Sepsis Overview
Sepsis is a clinical term used to describe symptomatic bacteremia, with or without organ dysfunction. This term is often misused and misapplied to patients with fever, leukocytosis, and hypotension due to other causes, including pseudosepsis. True sepsis is a common cause of hospitalization in the United States, including in elderly men who are more likely to develop urosepsis due to benign urinary tract obstruction caused by prostatic hypertrophy. Patients with diabetes, systemic lupus erythematosus (SLE), or alcoholism or who are taking steroids are also at an increased risk for bacteremia.
Sustained bacteremia, in contrast to transient bacteremia, may result in a sustained febrile response that may be associated with organ dysfunction. Septicemia refers to the active multiplication of bacteria in the bloodstream that results in an overwhelming infection.
For patient education information, see eMedicine's Blood and Lymphatic System Center, as well as Sepsis (Blood Infection).
Etiology and Pathophysiology of Sepsis
Sepsis is not a random occurrence and is usually associated with other conditions, such as perforation, compromise, or rupture of an intra-abdominal or pelvic structure.[1] Intrarenal infection (pyelonephritis), renal abscess (intrarenal or extrarenal), acute prostatitis, or prostatic abscess may cause urosepsis in immunocompetent hosts.
Sepsis or septic shock may be associated with the direct introduction of microbes into the bloodstream via intravenous (IV) infusion (eg, IV line, other device-associated infections). Meningococcemia from a respiratory source may also result in sepsis, with or without associated meningitis.
Bacteremia due to bacteruria (urosepsis) may complicate cystitis in compromised hosts, and sepsis may be caused by overwhelming pneumococcal infection in patients with impaired or absent splenic function.[2]
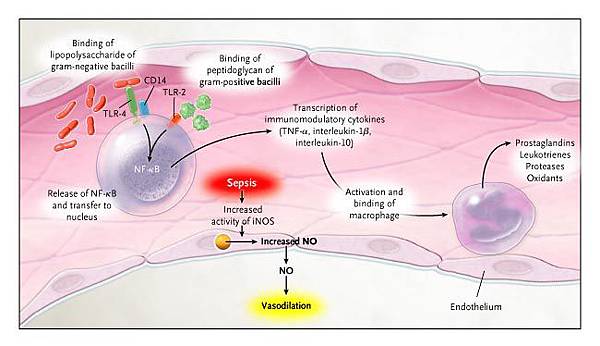
The pathophysiology of sepsis is complex and results from the effects of circulating bacterial products, mediated by cytokine release, caused by sustained bacteremia. Cytokines, previously termed endotoxins, are responsible for the clinically observable effects of the bacteremia in the host.[3, 4, 5] Impaired pulmonary, hepatic, or renal function may result from excessive cytokine release during the septic process.
Sepsis Evaluation
The patient’s history is essential in determining the likely source of the septic process. This, in turn, determines the appropriate antimicrobial therapy (see Management). In addition, the physical examination may be helpful in suggesting a potential source for sepsis.
Patients with nonspecific symptoms are usually acutely ill with fever, with or without shaking chills. Mental status may be impaired in the setting of fever or hypoperfusion. Patients with bacteremia from any source often display an increased breathing rate due to respiratory alkalosis. The skin of patients with sepsis may be warm or cold, depending on the adequacy of organ perfusion and dilatation of the superficial vessels of the skin.
IV line infections
An infected central IV line site immediately suggests the probable etiology.[6] However, it is important to note that only 50% of patients with central IV line infections have evidence of infection at the insertion site.
Suspect intravenous (IV)-line infections when other sources of sepsis are eliminated and the IV line has been in place for a prolonged period, usually longer than 1 week.[7, 8] Central IV lines are the lines most commonly associated with bacteremia or sepsis. Peripheral venous lines are almost never involved, and arterial lines are rarely associated with bacteremia, although it can still occur.
Gastrointestinal/genitourinary infections
As mentioned earlier, in many cases, the history is critical for diagnosis. Abdominal findings on physical examination may be unimpressive or absent.
Patients with an intra-abdominal or pelvic source of infection usually have a history of antecedent conditions that predispose to perforation or abscess (eg, chronic or retrocecal subacute appendicitis, diverticulitis, Crohn disease, previous abdominal surgery, cholecystitis).
Diffuse abdominal pain suggests pancreatitis (not sepsis) or generalized peritonitis, whereas right upper abdominal quadrant (RUQ) tenderness may suggest a gallbladder etiology (eg, cholecystitis, cholangitis), and tenderness in the right lower abdominal quadrant (RLQ) in a young adult suggests appendicitis or Crohn disease. Discrete tenderness over the left lower abdominal quadrant suggests diverticulitis, particularly in elderly patients.
A rectal examination may reveal exquisite tenderness due to a prostatic abscess or, more commonly, an enlarged noninflamed prostate suggestive of benign prostatic hypertrophy (BPH).
A urinary tract source is suggested by an antecedent history of pyelonephritis, stone disease, congenital abnormal collecting system, prostate enlargement, or previous prostate or renal surgery/procedures.[9, 10]
Costovertebral angle (CVA) tenderness with a temperature of 102°F defines acute pyelonephritis. Subacute or chronic pyelonephritis may manifest as only mild tenderness.
Diagnostic Considerations
Sepsis is not a random occurrence and is usually associated with other conditions (see Table 1 below)[11, 12, 13] ; the history is critical for the diagnosis and in determining the appropriate clinical management (see Sepsis Evaluation). Pseudosepsis, sepsis syndrome, and host immunocompetence also need to be considered. Table 1. Clinical Conditions Associated With Sepsis and Its Mimics
|
Associated With Sepsis |
Associated With Sepsis |
|
Gastrointestinal tract source |
Gastrointestinal tract source |
|
Genitourinary tract source |
Genitourinary tract source |
|
Pelvic source |
Upper respiratory tract source |
|
Lower respiratory tract source |
Lower respiratory tract source |
|
Intravascular source |
Skin/soft-tissue source |
|
Cardiovascular source |
Cardiovascular source |
|
Central nervous system source |
|
|
Adapted from: Cunha BA, Shea KW. Fever in the intensive care unit. Infect Dis Clin North Am. Mar 1996;10(1):185-209.[11] |
|
Pseudosepsis
Before embarking on a workup for sepsis or beginning empiric antibiotics, it is vital to first rule out the treatable causes of pseudosepsis early in the disease process. Consider other causes or conditions that mimic the clinical and hemodynamic parameters of sepsis (see Table 2 below). The causes of pseudosepsis need identification because they require supportive, rather than antimicrobial, therapy.
Pseudosepsis is a common cause of misdiagnosis in hospitalized patients, particularly in the emergency department and in medical and surgical intensive care units. The most common causes of pseudosepsis include gastrointestinal (GI) hemorrhage, pulmonary embolism, acute myocardial infarction (MI), acute pancreatitis (edematous or hemorrhagic), diuretic-induced hypovolemia, and relative adrenal insufficiency.
Patients with pseudosepsis may have fever, chills, leukocytosis, and a left shift, with or without hypotension. All causes of pseudosepsis produce Swan-Ganz catheter readings that are compatible with sepsis (eg, increased cardiac output, decreased peripheral resistance), which could misdirect the unwary clinician.
Table 2. Septic Syndromes, Noninfectious Conditions Mimicking Sepsis
|
Clinical Presentations |
Hemodynamic Parameters |
|
Hemorrhage |
Acute pancreatitis |
|
Pulmonary embolism |
Anaphylaxis |
|
Myocardial infarction |
Spinal cord injury |
|
Pancreatitis |
Adrenal insufficiency |
|
Diabetic (abdominal crisis) ketoacidosis |
|
|
Systemic lupus erythematosus flare with abdominal crisis |
|
|
Ventricular pseudoaneurysm |
|
|
Massive aspiration/atelectasis |
|
|
Systemic vasculitis |
|
|
Diuretic-induced hypovolemia |
Sepsis syndrome versus sepsis
Sepsis syndrome is the systemic response to infection in which there is evidence of abnormalities in organ perfusion and/or function. Sepsis is the condition in which microorganisms/pathogens are present in the blood or plasma, with or without organ dysfunction. See Table 3 below, for a comparison between the 2 conditions.
Table 3. Sepsis Syndrome Versus Sepsis
|
Sepsis Syndrome: |
Sepsis: Bacteremia From |
|
|
Parameters |
No definite source PLUS |
Proper identification/ process/ source PLUS |
|
Microbiologic |
Positive buffy coat smear result OR |
|
|
Hemodynamic |
⇓ Peripheral vascular disease (PVR) |
|
|
⇑ Cardiac output (CO) |
Left ventricular dilatation |
|
|
Laboratory |
⇓ Fibrinogen |
⇓ White blood cells (WBCs) |
|
⇓ Globulins |
⇓ Platelets |
|
|
⇓ Albumin |
||
|
Negative blood |
⇑ Fibrin split products |
|
|
Clinical |
≤102°F ± |
≥102°F |
|
GI = gastrointestinal; GU = genitourinary; PT/PTT = prothrombin time/partial thromboplastin time. |
||
Host immunocompetence
Otherwise healthy hosts with community-acquired pneumonia virtually never present with hypotension or sepsis; however, patients with decreased or absent splenic function may present with overwhelming pneumococcal sepsis. If an otherwise healthy patient with community-acquired pneumonia presents with shock and all of the other causes of pseudosepsis are ruled out, then it must be assumed that the patient is a compromised host with impaired or absent splenic function.
Blood and Bacterial Cultures
Obtain blood cultures in all patients upon admission to demonstrate the organism responsible for infection. Negative blood culture results are also necessary to include pseudosepsis in the differential diagnosis (see Diagnostic Considerations).[14]
Blood culture isolates might suggest the underlying disease process. Bacteroides fragilis suggests a colonic or pelvic source, whereas Klebsiella species or enterococci suggest a gallbladder or urinary tract source more frequently than an intra-abdominal source.
If central intravenous-line sepsis is suspected, remove the line and send the tip for semiquantitative bacterial culture. If the catheter-tip culture results are positive and demonstrate 15 or more colonies and if the catheter-tip isolate matches the blood-culture isolate, an infection associated with the central intravenous line is diagnosed.
Nasal colonization with methicillin-resistant S aureus (MRSA) is often viewed as a potential marker of subsequent risk for severe MRSA infection. However, a meta-analysis found nasal colonization with MRSA to be a poor predictor for the subsequent occurrence of MRSA lower respiratory tract infections and MRSA bloodstream infections that require antimicrobial treatment.[15] Thus, caution should be exercised in interpreting nasal cultures in patients in the intensive care unit (ICU).
Gram Staining
Obtain a buffy coat of the white cells from peripheral blood stained by acridine orange,[16] or use the Gram method to demonstrate bacteria responsible for the bacteremia or septic process. Although the yield is low, stained buffy coat smears, if positive, are the best rapid tests available to demonstrate organisms that cause bacteremia. If the stained buffy coat smear yields a positive result, it demonstrates the morphology of the bacteria that is causing the bacteremia, which provides rapid information on which to base empiric antimicrobial therapy.
Complete Blood Cell Count
A complete blood cell (CBC) count is usually not helpful because of the numerous conditions that mimic sepsis (eg, pseudosepsis) and that manifest as leukocytosis with variable degrees of a left shift (see Diagnostic Considerations). Leukocytosis with a left shift is a nonspecific diagnostic finding that is as common in noninfection as in infection.
Urine Studies
Obtain a urine Gram stain, urinalysis, and urine culture if urosepsis is suspected.
Chest Radiology
Chest radiography is important to rule out pneumonia and diagnose other causes of pulmonary infiltrates, such as the following:
- Pulmonary drug reactions
- Pulmonary emboli
- Pulmonary hemorrhage
- Primary or metastatic pulmonary neoplasms
- Lymphangitic spread of malignancies
- Large pleural effusions
- Pneumothorax
- Hydrothorax
- Fluid overload
- Congestive heart failure
- Acute MI
- Acute respiratory distress syndrome – This condition should suggest an intra-abdominal source (eg, acute pancreatitis).
Abdominal Ultrasonography
Perform abdominal ultrasonography if biliary tract obstruction is suspected based on the clinical presentation. However, abdominal ultrasonography is suboptimal for the detection of abscesses or perforated hollow organs.
Ultrasonograms in patients with cholecystitis may show a thickened gallbladder wall or biliary calculi but no dilatation of the common bile duct. Stones in the biliary tract are visible in patients with cholangitis, but the common bile duct is dilated.
Abdominal CT Scanning or MRI
Use computed tomography (CT) scanning or MRI of the abdomen if a nonbiliary intra-abdominal source of infection is suspected based on the history or physical examination findings. These 2 imaging modalities are superior to ultrasonography in demonstrating all lesions, except those related to the biliary tract.
An abdominal CT scan or MRI is also helpful in delineating intrarenal and extrarenal pathology.
Nuclear Imaging
Gallium or indium scanning has no place in the initial workup of sepsis, as patients with sepsis are acutely ill by definition, and rapid diagnostic tests (eg, computed tomography [CT] scanning or magnetic resonance imaging [MRI] of the abdomen, ultrasonography of the right upper quadrant) are time-critical, life-saving tools.[17]
Cardiac Studies
Obtain an electrocardiograph (ECG) and cardiac enzyme levels in patients in whom an acute myocardial infarction (MI) is likely. Remember that certain patients may present with a silent, asymptomatic MI, which should be included in the differential diagnosis of otherwise unexplained fever, leukocytosis, and hypotension. Silent MIs are common in elderly patients and in those who have undergone recent abdominal or pelvic surgery. They are also common in individuals with alcoholism, diabetes, and uremic conditions.
Thoracentesis/Paracentesis
Perform thoracentesis for diagnostic purposes in patients with substantial pleural effusion. Perform paracentesis in patients with gross ascites.
Swan-Ganz Catheterization
Use data obtained via a Swan-Ganz catheter to manage the fluid status of the patient and to assess left ventricular dysfunction in patients with acute myocardial infarction (MI).
Do not use Swan-Ganz hemodynamic parameters to diagnose sepsis. Although most patients with sepsis demonstrate an increased cardiac output with a low peripheral vascular resistance, the converse is not true. Most patients with Swan-Ganz readings compatible with sepsis without a definite intravenous abdominal or genitourinary source do not have sepsis, but rather pseudosepsis, as described in Diagnostic Considerations.
Histologic Findings
Histologic examination is generally unhelpful, as sepsis does not cause diagnostic findings in various organs.
Procalcitnonin levels
A study by Karlsson et al assessed the use of procalcitonin (PCT) levels as a predictor of survival in 242 adult patients with severe sepsis and septic shock.[18] Patients with septic shock had higher PCT concentrations than patients without. Although PCT levels did not differ between survivors and nonsurvivors (P = 0.64 and P = 0.99, respectively), mortality was lower in patients whose PCT levels decreased more than 50% (by 72 h) compared with those who had a decrease of less than 50% (12.2% vs 29.8%; P = 0.007).
A separate study from van Nieuwkoop et al examined the use of PCT levels in predicting bacteremia.[19] The study examined 581 patients, 136 of whom had bacteremia. PCT levels successfully identified 94-99% of patients with bacteremia. Use of PCT as a single decision tool may help reduce blood cultures by as much as 40%.
Overview of Sepsis Management
Early and aggressive medical therapy is indicated in patients with suspected sepsis.[20, 21, 22, 23, 24, 25, 26, 27, 28, 29] However, it is crucial to consider pseudosepsis as a cause of the presenting syndrome complex, as most causes of pseudosepsis are readily treatable and reversible if recognized and treated early. Patients with pseudosepsis require supportive therapy rather than antimicrobial treatment.
Patients with sepsis are generally ill and require bed rest or admission to the intensive care unit (ICU) for monitoring and treatment. Admission to an ICU or surgical ICU depends on the severity of the septic process and organ dysfunction, as well as the need for surgical intervention. Transfer to a facility able to perform diagnostic imaging tests or required surgical procedures if they are not available at the admitting hospital may be necessary.
Determine the likely source of the infection, and administer intravenous, empiric antimicrobial agents until culture results are available, when more narrow-spectrum agents can be used (see Antimicrobial Management). In addition, offer supportive therapy aimed at maintaining organ perfusion, and provide respiratory support, when necessary.[24, 30, 31]
Most patients are instructed to take nothing by mouth (NPO) and can tolerate a transient decrease in caloric intake over 1-2 weeks if their fluid and electrolyte balances are maintained.
Consultations
Obtain a consultation with a surgeon for patients with presumed intra-abdominal or pelvic sepsis. Early surgical consultation and involvement by the surgical team is essential, because many causes of sepsis involve a perforated viscus, abscess, or obstructing process that requires surgical intervention for cure or resolution of the infection. For example, peritonitis may result in abscesses, which may subsequently need to be drained. Inadequate correction of intra-abdominal perforation or drainage procedures may result in a continuance or relapse of the patient's septic condition.
Also obtain a consultation with an infectious disease specialist for all patients with sepsis included in the differential diagnosis (see Diagnostic Considerations).[23]
Surgical Intervention
Evaluation by a surgeon for patients with presumed intra-abdominal or pelvic sepsis is essential, as many causes of sepsis involve a perforated viscus, abscess, or obstructing process that requires surgical intervention for cure or resolution of the infection. The procedures used are dependent on the source of the infection, the severity of the sepsis, the patient’s clinical status, among other factors in individual scenarios.
Antimicrobial Management
Appropriate antimicrobial therapy depends on adequate coverage of the resident flora of the organ system presumed to be the source of the septic process.[20, 21, 22, 23, 32] Empiric monotherapy regimens include imipenem, meropenem, cefoperazone, piperacillin/tazobactam, or sulbactam/ampicillin. Combination therapeutic agents include clindamycin or metronidazole plus levofloxacin, aztreonam, trimethoprim/sulfamethoxazole (TMP-SMZ), or an aminoglycoside.
No single drug/regimen is superior to another. Alternative agents may be used alone or in combination, provided they have a low reactive potential and a good adverse-effect profile.[20, 21, 22, 23]
Antibiotics are normally continued until the septic process and surgical interventions have controlled the source of infection. Ordinarily, patients are treated for approximately 2 weeks.
As soon as patients are able to tolerate medications orally, the patient may be switched to an equivalent regimen of antibiotics by mouth in an intravenous (IV)-to-oral switch program.
A Cochrane Database Systemic Review investigated the potential use of de-escalation to replace empirical broad-spectrum antimicrobial treatment with a narrower antimicrobial therapy.[33] This process involves either changing the agent or discontinuing combination treatment according to the patient’s microbial culture results. Despite examining 436 studies, the review determined that no adequate, direct evidence concludes whether de-escalation is effective or safe for adults with sepsis, severe sepsis, or septic shock. Further research using randomized, controlled trials is needed.
Empiric therapy for IV-line infections
Because IV-line infections are most often due to Staphylococcus aureus (methicillin-sensitive S aureus [MSSA] or methicillin-resistant S aureus [MRSA]) and less commonly due to aerobic gram-negative bacilli, the preferred empiric therapy for IV-line infections is meropenem, cefoperazone, or cefepime plus additional coverage for staphylococci.[7, 8] If MRSA is prevalent in the institution, add linezolid, vancomycin, or daptomycin.
If coagulase-negative staphylococci are recovered from the blood (high-level bacteremia; ie, 3/4 or 4/4 blood cultures positive), avoid vancomycin for empiric therapy if possible, as this is a low-virulence organism.
Treatment of coagulase-negative staphylococcal central-line infection requires removal of the line. Vancomycin may be given, but central line removal is essential. If the central line cannot be removed for clinical reasons in a patient with MRSA or coagulase-negative staphylococcal infection, empiric suppressive vancomycin therapy is acceptable.
Minimize the use of vancomycin in order to prevent the emergence of Enterococcus faecium, a vancomycin-resistant species.[7]
Empiric therapy for biliary tract infections (cholecystitis/cholangitis)
The main biliary-tract pathogens include Escherichia coli, Klebsiella species, and Enterococcus faecalis. Coverage for staphylococci and anaerobes is not needed in the biliary tract. Anaerobes are important only in patients with diabetes who have Clostridium perfringens emphysematous cholecystitis. Preferred monotherapy for biliary-tract infections is with imipenem, meropenem, piperacillin, or cefoperazone.
Empiric therapy for intra-abdominal and pelvic infections
The main pathogens in the lower abdomen and pelvis include aerobic coliform gram-negative bacilli and B fragilis. Enterococci are permissive/opportunistic pathogens and do not require special coverage. Potent anti– B fragilis and aerobic gram-negative bacillary coverage are essential, in addition to surgical intervention when drainage or repair of intra-abdominal viscera is required.
Preferred monotherapy for intra-abdominal and pelvic infections is imipenem, meropenem, piperacillin/tazobactam, or ampicillin/sulbactam.
Preferred combination therapy for intra-abdominal and pelvic infections is clindamycin or metronidazole plus aztreonam, levofloxacin, or an aminoglycoside.
Empiric therapy for urosepsis
The primary uropathogens include gram-negative aerobic bacilli, such as coliforms or enterococci (E faecalis, not E faecium vancomycin-resistant enterococci). Pseudomonas aeruginosa, Enterobacter species, and Serratia species are rare uropathogens and are associated with urologic instrumentation.
Preferred monotherapy for urosepsis due to aerobic gram-negative bacilli is with aztreonam, levofloxacin, third- or fourth-generation cephalosporins, or an aminoglycoside. However, preferred monotherapy for urosepsis due to enterococci (E faecalis) is with ampicillin or vancomycin (penicillin-allergic).
Empiric therapy for community-acquired urosepsis is levofloxacin, aztreonam, or an aminoglycoside plus ampicillin. For nosocomial urosepsis, piperacillin, imipenem, or meropenem monotherapy is preferred.
Empiric therapy for S aureus, pneumococcal, and meningococcal sepsis
S aureus sepsis is usually associated with infection caused by devices or acute bacterial endocarditis. Empiric therapy may be with nafcillin, an anti-staphylococcal, cephalosporin, a carbapenem, linezolid, or clindamycin with or without rifampin.
Pneumococcal or meningococcal sepsis may be treated with penicillin G or a beta-lactam. In patients with associated meningococcal meningitis, the antibiotic selected should penetrate the cerebrospinal fluid and should be given in meningeal doses.
Empiric therapy for sepsis of unknown origin
The usual sources of sepsis are from the distal gastrointestinal (GI) tract, pelvis, or genitourinary (GU) tract. For IV-line infections, see above.
Organisms that should be covered from the GI/GU tract and pelvis include aerobic gram-negative bacilli (coliforms) and B fragilis. Enterococci are important pathogens in biliary - tract sepsis and urosepsis.
Preferred empiric monotherapy includes meropenem, imipenem, piperacillin/tazobactam, or cefoperazone.
Empiric combination therapy includes the following:
- Levofloxacin plus either clindamycin or metronidazole
- Aztreonam
- Cefepime plus either clindamycin or metronidazole, or
- Ceftriaxone or aminoglycoside plus metronidazole
Outpatient Management
If orally administered antibiotics are continued at home, advise the patient about possible adverse effects. If additional antimicrobial therapy is needed outside the hospital setting, it should be given orally, not intravenously. Do not allow the total course of antibiotics to exceed 3 weeks, except for the treatment of liver abscesses, which may require prolonged courses of oral antibiotics for cure or complete clinical resolution.
Coordinate surgical follow-up with the surgeon.
Prognosis
Sepsis is a common cause of mortality and morbidity worldwide. The prognosis of sepsis depends on the underlying status and host defenses, prompt and adequate surgical drainage of abscesses, relief of any obstruction of the intestinal or urinary tract, and appropriate and early empiric antimicrobial therapy with the drug spectrum appropriate to the presumed septic source. Thus, early and appropriate empiric antimicrobial therapy and surgical intervention are critical in decreasing mortality and morbidity.
The prognosis in most patients is good, except in those with intra-abdominal or pelvic abscesses due to organ perforation. The underlying physiologic condition of the host is the primary determinant of outcome.
A systematic review by Winters et al suggested that, beyond the standard 28-day in-hospital mortality endpoint, ongoing mortality rates in patients with sepsis remain elevated up to 2 years and beyond.[34] In addition, survivors consistently demonstrate impaired quality of life.[15]
Special Considerations
The most important medicolegal concerns regarding sepsis treatment include the following:
- Ensuring that the patient indeed does have sepsis
- Rapidly identifying its source
- Implementing effective treatments.
The most common error is failure to consider pseudosepsis as a cause of the presenting syndrome complex. Most causes of pseudosepsis are readily treatable and reversible if recognized and treated early (see Diagnostic Considerations).
Elderly or pregnant patients
Elderly patients may present with peritonitis and may not experience rebound tenderness of the abdomen.[35] An acute surgical abdomen in a pregnant patient may be difficult to diagnose, but, fortunately, most pregnant women are young, healthy, and physiologically strong.[36] The most common cause of sepsis in pregnancy is urosepsis due to an obstructed urinary tract, which may be caused by the hormone effects of pregnancy on the ureters (hydroureters) and the mechanical obstructing effect of the uterus impinging on the ureters.[36]





 留言列表
留言列表
 線上藥物查詢
線上藥物查詢 