.[18]
* In the roflumilast and placebo groups, the mean compliance with medications was 93% and 95% (M2-124) and 93% and 96% (M2-125).[18] In the pooled analysis, 14% and 11% of roflumilast and placebo recipients discontinued medications because of adverse events. During the first 12 weeks of treatment, 8% and 3% of roflumilast and placebo recipients withdrew because of adverse events (both trials); the subsequent probability of withdrawal because of adverse events was 9% for both roflumilast and placebo recipients (both trials).[18]
* In the M2-124 trial, the most frequently occurring adverse events (not related to COPD) occurring at a greater frequency in roflumilast than placebo recipients were weight loss, diarrhoea, nausea, back pain and influenza.[18] Treatment-emergent adverse events occurring in ≥2% of patients in at least one treatment group are shown in figure 1.[18] In general, these findings are consistent with those using pooled data from COPD clinical studies, with the most frequent adverse events being diarrhoea, weight loss, nausea, abdominal pain and headache.[11] In the pooled data, most adverse events were of mild or moderate severity, occurred in the first weeks of treatment and resolved with continued treatment.[11] * Gastrointestinal adverse effects are considered to be potentially dose-limiting effects of PDE4 inhibitors.[4] In the M2-124 trial, diarrhoea occurred in 8% and 3% of roflumilast and placebo recipients, and nausea was reported in 5% and 2% in the corresponding groups.[18] Vomiting was infrequent, with a rate in the pooled analysis of 1% and <1% in roflumilast and placebo groups.[18] In the pooled analysis of M2-124 and M2-125, the most common adverse events (not related to COPD) that were associated with medication discontinuation were diarrhoea, nausea and headache.[18]
* In the M2-124 trial, weight loss was observed in 12% and 3% of roflumilast and placebo recipients.[18] In the pooled analysis of M2-124 and M2-125, the mean weight change after 1 year in roflumilast recipients was −2.09 kg versus an increase of 0.08 kg in placebo recipients.[18] In roflumilast recipients, weight change occurred chiefly in the first 6 months of treatment. Roflumilast recipients with diarrhoea, nausea, vomiting or headache had a 2.60 kg weight loss, whereas the weight loss was 2.02 kg in roflumilast recipients without these symptoms.[18] Obese patients (body mass index [BMI] >30) reported the largest absolute weight loss.[18] Roflumilast (n = 91) and placebo (n = 35) recipients with weight loss during the M2-124 and M2-125 trials, had 3.2 and 1.3 kg gains in weight after 12 weeks of post-therapy follow-up.[29]
* A similar pattern of weight loss over 24 weeks of treatment was observed with roflumilast plus a long-acting bronchodilator in the M2-127 and M2-128 trials; however, there were no significant between-treatment differences in weight loss across BMI category subgroups,[24] although a different pattern of decline in the fat-free mass index and BMI suggested that there was preferential loss of fat mass in roflumilast recipients.[30]
* In the pooled analysis, 1% and <1% of patients in the roflumilast and placebo groups developed atrial fibrillation, while cardiovascular events in general occurred in 7% and 8% of patients.[18] Holter monitoring of 33 and 22 roflumilast and placebo recipients revealed no between-group differences in cardiac rhythm disturbances.[18]
* In roflumilast recipients, there was no increase in pulmonary infections during the trial period, including pneumonia (data not reported).[18]
* In clinical studies, roflumilast has been associated with psychiatric disorders, including insomnia (1% to <10%), anxiety (0.1% to <1%), depression (0.01% to 0.1%) and nervousness (0.01% to 0.1%).[11] In rare instances, suicidal ideation and behaviours, including completed suicide, were reported.[11] Special precautions are required for patients who are at increased risk for psychiatric disorders or suicide (section 5).

5. Dosage and Administration
In the EU, oral roflumilast is approved as maintenance treatment for adult patients with severe COPD (post-bronchodilator FEV1 <50% predicted) associated with chronic bronchitis and a history of frequent exacerbations, as an add-on to bronchodilator therapy.[11] It is not indicated as a rescue medication for acute bronchospasm. In clinical trials, the maintenance effects of roflumilast in patients with COPD have been evaluated for up to 1 year.[11]
The recommended roflumilast dosage is a single 500 μg tablet administered once daily.[11] Roflumilast may need several weeks to achieve its effect. It can be swallowed with water, with or without food, and should be taken at the same time each day.[11]
Underweight patients receiving roflumilast require ongoing monitoring of bodyweight, and in the case of unexplained clinically relevant weight loss roflumilast should be discontinued and further monitoring of bodyweight undertaken.[11]
Roflumilast should not be used in patients with severe immunological disorders, severe acute infections, cancers other than basal cell carcinoma, or patients treated with immunosuppressive medicinal products, as there is a lack of clinical experience with roflumilast in these populations.[11] Patients receiving long-term oral corticosteroids should not receive roflumilast. There is limited experience in using roflumilast in patients with latent infections, including herpes zoster, herpes viral infection, viral hepatitis and tuberculosis.[11]
Careful assessment is required in patients with current psychiatric symptoms or a history of psychiatric symptoms and in patients receiving treatments that are likely to cause psychiatric events.[11] As rare instances of suicidal behaviour have been observed in clinical trials, patients should be instructed to report suicidal ideation or mood or behavioural changes to their prescriber. Roflumilast is not recommended for patients with a history of depression associated with suicidal ideation or behaviour.[11]
Roflumilast should not be coadministered with theophylline, as there are no clinical data supporting maintenance treatment with roflumilast used concomitantly with theophylline.[11]
Local prescribing information should be consulted for detailed information regarding contraindications, warnings and precautions, use in special populations and possible drug interactions.
6. Roflumilast: Current Status
Roflumilast is authorized in the EU for maintenance treatment, as an add-on to bronchodilator therapy, in adult patients with severe COPD (post-bronchodilator FEV1 <50% predicted) associated with chronic bronchitis and a history of frequent exacerbations.
In randomized, double-blind, multinational trials, oral roflumilast was efficacious in patients with moderate to severe and severe COPD, including when used concomitantly with long-acting bronchodilators. Compared with placebo, roflumilast produced small but significant increases from baseline in lung function, and in trials where it was used with or without concomitant long-acting bronchodilators, it significantly reduced the rate of COPD exacerbations. Roflumilast was generally well tolerated. Trials comparing roflumilast plus long-acting bronchodilator with inhaled corticosteroids plus long-acting bronchodilator in COPD would be of considerable interest.
Acknowledgements and Disclosures
The manuscript was reviewed by: A.D. D'Urzo, Department of Family and Community Medicine, University of Toronto, Toronto, Ontario, Canada; O.E. Della Pasqua, Leiden/Amsterdam Centre for Drug Research, Leiden, the Netherlands, and GlaxoSmithKline Research and Development, Stockley Park, Uxbridge, UK; J.A. Falk, Division of Pulmonary and Critical Care Medicine, Department of Medicine, Cedars-Sinai Medical Center, Los Angeles, California, USA; A.F. Gelb, Pulmonary Division, Department of Medicine, Lakewood Regional Medical Center, Lakewood, California, USA; J. Walters, Menzies Research Institute, University of Tasmania, Hobart, Tasmania, Australia.
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from any comments were made on the basis of scientific and editorial merit.
Correction to Online version of this article
Vol. 70, No. 12, 2010
Page 1623: The first sentence of the fourth paragraph has been corrected from the original Online version to read: “The mean increases in pre- and post-bronchodilator FEV1 with roflumilast plus long-acting bronchodilator groups were 39 mL and 68 mL in the M2-127 trial and 65 mL and 74 mL in the M2-128 trial, while the corresponding changes in placebo plus long-acting bronchodilator groups were −10 mL and 8 mL and −16 mL and −7 mL (table I).”
References
1. Yao H, de Boer W, Rahman I. Targeting lung inflammation: novel therapies for the treatment of COPD. Curr Respir Med Rev 2008; 4 (1): 57-68
2. Global Initiative for Chronic Obstructive Lung Disease (GOLD). At-a-glance outpatient reference guide for chronic obstructive pulmonary disease (COPD) [online]. Available from URL: http://www.goldcopd.com [Accessed 2010 May 20]
3. Wedzicha JA, Seemungal TA. COPD exacerbations: defining their cause and prevention. Lancet 2007 Sep 1; 370 (9589): 786-96
Cited Here... | PubMed | CrossRef
4. Hansel TT, Barnes JJ. New drugs for exacerbations of chronic obstructive pulmonary disease. Lancet 2009 Aug 29; 374 (9691): 744-55
Cited Here... | PubMed | CrossRef
5. Burgel P-R, Nesme-Meyer P, Chanez P, et al. Cough and sputum production are associated with frequent exacerbations and hospitalizations in COPD subjects. Chest 2009 Apr; 135 (4): 975-82
Cited Here... | PubMed | CrossRef
6. Barnes PJ, Celli BR. Systemic manifestations and comorbidities of COPD. Eur Respir J 2009; 33 (5): 1165-85
Cited Here... | PubMed | CrossRef
7. Halpin D. Mortality in COPD: inevitable or preventable? Insights from the cardiovascular arena. COPD 2008 Jun; 5 (3): 187-200
8. O'Donnell DE. Assessment of bronchodilator efficacy in symptomatic COPD: is spirometry useful? Chest 2000 Feb; 117 (2 Suppl.): 42s-7s
Cited Here... | PubMed | CrossRef
9. Celli BR, MacNee W. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. ATS/ERS Task Force. Eur Respir J 2004; 23: 932-46
Cited Here... | PubMed | CrossRef
10. Hatzelmann A, Schudt C. Anti-inflammatory and immunomodulatory potential of the novel PDE4 inhibitor roflumilast in vitro. J Pharmacol Exp Ther 2001; 297 (1): 267-79
11. European Medicines Agency. Daxas 500 micograms film-coated tablets: summary of product characteristics [online]. Available from URL: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/001179/WC500095209.pdf [Accessed 2010 Aug 2]
12. Nycomed. Nycomed and Forest Laboratories receive complete FDA response letter for roflumilast - no additional patient trials were requested [media release]. 2010 May 17 [online]. Available from URL: http://www.nycomed.com/News/NewsReleases/2010/Nycomed%20and%20Forest%20Laboratories%20receive%20complete%20FDA%20response%20letter%20for%20Roflumilast.aspx
13. Hatzelmann A, Morcillo EJ, Lungarella G, et al. The preclinical pharmacology of roflumilast: a selective, oral phosphodiesterase inhibitor in development for chronic obstructive pulmonary disease. Pulm Pharmacol Ther 2010 Aug; 23 (4): 235-56
Cited Here... | PubMed | CrossRef
14. Wohlsen A, Hirrle A, Tenor H, et al. Effect of cyclic AMP-elevating agents on airway ciliary beat frequency in central and lateral airways in rat precision-cut lung slices. Eur J Pharmacol 2010 Jun 10; 635 (1-3): 177-83
Cited Here... | PubMed | CrossRef
15. Martorana PA, Beume R, Lucattelli M, et al. Roflumilast fully prevents emphysema in mice chronically exposed to cigarette smoke. Am J Resp Crit Care Med 2005 Oct; 172 (7): 848-53
Cited Here... | PubMed | CrossRef
16. Hohlfeld JM, Schoenfeld K, Lavae-Mokhtari M, et al. Roflumilast attenuates pulmonary inflammation upon segmental endotoxin challenge in healthy subjects: a randomized placebo-controlled trial. Pulm Pharmacol Ther 2008 Aug; 21 (4): 616-23
Cited Here... | PubMed | CrossRef
17. Grootendorst DC, Gauw SA, Verhoosel RM, et al. Reduction in sputum neutrophil and eosinophil numbers by the PDE4 inhibitor roflumilast in patients with COPD. Thorax 2007 Dec; 62 (12): 1081-7
Cited Here... | View Full Text | PubMed | CrossRef
18. Calverley PM, Rabe KF, Goehring UM, et al. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet 2009 Aug 29; 374 (9691): 685-94
Cited Here... | PubMed | CrossRef
19. Bethke TD, Böhmer GM, Hermann R, et al. Dose-proportional intraindividual single- and repeated-dose pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor. J Clin Pharmacol 2007 Jan; 47 (1): 26-36
Cited Here... | View Full Text | PubMed | CrossRef
20. Hauns B, Hermann R, Hünnemeyer A, et al. Investigation of a potential food effect on the pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor, in healthy subjects. J Clin Pharmacol 2006 Oct; 46 (10): 1146-53
Cited Here... | View Full Text | PubMed | CrossRef
21. Lahu G, Hünnemeyer A, Diletti E, et al. Population pharmacokinetic modelling of roflumilast and roflumilast N-oxide by total phosphodiesterase-4 inhibitory activity and development of a population pharmacodynamic-adverse event model. Clin Pharmacokinet 2010; 49 (9): 589-606
Cited Here... | View Full Text | PubMed | CrossRef
22. Rabe KF, Bateman ED, O'Donnell D, et al. Roflumilast - an oral anti-inflammatory treatment for chronic obstructive pulmonary disease: a randomised controlled trial. Lancet 2005 Aug 13-2005; 366 (9485): 563-71
23. Calverley PM, Sanchez-Toril F, McIvor A, et al. Effect of 1-year treatment with roflumilast in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2007 Jul 15; 176 (2): 154-61
Cited Here... | PubMed | CrossRef
24. Fabbri LM, Calverley PM, Izquierdo-Alonso JL, et al. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with longacting bronchodilators: two randomised clinical trials. Lancet 2009 Aug 29; 374 (9691): 695-703
Cited Here... | PubMed | CrossRef
25. Jones PW, Quirk FH, Baveystock CM. The St Georges Respiratory Questionnnaire. Respir Med 1991 Sep; 85 Suppl. B: 25-31
Cited Here... | PubMed | CrossRef
26. Mahler DA, Weinberg DH, Wells CK, et al. The measurement of dyspnea: contents, interobserver agreement, and physiologic correlates of two clinical indices. Chest 1984 Jun; 85 (6): 751-8
Cited Here... | PubMed | CrossRef
27. Eakin EG, Resnikoff PM, Prewitt LM, et al. Validation of a new dyspnea measure: the UCSD Shortness of Breath Questionnaire. Chest 1998 Mar; 113 (3): 619-24
Cited Here... | PubMed | CrossRef
28. Rutten-van Mölken MP, van Nooten FE, Lindemann M, et al. A 1-year prospective cost-effectiveness analysis of roflumilast for the treatment of patients with severe chronic obstructive pulmonary disease. Pharmacoeconomics 2007; 25 (8): 695-711
Cited Here... | View Full Text | PubMed | CrossRef
29. Martinez FJ, Rabe KF, Wouters EFM, et al. Time course and reversibility of weight decrease with roflumilast, a phosphodiesterase 4 inhibitor [abstract no. A4441]. 2010 American Thoracic Society (ATS) International Conference; 2010 May 14-19; New Orleans (LA)
30. Wouters EFM, Teichmann P, Brose M, et al. Effects of roflumilast, a phosphodiesterase 4 inhibitor, on body composition in chronic obstructive pulmonary disease [abstract no. A4473]. 2010 American Thoracic Society (ATS) International Conference; 2010 May 14-19; New Orleans (LA)
Keywords:
Adis-Drug-Profiles; Chronic-obstructive-pulmonary-disease; Research-and-development; Roflumilast, general






 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 